INTRODUCTION
METHODS
Study population
Covariates
Study design and definition of diseases
Statistical analysis
RESULTS
Baseline characteristics
Table 1.
| Characteristic | 40 years | 66 years | P value |
|---|---|---|---|
| Number | 2,905,752 | 1,239,569 | |
| Male sex | 1,439,587 (49.5) | 560,882 (45.3) | <0.0001 |
| BMI, kg/m2 | 23.54±3.36 | 24.17±2.99 | <0.0001 |
| WC, cm | 78.69±9.43 | 82.55±8.15 | <0.0001 |
| Height, cm | 165.61±8.44 | 158.95±8.29 | <0.0001 |
| Body weight, kg | 64.91±12.46 | 61.16±9.44 | <0.0001 |
| Systolic blood pressure, mm Hg | 118.47±13.74 | 127.78±15.15 | <0.0001 |
| Diastolic blood pressure, mm Hg | 74.67±10.04 | 77.76±9.69 | <0.0001 |
| Fasting blood glucose, mg/dL | 93.75±15.86 | 97.87±16.2 | <0.0001 |
| Total cholesterol, mg/dL | 193.56±34.54 | 199.55±37.74 | <0.0001 |
| HDL-C, mg/dL | 56.34±16.74 | 54.35±17.08 | <0.0001 |
| LDL-C, mg/dL | 112.76±35.96 | 119.26±39.13 | <0.0001 |
| TG, mg/dL | 105.8 (105.72–105.87) | 117.13 (117.02–117.23) | <0.0001 |
| Current smokinga | 804,080 (27.7) | 155,769 (12.6) | <0.0001 |
| Alcohol drinkingb | 233,057 (8.0) | 58,234 (4.7) | <0.0001 |
| Physically activec | 479,820 (16.5) | 304,313 (24.6) | <0.0001 |
| Low incomed | 522,199 (18.0) | 297,638 (24.0) | <0.0001 |
| HTN | 334,549 (11.5) | 618,878 (49.9) | <0.0001 |
| Dyslipidemia | 338,285 (11.6) | 425,781 (34.4) | <0.0001 |
| CKDe | 67,008 (2.3) | 127,682 (10.3) | <0.0001 |
| Abdominal obesityf | 459,249 (15.8) | 355,044 (28.6) | <0.0001 |
| General obesityg | 879,501 (30.3) | 456,224 (36.8) | <0.0001 |
Values are presented as number (%), mean±standard deviation, or geometric mean (95% confidence interval).
BMI, body mass index; WC, waist circumfer ence; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TG, triglyceride; HTN, hypertension; CKD, chronic kidney disease.
a Current smoking: those who were smoking at the time of interview and who had smoked >100 cigarettes in their lifetime,
Risk of DM development according to age
Table 2.
| Variable | Number | DM | Incidence rate, 1,000 person-years | Model 1 | Model 2 | Model 3 | |||
|---|---|---|---|---|---|---|---|---|---|
| 40-year-old group | |||||||||
