Abstract
Objectives
Methods
Results
Conclusion
Notes
This study was partially supported by a research grant from Hanmi Pharmaceutical Co. Ltd. (research ID: HM-IIT-ESO-004). This was an investigator-initiated study, and the funding company had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. No other potential conflicts of interest relevant to this article were reported.
AUTHOR CONTRIBUTIONS
Conceptualization: all authors. Methodology: SKK, EJJ, JHS, DIS, SMJ, BJL, ISP, JGC, YHP. Software: SKK, SJP, YHP. Validation: all authors. Investigation: SKK, EJJ, JHS, DIS, SMJ, BJL, ISP, JGC, YHP. Formal analysis: SKK, SJP, YHP. Data curation: SKK, SJP. Project administration: SKK, YHP. Funding acquisition: SKK, YHP. Writing–original draft: SKK, SJP, YHP. Writing–review & editing: all authors.
ACKNOWLEDGMENTS
Supplementary materials
Supplementary Material 2.
Supplementary Material 3.
REFERENCES
Fig. 1.
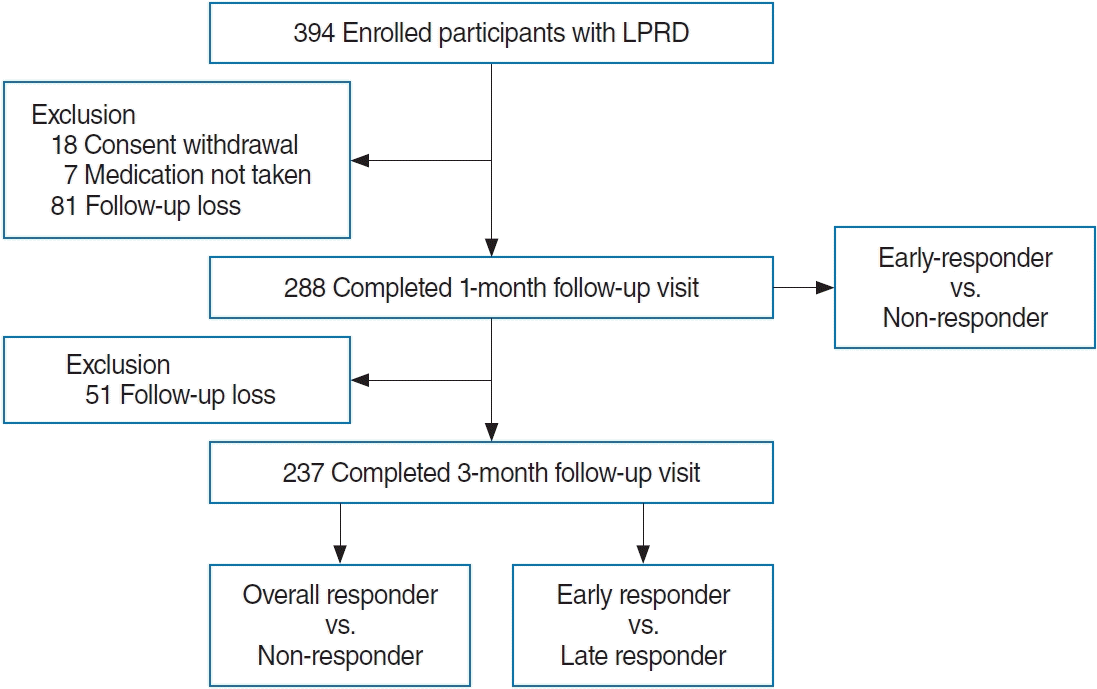
Table 1.
| Factor | Responder (n=109, 37.85%) | Non-responder (n=179, 62.15%) | Total (n=288) | P-value | ||||
|---|---|---|---|---|---|---|---|---|
| Clinical characteristics | ||||||||
| Sex | Male | 45 (41.28) | 79 (44.13) | 124 (43.06) | 0.636 | |||
| Female | 64 (58.72) | 100 (55.87) | 164 (56.94) | |||||
| Age (yr) | Average±SD | 54.51±11.45 | 55.40±12.47 | 55.07±12.08 | 0.346 | |||
| Median | 55 | 56 | 56 | |||||
| Range | 21–82 | 19–88 | 19–88 | |||||
| BMI change (kg/m2) | Average±SD | −0.13±0.83 | −0.01±1.03 | −0.05±0.96 | 0.074 | |||
| Median | 0 | 0 | 0 | |||||
| Range | −6.41–1.67 | −9.83–7.17 | −9.83–7.17 | |||||
| GERD history | (−) | 74 (67.89) | 100 (55.87) | 174 (60.42) | 0.043* | |||
| (+) | 35 (32.11) | 79 (44.13) | 114 (39.58) | |||||
| LPRD family history | (−) | 85 (77.98) | 152 (84.92) | 237 (82.29) | 0.135 | |||
| (+) | 24 (22.02) | 27 (15.08) | 51 (17.71) | |||||
| Pre-treatment RSI | Average±SD | 15.84±4.02 | 16.81±4.12 | 16.44±4.10 | 0.012* | |||
| Median | 14 | 15 | 15 | |||||
| Range | 13–34 | 13–35 | 13–35 | |||||
| Pre-treatment RFS | Average±SD | 11.51±2.76 | 11.00±2.48 | 11.19±2.60 | 0.117 | |||
| Median | 12 | 11 | 11 | |||||
| Range | 7–18 | 7–18 | 7–18 | |||||
| Number of concurrent medications | Average±SD | 1.88±3.64 | 3.78±6.60 | 3.03±5.73 | 0.007* | |||
| Median | 0 | 1 | 1 | |||||
| Range | 0–20 | 0–42 | 0–42 | |||||
| Hospital anxiety and depression scale (HAD) | ||||||||
| HAD-A total | Average±SD | 6.63±3.21 | 7.16±3.39 | 6.96±3.32 | 0.212 | |||
| Median | 6 | 7 | 6 | |||||
| Range | 1–16 | 2–17 | 1–17 | |||||
| HAD-A presence | (−) | 71 (65.14) | 103 (57.54) | 174 (60.42) | 0.201 | |||
| (+) | 38 (34.86) | 76 (42.46) | 114 (39.58) | |||||
| HAD-D total | Average±SD | 7.21±3.05 | 7.54±3.05 | 7.41±3.05 | 0.389 | |||
| Median | 7 | 7 | 7 | |||||
| Range | 0–15 | 0–16 | 0–16 | |||||
| HAD-D presence | (−) | 57 (52.29) | 91 (50.84) | 148 (51.39) | 0.811 | |||