| BMI (per 1) | 1.251 (1.249–1.252) | 1.249 (1.247–1.251) | 1.214 (1.212–1.216) | ||||||
| General obesitya (BMI ≥25 kg/m2) | |||||||||
| No | 2,026,251 | 26,827 | 2.2984 | 1 (ref) | 1 (ref) | 1 (ref) | |||
| Yes | 879,501 | 56,766 | 11.6298 | 4.412 (4.347–4.479) | 4.402 (4.336–4.468) | 3.566 (3.512–3.622) | |||
| BMI, kg/m2 | |||||||||
| <18.5 | 116,685 | 580 | 0.8815 | 0.617 (0.568–0.671) | 0.605 (0.556–0.657) | 0.622 (0.572–0.676) | |||
| 18.5–22.9 | 1,266,476 | 11,245 | 1.5377 | 1 (ref) | 1 (ref) | 1(ref) | |||
| 23–24.9 | 643,090 | 15,002 | 4.0534 | 2.333 (2.276–2.391) | 2.355 (2.298–2.414) | 2.203 (2.149–2.258) | |||
| 25–29.9 | 759,698 | 40,725 | 9.5598 | 5.256 (5.145–5.370) | 5.269 (5.157–5.383) | 4.419 (4.324–4.516) | |||
| ≥30 | 119,803 | 16,041 | 25.8282 | 14.947 (14.587–15.317) | 14.761 (14.404–15.126) | 10.351 (10.091–10.617) | |||
| P for trend | <0.001 | <0.001 | <0.001 | ||||||
| WC (per 5) | 1.591 (1.585–1.596) | 1.584 (1.578–1.589) | 1.490 (1.484–1.495) | ||||||
| Abdominal obesityb (WC ≥90/85 cm) | |||||||||
| No | 2,446,503 | 45,278 | 3.2215 | 1 (ref) | 1 (ref) | 1 (ref) | |||
| Yes | 459,249 | 38,315 | 15.3379 | 4.164 (4.107–4.222) | 4.104 (4.047–4.162) | 3.231 (3.184–3.278) | |||
| WC (men/women), cm | |||||||||
| <80/75 | 1,292,629 | 10,527 | 1.4104 | 0.236 (0.230–0.242) | 0.236 (0.230–0.242) | 0.269 (0.262–0.275) | |||
| 80–84.9/75–79.9 | 665,176 | 15,252 | 3.9902 | 0.586 (0.574–0.598) | 0.588 (0.576–0.601) | 0.629 (0.615–0.642) | |||
| 85–89.9/80–84.9 | 488,698 | 19,499 | 7.0419 | ||||||
| 90–94.9/85–89.9 | 268,210 | 16,912 | 11.3561 | 1.588 (1.555–1.621) | 1.576 (1.544–1.609) | 1.465 (1.436–1.496) | |||
| 95–99.9/90–94.9 | 118,578 | 10,817 | 16.9227 | 2.401 (2.346–2.458) | 2.367 (2.312–2.423) | 2.043 (1.995–2.092) | |||
| ≥100/95 | 72,461 | 10,586 | 28.6412 | 4.203 (4.105–4.304) | 4.110 (4.014–4.209) | 3.225 (3.148–3.304) | |||
| P for trend | <0.0001 | <0.0001 | <0.0001 | ||||||
| 66-year-old group | |||||||||
| BMI (per 1) | 1.134 (1.132–1.136) | 1.137 (1.135–1.139) | 1.117 (1.115–1.119) | ||||||
| General obesitya (BMI ≥25 kg/m2) | |||||||||
| No | 783,345 | 59,369 | 13.7884 | 1 (ref) | 1 (ref) | 1 (ref) | |||
| Yes | 456,224 | 63,135 | 25.968 | 1.902 (1.880–1.923) | 1.933 (1.911–1.955) | 1.739 (1.719–1.759) | |||
| BMI, kg/m2 | |||||||||
| <18.5 | 28,362 | 1,480 | 9.3677 | 0.801 (0.760–0.844) | 0.770 (0.731–0.811) | 0.841 (0.798–0.886) | |||
| 18.5–22.9 | 407,126 | 26,089 | 11.5967 | 1 (ref) | 1 (ref) | 1 (ref) | |||
| 23–24.9 | 347,857 | 31,800 | 16.7541 | 1.449 (1.426–1.473) | 1.481 (1.456–1.505) | 1.383 (1.360–1.406) | |||
| 25–29.9 | 415,689 | 54,590 | 24.5226 | 2.136 (2.104–2.167) | 2.194 (2.162–2.227) | 1.945 (1.916–1.974) | |||
| ≥30 | 40,535 | 8,545 | 41.6512 | 3.738 (3.648–3.831) | 3.817 (3.724–3.912) | 3.155 (3.078–3.235) | |||
| P for trend | <0.001 | <0.001 | <0.001 | ||||||
| WC (per 5) | 1.294 (1.289–1.298) | 1.296 (1.292–1.301) | 1.254 (1.250–1.259) | ||||||
| Abdominal obesityb (WC ≥90/85 cm) | |||||||||
| No | 884,525 | 69,546 | 14.3296 | 1 (ref) | 1 (ref) | 1 (ref) | |||
| Yes | 355,044 | 52,958 | 28.1141 | 1.980 (1.957–2.002) | 1.991 (1.968–2.014) | 1.799 (1.778–1.820) | |||
| WC (men/women), cm | |||||||||
| <80/75 | 279,645 | 14,852 | 9.6115 | 0.509 (0.499–0.519) | 0.500 (0.490–0.510) | 0.554 (0.543–0.565) | |||
| 80–84.9/79.9 | 293,367 | 22,826 | 14.1726 | 0.750 (0.738–0.763) | 0.747 (0.734–0.759) | 0.775 (0.762–0.788) | |||
| 85–89.9/80–84.9 | 311,513 | 31,868 | 18.7736 | ||||||
| 90–94.9/85–89.9 | 206,276 | 26,829 | 24.211 | 1.291 (1.270–1.312) | 1.292 (1.271–1.313) | 1.252 (1.232–1.273) | |||
| 95–99.9/90–94.9 | 98,510 | 15,901 | 30.6169 | 1.649 (1.618–1.680) | 1.648 (1.616–1.679) | 1.550 (1.521–1.580) | |||
| ≥100/95 | 50,258 | 10,228 | 39.9227 | 2.173 (2.125–2.222) | 2.164 (2.116–2.213) | 1.975 (1.931–2.020) | |||
| P for trend | <0.0001 | <0.0001 | <0.0001 | ||||||
Risk of DM development was expressed as hazard ratio with (95% confidence interval). Model 1 was adjusted for age and sex; Model 2 was adjusted for age, sex, smoking status, alcohol consumption, physical activity, and income level; Model 3 was adjusted for age, sex, smoking status, alcohol consumption, physical activity, income, hypertension, dyslipidemia, and chronic kidney disease.
BMI, body mass index; WC, waist circumference; DM, diabetes mellitus.
Fig. 1.
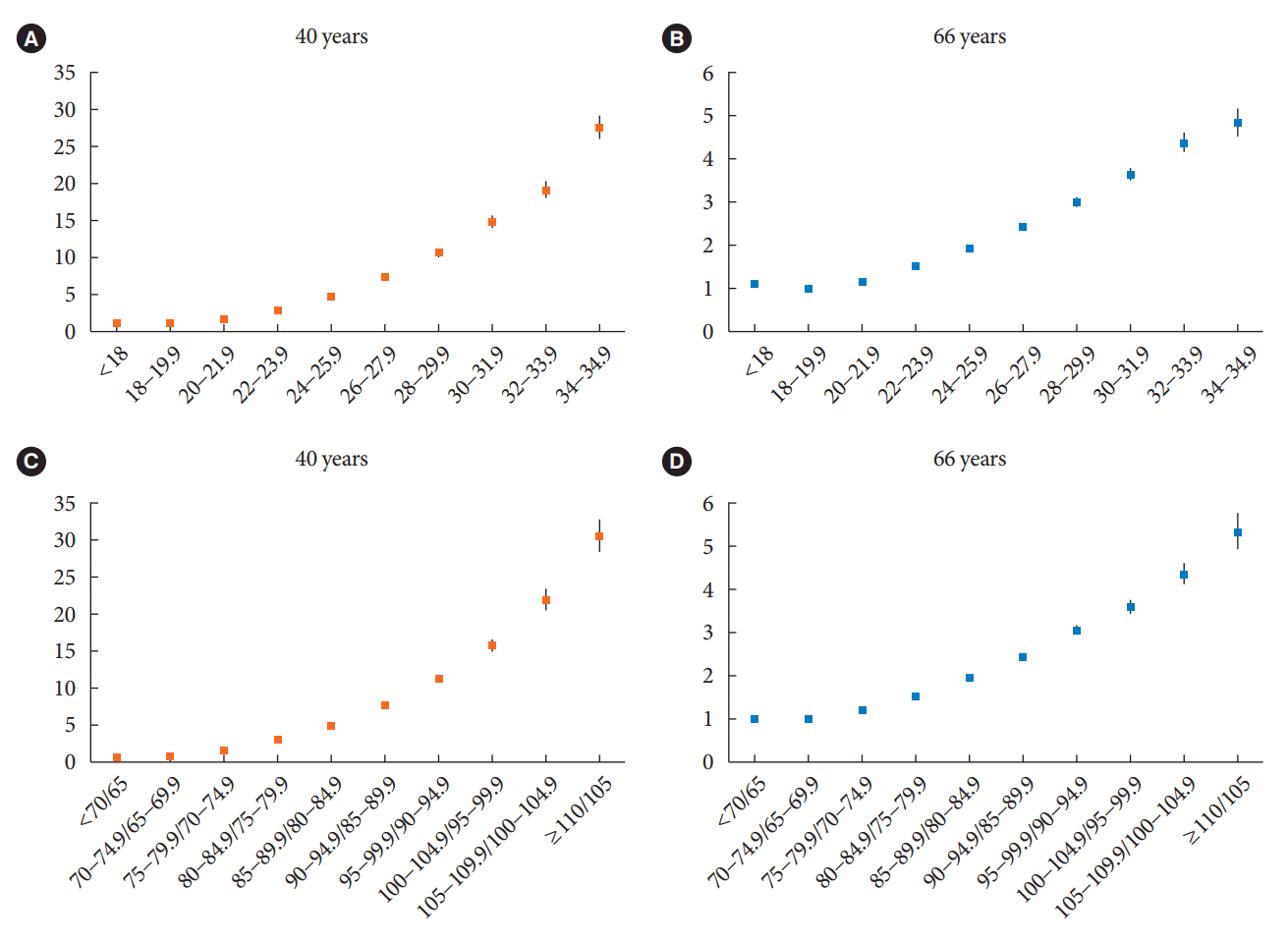
Table 3.