| (+) | 52 (47.71) | 88 (49.16) | 140 (48.61) | |||||
| Treatment compliance | ||||||||
| PPI medication (%) | Average±SD | 98.82±5.78 | 98.27±6.13 | 98.48±6.00 | 0.274 | |||
| Median | 100 | 100 | 100 | |||||
| Range | 53.57–100 | 53.57–100 | 53.57–100 | |||||
| Sleep habitsa) | (−) | 66 (60.55) | 129 (72.07) | 195 (67.71) | 0.043* | |||
| (+) | 43 (39.45) | 50 (27.93) | 93 (32.29) | |||||
| Dietary habit 1b) | (−) | 49 (44.95) | 75 (41.90) | 124 (43.06) | 0.612 | |||
| (+) | 60 (55.05) | 104 (58.10) | 164 (56.94) | |||||
| Dietary habit 2c) | (−) | 51 (46.79) | 89 (49.72) | 140 (48.61) | 0.629 | |||
| (+) | 58 (53.21) | 90 (50.28) | 148 (51.39) | |||||
| Dietary habit 3d) | (−) | 55 (50.46) | 89 (49.72) | 144 (50.00) | 0.903 | |||
| (+) | 54 (49.54) | 90 (50.28) | 144 (50.00) | |||||
| Initial alcohol consumption | (−) | 68 (62.39) | 88 (49.16) | 156 (54.17) | 0.029* | |||
| (+) | 41 (37.61) | 91 (50.84) | 132 (45.83) | |||||
| Alcohol restriction | (−) | 80 (73.39) | 118 (65.92) | 198 (68.75) | 0.185 | |||
| (+) | 29 (26.61) | 61 (34.08) | 90 (31.25) | |||||
| Initial smoking | (−) | 89 (81.65) | 131 (73.18) | 220 (76.39) | 0.101 | |||
| (+) | 20 (18.35) | 48 (26.82) | 68 (23.61) | |||||
| Smoking restriction | (−) | 98 (89.91) | 155 (86.59) | 253 (87.85) | 0.404 | |||
| (+) | 11 (10.09) | 24 (13.41) | 35 (12.15) | |||||
| Past and current medical history | ||||||||
| Past medical history | (−) | 104 (95.41) | 168 (93.85) | 272 (94.44) | 0.576 | |||
| (+) | 5 (4.59) | 11 (6.15) | 16 (5.56) | |||||
| SOC | ||||||||
| Cardiac | (−) | 109 (100.00) | 176 (98.32) | 285 (98.96) | 0.292 | |||
| (+) | 0 | 3 (1.68) | 3 (1.04) | |||||
| Pulmonary | (−) | 108 (99.08) | 178 (99.44) | 286 (99.31) | 1.000 | |||
| (+) | 1 (0.92) | 1 (0.56) | 2 (0.69) | |||||
| Thyroid | (−) | 108 (99.08) | 175 (97.77) | 283 (98.26) | 0.653 | |||
| (+) | 1 (0.92) | 4 (2.23) | 5 (1.74) | |||||
| PT | ||||||||
| Coronary disease | (−) | 109 (100.00) | 178 (99.44) | 287 (99.65) | 1.000 | |||
| (+) | 0 | 1 (0.56) | 1 (0.35) | |||||
| Thyroid, malignant tumor | (−) | 108 (99.08) | 176 (98.32) | 284 (98.61) | 1.000 | |||
| (+) | 1 (0.92) | 3 (1.68) | 4 (1.39) | |||||
| Current medical history | (−) | 77 (70.64) | 106 (59.22) | 183 (63.54) | 0.051 | |||
| (+) | 32 (29.36) | 73 (40.78) | 105 (36.46) | |||||
| SOC | ||||||||
| Cardiac | (−) | 97 (88.99) | 142 (79.33) | 239 (92.99) | 0.034* | |||
| (+) | 12 (11.01) | 37 (20.67) | 49 (17.01) | |||||
| Pulmonary | (−) | 107 (98.17) | 176 (98.32) | 283 (98.26) | 1.000 | |||
| (+) | 2 (1.83) | 3 (1.68) | 5 (1.74) | |||||
| Endocrine and metabolic | (−) | 96 (88.07) | 157 (87.71) | 253 (87.85) | 0.927 | |||
| (+) | 13 (11.93) | 22 (12.29) | 35 (12.15) | |||||
| Thyroid | (−) | 104 (95.41) | 159 (88.83) | 263 (91.32) | 0.054 | |||
| (+) | 5 (4.59) | 20 (11.17) | 25 (8.68) | |||||
| PT | ||||||||