| Variable | Number | DM | Incidence rate, 1,000 person-year | Model 1 | Model 2 | Model 3 | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| 40-year-old group | ||||||||||
| BMI (per 1) | ||||||||||
| Men | 1.243 (1.241–1.246) | 1.242 (1.239–1.244) | 1.205 (1.202–1.208) | |||||||
| Women | 1.259 (1.257–1.262) | 1.257 (1.254–1.260) | 1.219 (1.216–1.222) | |||||||
| General obesitya (BMI ≥25 kg/m2) | ||||||||||
| Men | ||||||||||
| No | 840,370 | 17,418 | 3.6125 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 599,217 | 42,040 | 12.7192 | 3.540 (3.478–3.603) | 3.538 (3.476–3.601) | 2.881 (2.829–2.934) | ||||
| Women | ||||||||||
| No | 1,185,881 | 9,409 | 1.3735 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 280,284 | 14,726 | 9.3448 | 6.819 (6.645–6.998) | 6.777 (6.604–6.955) | 5.256 (5.117–5.400) | ||||
| BMI, kg/m2 | ||||||||||
| Men | ||||||||||
| <18.5 | 25,549 | 282 | 1.9191 | 0.750 (0.666–0.845) | 0.718 (0.637–0.809) | 0.766 (0.680–0.863) | ||||
| 18.5–22.9 | 436,903 | 6,453 | 2.5621 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 23–24.9 | 377,918 | 10,683 | 4.9551 | 1.937 (1.878–1.998) | 1.969 (1.909–2.031) | 1.807 (1.752–1.864) | ||||
| 25–29.9 | 525,991 | 31,133 | 10.6246 | 4.168 (4.058–4.282) | 4.201 (4.090–4.316) | 3.463 (3.370–3.558) | ||||
| ≥30 | 73,226 | 10,907 | 29.0887 | 11.555 (11.205–11.917) | 11.459 (11.111–11.818) | 7.944 (7.694–8.202) | ||||
| P for trend | <0.0001 | <0.0001 | <0.0001 | |||||||
| Women | ||||||||||
| <18.5 | 91,136 | 298 | 0.5831 | 0.589 (0.524–0.662) | 0.581 (0.517–0.653) | 0.598 (0.532–0.672) | ||||
| 18.5–22.9 | 829,573 | 4,792 | 0.9995 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 23–24.9 | 265,172 | 4,319 | 2.7953 | 2.788 (2.675–2.905) | 2.791 (2.679–2.908) | 2.588 (2.483–2.697) | ||||
| 25–29.9 | 233,707 | 9,592 | 7.2135 | 7.221 (6.975–7.476) | 7.193 (6.948–7.447) | 5.939 (5.733–6.152) | ||||
| ≥30 | 46,577 | 5,134 | 20.8608 | 21.110 (20.295–21.959) | 20.787 (19.983–21.623) | 13.851 (13.287–14.440) | ||||
| P for trend | <0.001 | <0.001 | <0.001 | |||||||
| WC (per 5) | ||||||||||
| Men | 1.549 (1.542–1.556) | 1.541 (1.534–1.548) | 1.446 (1.439–1.453) | |||||||
| Women | 1.653 (1.644–1.662) | 1.647 (1.639–1.656) | 1.539 (1.530–1.548) | |||||||
| Abdominal obesityb (WC ≥90/85 cm) | ||||||||||
| Men | ||||||||||
| No | 1,126,905 | 31,287 | 4.8632 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 312,682 | 28,171 | 16.6361 | 3.446 (3.391–3.501) | 3.392 (3.338–3.447) | 2.722 (2.676–2.768) | ||||
| Women | ||||||||||
| No | 1,319,598 | 13,991 | 1.8357 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 146,567 | 10,144 | 12.6061 | 6.912 (6.737–7.091) | 6.817 (6.645–6.994) | 4.991 (4.858–5.128) | ||||
| WC, cm | ||||||||||
| Men | ||||||||||
| <80 | 419,056 | 5,657 | 2.3466 | 0.290 (0.281–0.299) | 0.289 (0.280–0.298) | 0.337 (0.326–0.347) | ||||
| 80–84.9 | 386,738 | 11,009 | 4.977 | 0.615 (0.600–0.631) | 0.618 (0.603–0.634) | 0.661 (0.645–0.677) | ||||
| 85–89.9 | 321,111 | 14,621 | 8.0745 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 90–94.9 | 187,252 | 12,954 | 12.5149 | 1.555 (1.518–1.592) | 1.542 (1.506–1.578) | 1.439 (1.405–1.473) | ||||
| 95–99.9 | 80,393 | 8,076 | 18.7646 | 2.343 (2.280–2.408) | 2.305 (2.244–2.369) | 2.007 (1.953–2.063) | ||||
| ≥100 | 45,037 | 7,141 | 31.334 | 3.944 (3.834–4.057) | 3.849 (3.741–3.959) | 3.064 (2.977–3.154) | ||||
| P for trend | <0.0001 | <0.0001 | <0.0001 | |||||||
| Women | ||||||||||
| <75 | 873,573 | 4,870 | 0.9638 | 0.189 (0.182–0.197) | 0.190 (0.183–0.198) | 0.218 (0.210–0.227) | ||||
| 75–79.9 | 278,438 | 4,243 | 2.6347 | 0.517 (0.496–0.538) | 0.518 (0.497–0.540) | 0.555 (0.532–0.578) | ||||
| 80–84.9 | 167,587 | 4,878 | 5.0906 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 85–89.9 | 80,958 | 3,958 | 8.7149 | 1.717 (1.647–1.791) | 1.709 (1.639–1.782) | 1.574 (1.510–1.642) | ||||
| 90–94.9 | 38,185 | 2,741 | 13.1263 | 2.598 (2.479–2.722) | 2.573 (2.456–2.697) | 2.154 (2.055–2.258) | ||||
| ≥95 | 27,424 | 3,445 | 24.3105 | 4.846 (4.640–5.063) | 4.762 (4.559–4.975) | 3.508 (3.355–3.669) | ||||
| P for trend | <0.001 | <0.001 | <0.001 | |||||||
| 66-year-old group | ||||||||||
| BMI (per 1) | ||||||||||
| Men | 1.127 (1.124–1.130) | 1.135 (1.131–1.138) | 1.114 (1.111–1.117) | |||||||
| Women | 1.139 (1.136–1.142) | 1.139 (1.136–1.141) | 1.118 (1.116–1.121) | |||||||