| Hypertension | (−) | 98 (89.91) | 145 (81.01) | 243 (84.38) | 0.044* | |||
| (+) | 11 (10.09) | 34 (18.99) | 45 (15.63) | |||||
| Diabetes mellitus | (−) | 103 (94.50) | 171 (95.53) | 288 (95.14) | 0.692 | |||
| (+) | 6 (5.50) | 8 (4.47) | 14 (4.86) | |||||
| Dyslipidemia | (−) | 102 (93.58) | 163 (91.06) | 265 (92.01) | 0.445 | |||
| (+) | 7 (6.42) | 16 (8.94) | 23 (7.99) | |||||
| Thyroid, malignant tumor | (−) | 105 (96.33) | 164 (91.62) | 269 (93.40) | 0.118 | |||
| (+) | 4 (3.67) | 15 (8.38) | 19 (6.60) | |||||
| Past and current medication history | ||||||||
| Past medication history | (−) | 86 (78.90) | 110 (61.45) | 196 (68.06) | 0.002* | |||
| (+) | 23 (21.10) | 69 (38.55) | 92 (31.94) | |||||
| AC | ||||||||
| Cardiac | (−) | 97 (88.99) | 149 (83.24) | 246 (85.42) | 0.180 | |||
| (+) | 12 (11.01) | 30 (16.76) | 42 (14.58) | |||||
| Pulmonary | (−) | 106 (97.25) | 163 (91.06) | 269 (93.40) | 0.040* | |||
| (+) | 3 (2.75) | 16 (8.94) | 19 (6.60) | |||||
| Endocrine | (−) | 101 (92.66) | 149 (83.24) | 250 (86.81) | 0.022* | |||
| (+) | 8 (7.34) | 30 (16.76) | 38 (13.19) | |||||
| TC | ||||||||
| Anti-hypertensive | (−) | 100 (91.74) | 154 (86.03) | 254 (88.19) | 0.145 | |||
| (+) | 9 (8.26) | 25 (13.97) | 34 (11.81) | |||||
| Mucoactive | (−) | 106 (97.25) | 163 (91.06) | 269 (93.40) | 0.040* | |||
| (+) | 3 (2.75) | 16 (8.94) | 19 (6.60) | |||||
| Hypoglycemic agent | (−) | 105 (96.33) | 173 (96.65) | 278 (96.53) | 1.000 | |||
| (+) | 4 (3.67) | 6 (3.35) | 10 (3.47) | |||||
| Thyroid hormone | (−) | 108 (99.08) | 164 (91.62) | 272 (94.44) | 0.007* | |||
| (+) | 1 (0.92) | 15 (8.38) | 16 (5.56) | |||||
| Current medication history | (−) | 66 (60.55) | 97 (54.19) | 163 (56.60) | 0.291 | |||
| (+) | 43 (39.45) | 82 (45.81) | 125 (43.40) | |||||
| AC | ||||||||
| Cardiac | (−) | 101 (92.66) | 155 (86.59) | 256 (88.89) | 0.112 | |||
| (+) | 8 (7.34) | 24 (13.41) | 32 (11.11) | |||||
| Endocrine | (−) | 101 (92.66) | 149 (83.24) | 250 (86.81) | 0.022* | |||
| (+) | 8 (7.34) | 30 (16.76) | 38 (13.19) | |||||
| Salivary gland | (−) | 109 (100.00) | 171 (95.53) | 280 (97.22) | 0.026* | |||
| (+) | 0 | 8 (4.47) | 8 (2.78) | |||||
| TC | ||||||||
| Anti-hypertensive | (−) | 103 (94.50) | 157 (87.71) | 260 (90.28) | 0.059 | |||
| (+) | 6 (5.50) | 22 (12.29) | 28 (9.72) | |||||
| Antiemetic | (−) | 109 (100.00) | 172 (96.06) | 281 (97.57) | 0.047* | |||
| (+) | 0 | 7 (3.91) | 7 (2.43) | |||||
| Hypoglycemic agent | (−) | 105 (96.33) | 173 (96.65) | 278 (96.53) | 1.000 | |||
| (+) | 4 (3.67) | 6 (3.35) | 10 (3.47) | |||||
| Thyroid hormone | (−) | 107 (98.17) | 164 (91.62) | 271 (94.10) | 0.022* | |||
| (+) | 2 (1.83) | 15 (8.38) | 17 (5.90) | |||||
| Sialogogue | (−) | 109 (100.00) | 171 (95.53) | 280 (97.22) | 0.026* | |||
| (+) | 0 | 8 (4.47) | 8 (2.78) | |||||
Values are presented as number (%) unless otherwise indicated.
SD, standard deviation; BMI, body mass index; GERD, gastroesophageal reflux disease; LPRD, laryngopharyngeal reflux disease; RSI, reflux symptom index; RFS, reflux finding score; HAD-A, hospital anxiety and depression scale-anxiety; HAD-D, hospital anxiety and depression scale-depression; PPI, proton pump inhibitor; SOC, system organ class; PT, preferred term; AC, anatomical code; TC, therapeutic code.
Table 2.
| Factor | Responder (n=162, 68.35%) | Non-responder (n=75, 31.65%) | Total (n=237) | P-value | ||||
|---|---|---|---|---|---|---|---|---|
| Clinical characteristics | ||||||||
| Sex | Male | 73 (45.06) | 33 (44.00) | 106 (44.73) | 0.879 | |||
| Female | 89 (54.94) | 42 (56.00) | 131 (55.27) | |||||
| Age (yr) | Average±SD | 55.08±11.25 | 54.81±12.98 | 55.00±11.08 | 0.963 | |||
| Median | 56 | 56 | 56 | |||||
| Range | 21–82 | 19–88 | 19–88 | |||||
| BMI change (kg/m2) | Average±SD | −0.08±0.79 | −0.16±1.18 | −0.10±0.93 | 0.722 | |||
| Median | 0 | 0 | 0 | |||||
| Range | −6.41–4.45 | −9.83–1.06 | −9.83–4.45 | |||||
| GERD history | (−) | 105 (64.81) | 39 (52.00) | 144 (60.76) | 0.060 | |||
| (+) | 57 (35.19) | 36 (48.00) | 93 (39.24) | |||||
| LPRD family history | (−) | 128 (79.01) | 64 (85.33) | 192 (81.01) | 0.249 | |||
| (+) | 34 (20.99) | 11 (14.67) | 45 (18.99) | |||||
| Pre-treatment RSI | Average±SD | 16.27±4.28 | 17.48±4.28 | 16.65±4.31 | 0.010* | |||
| Median | 15 | 16 | 15 | |||||
| Range | 13–35 | 13–30 | 13–35 | |||||
| Pre-treatment RFS | Average±SD | 11.57±2.71 | 10.59±2.47 | 11.26±2.67 | 0.012* | |||
| Median | 12 | 10 | 11 | |||||
| Range | 7–18 | 7–18 | 7–18 | |||||
| Number of concurrent medications | Average±SD | 2.64±5.46 | 4.21±7.19 | 3.14±6.09 | 0.153 | |||
| Median | 0.5 | 1 | 1 | |||||
| Range | 0–42 | 0–37 | 0–42 | |||||
| Hospital anxiety and depression scale (HAD) | ||||||||
| HAD-A total | Average±SD | 6.64±3.25 | 7.68±3.48 | 6.97±3.35 | 0.021* | |||
| Median | 6 | 8 | 6 | |||||
| Range | 1–16 | 2–17 | 1–17 | |||||
| HAD-A presence | (−) | 105 (64.18) | 37 (49.33) | 142 (59.92) | 0.024* | |||
| (+) | 57 (35.19) | 38 (50.67) | 95 (40.08) | |||||
| HAD-D total | Average±SD | 7.03±2.97 | 8.19±3.15 | 7.40±3.07 | 0.018* | |||
| Median | 7 | 8 | 7 | |||||
| Range | 0–15 | 2–16 | 0–16 | |||||
| HAD-D presence | (−) | 88 (54.32) | 35 (46.67) | 123 (51.90) | 0.273 | |||
| (+) | 74 (45.68) | 40 (53.33) | 114 (48.10) | |||||
| Treatment compliance | ||||||||
| PPI medication (%) | Average±SD | 98.37±6.12 | 98.38±7.19 | 98.38±6.46 | 0.664 | |||
| Median | 100 | 100 | 100 | |||||
| Range | 53.57–100 | 53.57–100 | 53.57–100 | |||||
| Sleep habitsa) | (−) | 107 (66.05) | 55 (73.33) | 162 (68.35) | 0.262 | |||
| (+) | 55 (33.95) | 20 (26.67) | 75 (31.65) | |||||
| Dietary habit 1b) | (−) | 64 (39.51) | 32 (42.67) | 96 (40.51) | 0.645 | |||
| (+) | 98 (60.49) | 43 (57.33) | 141 (59.49) | |||||
| Dietary habit 2c) | (−) | 78 (48.15) | 35 (46.67) | 113 (47.68) | 0.832 | |||
| (+) | 84 (51.85) | 40 (53.33) | 124 (52.32) | |||||
| Dietary habit 3d) | (−) | 83 (51.23) | 35 (46.67) | 118 (49.79) | 0.513 | |||
| (+) | 79 (48.77) | 40 (53.33) | 119 (50.21) | |||||
| Initial alcohol consumption | (−) | 89 (54.94) | 38 (50.67) | 127 (53.59) | 0.540 | |||
| (+) | 73 (45.06) | 37 (49.33) | 110 (46.41) | |||||