| General obesitya (BMI ≥25 kg/m2) | ||||||||||
| Men | ||||||||||
| No | 372,352 | 31,461 | 15.4186 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 188,530 | 27,002 | 27.2769 | 1.768 (1.739–1.797) | 1.823 (1.793–1.853) | 1.653 (1.626–1.681) | ||||
| Women | ||||||||||
| No | 410,993 | 27,908 | 12.32 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 267,694 | 36,133 | 25.069 | 2.033 (2.002–2.065) | 2.036 (2.004–2.068) | 1.819 (1.790–1.848) | ||||
| BMI, kg/m2 | ||||||||||
| Men | ||||||||||
| <18.5 | 14,978 | 980 | 11.7237 | 0.884 (0.829–0.943) | 0.841 (0.789–0.898) | 0.907 (0.850–0.968) | ||||
| 18.5–22.9 | 195,316 | 14,323 | 13.2616 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 23–24.9 | 162,058 | 16,158 | 18.4277 | 1.389 (1.358–1.421) | 1.438 (1.406–1.471) | 1.347 (1.317–1.378) | ||||
| 25–29.9 | 178,104 | 24,681 | 26.3015 | 1.981 (1.941–2.023) | 2.077 (2.034–2.120) | 1.854 (1.816–1.894) | ||||
| ≥30 | 10,426 | 2,321 | 45.0361 | 3.388 (3.242–3.540) | 3.558 (3.405–3.718) | 2.974 (2.844–3.109) | ||||
| P for trend | <0.0001 | <0.0001 | <0.0001 | |||||||
| Women | ||||||||||
| <18.5 | 13,384 | 500 | 6.7206 | 0.668 (0.611–0.731) | 0.655 (0.599–0.716) | 0.724 (0.662–0.792) | ||||
| 18.5–22.9 | 211,810 | 11,766 | 10.0595 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 23–24.9 | 185,799 | 15,642 | 15.317 | 1.522 (1.486–1.559) | 1.529 (1.493–1.566) | 1.428 (1.394–1.462) | ||||
| 25–29.9 | 237,585 | 29,909 | 23.2263 | 2.307 (2.258–2.356) | 2.315 (2.266–2.365) | 2.042 (1.998–2.087) | ||||
| ≥30 | 30,109 | 6,224 | 40.5156 | 4.023 (3.901–4.148) | 4.013 (3.891–4.138) | 3.279 (3.179–3.383) | ||||
| P for trend | <0.001 | <0.001 | <0.001 | |||||||
| WC (per 5) | ||||||||||
| Men | 1.277 (1.270–1.283) | 1.284 (1.277–1.291) | 1.244 (1.237–1.250) | |||||||
| Women | 1.307 (1.301–1.313) | 1.306 (1.300–1.312) | 1.262 (1.256–1.268) | |||||||
| Abdominal obesityb (WC ≥90/85 cm) | ||||||||||
| Men | ||||||||||
| No | 415,340 | 35,776 | 15.7715 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 145,542 | 22,687 | 29.7736 | 1.886 (1.854–1.917) | 1.910 (1.879–1.943) | 1.736 (1.707–1.766) | ||||
| Women | ||||||||||