| Alcohol restriction | (−) | 38 (23.46) | 21 (28.00) | 59 (24.89) | 0.557 | |||
| (+) | 115 (70.99) | 56 (74.67) | 171 (75.11) | |||||
| Initial smoking | (−) | 124 (76.54) | 54 (72.00) | 178 (75.11) | 0.452 | |||
| (+) | 38 (23.46) | 21 (28.00) | 59 (24.89) | |||||
| Smoking restriction | (−) | 16 (9.88) | 10 (13.33) | 26 (10.97) | 0.428 | |||
| (+) | 146 (90.12) | 65 (86.67) | 211 (89.03) | |||||
| Past and current medical history | ||||||||
| Past medical history | (−) | 152 (93.83) | 72 (96.00) | 224 (94.51) | 0.760 | |||
| (+) | 10 (6.17) | 3 (4.00) | 16 (5.56) | |||||
| SOC | ||||||||
| Cardiac | (−) | 160 (98.77) | 75 (100.00) | 235 (99.16) | 1.000 | |||
| (+) | 2 (1.23) | 0 | 2 (0.84) | |||||
| Pulmonary | (−) | 160 (98.77) | 75 (100.00) | 235 (99.16) | 1.000 | |||
| (+) | 2 (1.23) | 0 | 2 (0.84) | |||||
| Thyroid | (−) | 159 (98.15) | 74 (98.67) | 233 (98.31) | 1.000 | |||
| (+) | 3 (1.85) | 1 (1.33) | 4 (1.69) | |||||
| PT | ||||||||
| Coronary disease | (−) | 161 (99.38) | 75 (100.00) | 237 (99.58) | 1.000 | |||
| (+) | 1 (0.62) | 0 | 1 (0.42) | |||||
| Thyroid, malignant tumor | (−) | 159 (98.15) | 74 (98.67) | 233 (98.31) | 1.000 | |||
| (+) | 3 (1.85) | 1 (1.33) | 4 (1.69) | |||||
| Current medical history | (−) | 111 (68.52) | 40 (53.33) | 151 (63.71) | 0.024* | |||
| (+) | 51 (31.48) | 35 (46.67) | 86 (36.29) | |||||
| SOC | ||||||||
| Cardiac | (−) | 137 (84.57) | 60 (80.00) | 197 (83.12) | 0.383 | |||
| (+) | 25 (15.43) | 15 (20.00) | 40 (16.88) | |||||
| Pulmonary | (−) | 159 (98.15) | 74 (98.67) | 233 (98.31) | 1.000 | |||
| (+) | 3 (1.85) | 1 (1.33) | 4 (1.69) | |||||
| Endocrine and metabolic | (−) | 139 (85.80) | 67 (89.33) | 206 (86.92) | 0.453 | |||
| (+) | 23 (14.20) | 8 (10.67) | 31 (13.08) | |||||
| Thyroid | (−) | 155 (95.68) | 61 (81.33) | 216 (91.14) | 0.0003* | |||
| (+) | 7 (4.32) | 14 (18.67) | 21 (8.86) | |||||
| PT | ||||||||
| Hypertension | (−) | 138 (85.19) | 62 (82.67) | 200 (84.39) | 0.619 | |||
| (+) | 24 (14.81) | 13 (17.33) | 37 (15.61) | |||||
| Diabetes mellitus | (−) | 153 (94.44) | 72 (96.00) | 225 (94.94) | 0.757 | |||
| (+) | 9 (5.56) | 3 (4.00) | 12 (5.06) | |||||
| Dyslipidemia | (−) | 147 (90.74) | 70 (93.33) | 217 (91.56) | 0.504 | |||
| (+) | 15 (9.26) | 5 (6.67) | 20 (8.44) | |||||
| Thyroid, malignant tumor | (−) | 158 (97.53) | 63 (84.00) | 221 (93.25) | 0.0001* | |||
| (+) | 4 (2.47) | 12 (16.00) | 16 (6.75) | |||||
| Past and current medication history | ||||||||
| Past medication history | (−) | 119 (73.46) | 46 (61.33) | 165 (69.62) | 0.059 | |||
| (+) | 43 (26.54) | 29 (38.67) | 72 (30.38) | |||||
| AC | ||||||||
| Cardiac | (−) | 138 (85.19) | 69 (92.00) | 207 (87.34) | 0.142 | |||
| (+) | 24 (14.81) | 6 (8.00) | 30 (12.66) | |||||
| Pulmonary | (−) | 154 (95.06) | 66 (88.00) | 220 (92.83) | 0.050 | |||
| (+) | 8 (4.94) | 9 (12.00) | 17 (7.17) | |||||
| Endocrine | (−) | 145 (89.51) | 61 (81.33) | 206 (86.92) | 0.083 | |||
| (+) | 17 (10.49) | 14 (18.67) | 31 (13.08) | |||||
| TC | ||||||||
| Anticoagulant | (−) | 150 (92.59) | 74 (98.67) | 224 (94.51) | 0.068 | |||
| (+) | 12 (7.41) | 1 (1.33) | 13 (5.49) | |||||
| Anti-hypertensives | (−) | 142 (87.65) | 70 (93.33) | 212 (89.45) | 0.186 | |||
| (+) | 20 (12.35) | 5 (6.67) | 25 (10.55) | |||||
| Mucoactive | (−) | 154 (95.06) | 66 (88.00) | 220 (92.83) | 0.050 | |||
| (+) | 8 (4.94) | 9 (12.00) | 17 (7.17) | |||||
| Hypoglycemic agent | (−) | 155 (95.68) | 74 (98.67) | 229 (96.62) | 0.441 | |||
| (+) | 7 (4.32) | 1 (1.33) | 8 (3.38) | |||||
| Thyroid hormone | (−) | 159 (98.15) | 65 (86.67) | 224 (94.51) | 0.001* | |||
| (+) | 3 (1.85) | 10 (13.33) | 13 (5.49) | |||||
| Osteoporosis | (−) | 159 (98.15) | 70 (93.33) | 229 (96.62) | 0.113 | |||
| (+) | 3 (1.85) | 5 (6.67) | 8 (3.38) | |||||
| Current medication history | (−) | 95 (58.64) | 46 (61.33) | 141 (59.49) | 0.695 | |||
| (+) | 67 (41.36) | 29 (38.67) | 96 (40.51) | |||||
| AC | ||||||||
| Cardiac | (−) | 145 (89.51) | 68 (90.67) | 213 (89.87) | 0.783 | |||
| (+) | 17 (10.49) | 7 (9.33) | 24 (10.13) | |||||
| Pulmonary | (−) | 133 (82.10) | 61 (81.33) | 194 (81.86) | 0.887 | |||
| (+) | 29 (17.90) | 14 (18.67) | 43 (18.14) | |||||
| Endocrine | (−) | 145 (89.51) | 60 (80.00) | 205 (86.50) | 0.046* | |||
| (+) | 17 (10.49) | 15 (20.00) | 32 (13.50) | |||||
| Salivary gland | (−) | 160 (98.77) | 71 (94.67) | 231 (97.47) | 0.082 | |||
| (+) | 2 (1.23) | 4 (5.33) | 6 (2.53) | |||||
| TC | ||||||||
| Anticoagulant | (−) | 154 (95.06) | 75 (100.00) | 229 (96.62) | 0.059 | |||
| (+) | 8 (4.94) | 0 | 8 (3.38) | |||||
| Anti-hypertensives | (−) | 146 (90.12) | 69 (92.00) | 215 (90.72) | 0.643 | |||
| (+) | 16 (9.88) | 6 (8.00) | 22 (9.28) | |||||
| Gastrointestinal protectant | (−) | 154 (95.06) | 65 (86.67) | 219 (92.41) | 0.023* | |||
| (+) | 8 (4.94) | 10 (13.33) | 18 (7.59) | |||||
| Antiemetic | (−) | 161 (99.38) | 69 (92.00) | 230 (97.05) | 0.005* | |||
| (+) | 1 (0.62) | 6 (8.00) | 7 (2.95) | |||||
| Hypoglycemic agent | (−) | 156 (96.30) | 73 (97.33) | 229 (96.62) | 1.000 | |||
| (+) | 6 (3.70) | 2 (2.67) | 8 (3.38) | |||||
| Thyroid hormone | (−) | 157 (96.91) | 65 (86.67) | 222 (93.67) | 0.007* | |||
| (+) | 5 (3.09) | 10 (13.33) | 15 (6.33) | |||||
| Sialogogue | (−) | 160 (98.77) | 71 (94.67) | 231 (97.47) | 0.082 | |||
| (+) | 2 (1.23) | 4 (5.33) | 6 (2.53) | |||||
Values are presented as number (%) unless otherwise indicated.
SD, standard deviation; BMI, body mass index; GERD, gastroesophageal reflux disease; LPRD, laryngopharyngeal reflux disease; RSI, reflux symptom index; RFS, reflux finding score; HAD-A, hospital anxiety and depression scale-anxiety; HAD-D, hospital anxiety and depression scale-depression; PPI, proton pump inhibitor; SOC, system organ class; PT, preferred term; AC, anatomical code; TC, therapeutic code.
Table 3.
| Factor | Early responder (n=92, 56.79%) | Late responder (n=70, 43.21%) | Total (n=162) | P-value | ||||
|---|---|---|---|---|---|---|---|---|
| Clinical characteristics | ||||||||
| Sex | Male | 41 (44.57) | 32 (45.71) | 73 (45.06) | 0.884 | |||
| Female | 51 (55.43) | 38 (54.29) | 89 (54.94) | |||||
| Age (yr) | Average±SD | 54.10±11.65 | 56.37±10.64 | 55.08±11.25 | 0.160 | |||
| Median | 54.5 | 57 | 56 | |||||
| Range | 21–82 | 33–75 | 21–82 | |||||
| BMI change (kg/m2) | Average±SD | −0.16±0.88 | 0.04±0.65 | −0.08±0.79 | 0.065 | |||
| Median | 0 | 0 | 0 | |||||
| Range | −6.41–1.67 | −1.54–4.45 | −6.41–4.45 | |||||
| GERD history | (−) | 64 (69.57) | 41 (58.57) | 105 (64.81) | 0.147 | |||
| (+) | 28 (30.43) | 29 (41.43) | 57 (35.19) | |||||
| LPRD family history | (−) | 69 (75.00) | 59 (84.29) | 128 (79.01) | 0.151 | |||