| No | 469,185 | 33,770 | 13.0643 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| Yes | 209,502 | 30,271 | 26.9868 | 2.064 (2.032–2.096) | 2.059 (2.027–2.091) | 1.852 (1.823–1.881) | ||||
| WC, cm | ||||||||||
| Men | ||||||||||
| <80 | 133,927 | 8,219 | 11.053 | 0.544 (0.529–0.558) | 0.528 (0.514–0.542) | 0.581 (0.566–0.597) | ||||
| 80–84.9 | 141,580 | 12,255 | 15.861 | 0.780 (0.762–0.799) | 0.773 (0.755–0.792) | 0.801 (0.782–0.820) | ||||
| 85–89.9 | 139,833 | 15,302 | 20.3443 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 90–94.9 | 91,309 | 12,585 | 26.0027 | 1.278 (1.248–1.308) | 1.282 (1.252–1.312) | 1.245 (1.216–1.275) | ||||
| 95–99.9 | 37,933 | 6,599 | 33.6331 | 1.651 (1.604–1.700) | 1.656 (1.609–1.705) | 1.565 (1.520–1.611) | ||||
| ≥100 | 16,300 | 3,503 | 42.8295 | 2.104 (2.028–2.182) | 2.104 (2.028–2.182) | 1.935 (1.865–2.008) | ||||
| P for trend | <0.0001 | <0.0001 | <.0001 | |||||||
| Women | ||||||||||
| <75 | 145,718 | 6,633 | 8.2743 | 0.473 (0.460–0.486) | 0.471 (0.458–0.485) | 0.523 (0.508–0.538) | ||||
| 75–79.9 | 151,787 | 10,571 | 12.6156 | 0.720 (0.703–0.738) | 0.720 (0.703–0.738) | 0.748 (0.730–0.767) | ||||
| 80–84.9 | 171,680 | 16,566 | 17.5238 | 1 (ref) | 1 (ref) | 1 (ref) | ||||
| 85–89.9 | 114,967 | 14,244 | 22.8216 | 1.302 (1.273–1.332) | 1.300 (1.271–1.330) | 1.257 (1.229–1.286) | ||||
| 90–94.9 | 60,577 | 9,302 | 28.7856 | 1.642 (1.601–1.684) | 1.636 (1.595–1.678) | 1.532 (1.493–1.572) | ||||
| ≥95 | 33,958 | 6,725 | 38.5595 | 2.200 (2.139–2.264) | 2.186 (2.124–2.248) | 1.977 (1.922–2.034) | ||||
| P for trend | <0.001 | <0.001 | <0.001 | |||||||
Risk of DM development was expressed as hazard ratio with (95% confidence interval). Model 1 was adjusted for age, sex; Model 2 was adjusted for age, sex, smoking status, alcohol consumption, physical activity, and income level; Model 3 was adjusted for age, sex, smoking status, alcohol consumption, physical activity, income, hypertension, and dyslipidemia.
BMI, body mass index; WC, waist circumference; DM, diabetes mellitus.
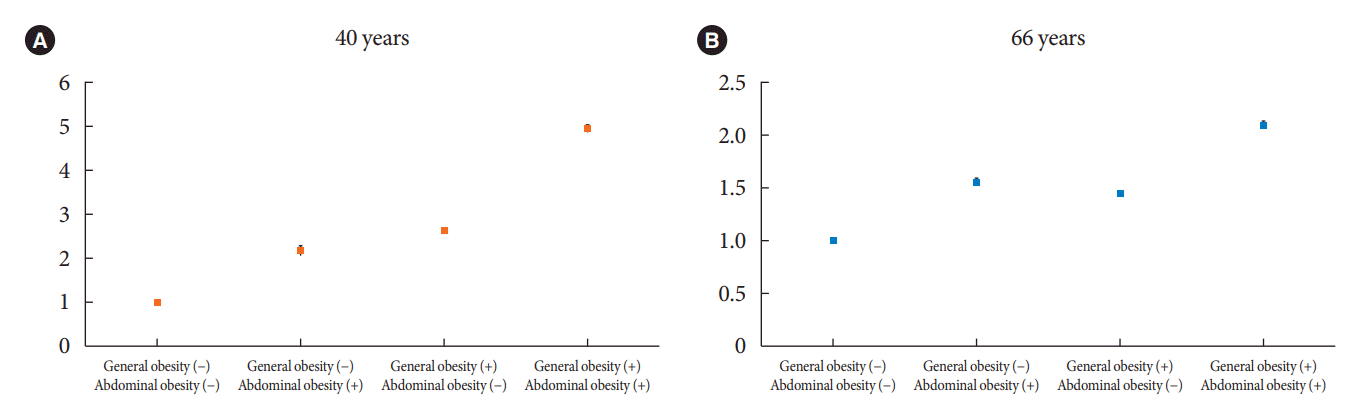
Risk of DM development according to the presence or absence of general and abdominal obesity
Fig. 2.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download