| (+) | 23 (25.00) | 11 (15.71) | 34 (20.99) | |||||
| Pre-treatment RSI | Average±SD | 15.86±4.19 | 16.80±4.36 | 16.27±4.28 | 0.042* | |||
| Median | 14 | 15 | 15 | |||||
| Range | 13–34 | 13–35 | 13–35 | |||||
| Pre-treatment RFS | Average±SD | 11.61±2.81 | 11.51±2.60 | 11.57±2.71 | 0.776 | |||
| Median | 12 | 11 | 12 | |||||
| Range | 7–18 | 7–18 | 7–18 | |||||
| Number of concurrent medications | Average±SD | 1.92±3.87 | 3.59±6.95 | 2.64±5.46 | 0.033* | |||
| Median | 0 | 1 | 0.5 | |||||
| Range | 0–20 | 0–42 | 0–42 | |||||
| Hospital anxiety and depression scale (HAD) | ||||||||
| HAD-A total | Average±SD | 6.46±3.09 | 6.87±3.47 | 6.64±3.25 | 0.551 | |||
| Median | 6 | 6.5 | 6 | |||||
| Range | 1–15 | 2–16 | 1–16 | |||||
| HAD-A presence | (−) | 62 (67.39) | 43 (61.43) | 105 (64.81) | 0.431 | |||
| (+) | 30 (32.61) | 27 (38.57) | 57 (35.19) | |||||
| HAD-D total | Average±SD | 7.24±2.96 | 6.76±2.99 | 7.03±2.97 | 0.396 | |||
| Median | 7 | 7 | 7 | |||||
| Range | 1–15 | 0–14 | 0–15 | |||||
| HAD-D presence | (−) | 47 (51.09) | 41 (58.57) | 88 (54.32) | 0.344 | |||
| (+) | 45 (48.91) | 29 (41.43) | 74 (45.68) | |||||
| Treatment compliance | ||||||||
| PPI medication (%) | Average±SD | 98.60±6.27 | 98.07±5.95 | 98.37±6.12 | 0.529 | |||
| Median | 100 | 100 | 100 | |||||
| Range | 53.57–100 | 71.43–100 | 53.57–100 | |||||
| Sleep habitsa) | (−) | 56 (60.87) | 51 (72.86) | 107 (66.05) | 0.111 | |||
| (+) | 36 (39.13) | 19 (27.14) | 55 (33.95) | |||||
| Dietary habit 1b) | (−) | 39 (42.39) | 25 (35.71) | 64 (39.51) | 0.389 | |||
| (+) | 53 (57.61) | 45 (64.29) | 98 (60.49) | |||||
| Dietary habit 2c) | (−) | 41 (44.57) | 37 (52.86) | 78 (48.15) | 0.295 | |||
| (+) | 51 (55.43) | 33 (47.14) | 84 (51.85) | |||||
| Dietary habit 3d) | (−) | 47 (51.09) | 36 (51.43) | 83 (51.23) | 0.966 | |||
| (+) | 45 (48.91) | 34 (48.57) | 79 (48.77) | |||||
| Initial alcohol consumption | (−) | 56 (60.87) | 33 (47.14) | 89 (54.94) | 0.082 | |||
| (+) | 36 (39.13) | 37 (52.86) | 73 (45.06) | |||||
| Alcohol restriction | (−) | 24 (26.09) | 23 (32.86) | 47 (29.01) | 0.347 | |||
| (+) | 68 (73.91) | 47 (67.14) | 115 (70.99) | |||||
| Initial smoking | (−) | 74 (80.43) | 50 (71.43) | 124 (76.54) | 0.180 | |||
| (+) | 18 (19.57) | 20 (28.57) | 38 (23.46) | |||||
| Smoking restriction | (−) | 8 (8.70) | 8 (11.43) | 16 (9.88) | 0.564 | |||
| (+) | 84 (91.30) | 62 (88.57) | 146 (90.12) | |||||
| Past and current medical history | ||||||||
| Past medical history | (−) | 88 (95.65) | 64 (91.43) | 152 (93.83) | 0.331 | |||
| (+) | 4 (4.35) | 6 (8.57) | 10 (6.17) | |||||
| SOC | ||||||||
| Cardiac | (−) | 92 (100.00) | 68 (97.14) | 160 (98.77) | 0.185 | |||
| (+) | 0 | 2 (2.86) | 2 (1.23) | |||||
| Pulmonary | (−) | 91 (98.91) | 69 (98.57) | 160 (98.77) | 1.000 | |||
| (+) | 1 (1.09) | 1 (1.43) | 2 (1.23) | |||||
| Thyroid | (−) | 91 (98.91) | 68 (97.14) | 159 (98.15) | 0.579 | |||
| (+) | 1 (1.09) | 2 (2.86) | 3 (1.85) | |||||
| PT | ||||||||
| Coronary disease | (−) | 92 (100.00) | 69 (98.57) | 161 (99.38) | 0.432 | |||
| (+) | 0 | 1 (1.43) | 1 (0.62) | |||||
| Thyroid, malignant tumor | (−) | 91 (98.91) | 68 (97.14) | 159 (98.15) | 0.579 | |||
| (+) | 1 (1.09) | 2 (2.86) | 3 (1.85) | |||||
| Current medical history | (−) | 66 (71.74) | 45 (64.29) | 111 (68.52) | 0.312 | |||
| (+) | 26 (28.26) | 25 (35.71) | 51 (31.48) | |||||
| SOC | ||||||||
| Cardiac | (−) | 82 (89.13) | 55 (78.57) | 137 (84.57) | 0.065 | |||
| (+) | 10 (10.87) | 15 (21.43) | 25 (15.43) | |||||
| Pulmonary | (−) | 90 (97.83) | 69 (98.57) | 159 (98.15) | 1.000 | |||
| (+) | 2 (2.17) | 1 (1.43) | 3 (1.85) | |||||
| Endocrine and metabolic | (−) | 81 (88.04) | 58 (82.86) | 139 (85.80) | 0.349 | |||
| (+) | 11 (11.96) | 12 (17.14) | 23 (14.20) | |||||
| Thyroid | (−) | 88 (95.65) | 67 (95.71) | 155 (95.68) | 1.000 | |||
| (+) | 4 (4.35) | 3 (4.29) | 7 (4.32) | |||||
| PT | ||||||||
| Hypertension | (−) | 83 (90.22) | 55 (78.57) | 138 (85.19) | 0.039* | |||
| (+) | 9 (9.78) | 15 (21.43) | 24 (14.81) | |||||
| Diabetes mellitus | (−) | 88 (95.65) | 65 (92.86) | 153 (94.44) | 0.502 | |||
| (+) | 4 (4.35) | 5 (7.14) | 9 (5.56) | |||||
| Dyslipidemia | (−) | 86 (93.48) | 61 (87.14) | 147 (90.74) | 0.168 | |||
| (+) | 6 (6.52) | 9 (12.86) | 15 (9.26) | |||||
| Thyroid, malignant tumor | (−) | 89 (96.74) | 69 (98.57) | 158 (97.53) | 0.634 | |||
| (+) | 3 (3.26) | 1 (1.43) | 4 (2.47) | |||||
| Past and current medication history | ||||||||
| Past medication history | (−) | 74 (80.43) | 45 (64.29) | 119 (73.46) | 0.021* | |||
| (+) | 18 (19.57) | 25 (35.71) | 43 (26.54) | |||||
| AC | ||||||||
| Cardiac | (−) | 82 (89.13) | 56 (80.00) | 138 (85.19) | 0.105 | |||
| (+) | 10 (10.87) | 14 (20.00) | 24 (14.81) | |||||
| Pulmonary | (−) | 89 (96.74) | 65 (92.86) | 154 (95.06) | 0.293 | |||
| (+) | 3 (3.26) | 5 (7.14) | 8 (4.94) | |||||
| Endocrine | (−) | 86 (93.48) | 59 (84.29) | 145 (89.51) | 0.059 | |||
| (+) | 6 (6.52) | 11 (15.71) | 17 (10.49) | |||||
| TC | ||||||||
| Anti-hypertensives | (−) | 84 (91.30) | 58 (82.86) | 142 (87.65) | 0.105 | |||
| (+) | 8 (8.70) | 12 (17.14) | 20 (12.35) | |||||
| Mucoactive | (−) | 89 (96.74) | 65 (92.86) | 154 (95.06) | 0.293 | |||
| (+) | 3 (3.26) | 5 (7.14) | 8 (4.94) | |||||
| Hypoglycemic agent | (−) | 90 (97.83) | 65 (92.86) | 155 (95.68) | 0.241 | |||
| (+) | 2 (2.17) | 5 (7.14) | 7 (4.32) | |||||
| Thyroid hormone | (−) | 91 (98.97) | 68 (97.14) | 159 (98.15) | 0.579 | |||
| (+) | 1 (1.09) | 2 (2.86) | 3 (1.85) | |||||
| Current medication history | (−) | 58 (63.04) | 37 (52.86) | 95 (58.64) | 0.192 | |||
| (+) | 34 (36.96) | 33 (47.14) | 67 (41.36) | |||||
| AC | ||||||||
| Cardiac | (−) | 86 (93.48) | 59 (84.29) | 145 (89.51) | 0.059 | |||
| (+) | 6 (6.52) | 11 (15.71) | 17 (10.49) | |||||
| Endocrine | (−) | 86 (93.48) | 59 (84.29) | 145 (89.51) | 0.059 | |||
| (+) | 6 (6.52) | 11 (15.71) | 17 (10.49) | |||||
| Salivary gland | (−) | 92 (100.00) | 68 (97.14) | 160 (98.77) | 0.185 | |||
| (+) | 0 | 2 (2.86) | 2 (1.23) | |||||
| TC | ||||||||
| Anti-hypertensives | (−) | 87 (94.57) | 59 (84.29) | 146 (90.12) | 0.030* | |||
| (+) | 5 (5.43) | 11 (15.71) | 16 (9.88) | |||||
| Hypoglycemic agent | (−) | 90 (97.83) | 66 (94.29) | 156 (96.30) | 0.404 | |||
| (+) | 2 (2.17) | 4 (5.71) | 6 (3.70) | |||||
| Anti-hyperlipidemic | (−) | 89 (96.74) | 63 (90.00) | 152 (93.83) | 0.102 | |||
| (+) | 3 (3.26) | 7 (10.00) | 10 (6.17) | |||||
| Thyroid hormone | (−) | 90 (97.83) | 67 (95.71) | 157 (96.91) | 0.653 | |||
| (+) | 2 (2.17) | 3 (4.29) | 5 (3.09) | |||||
| Sialogogue | (−) | 92 (100.00) | 68 (97.14) | 160 (98.77) | 0.185 | |||
| (+) | 0 | 2 (2.86) | 2 (1.23) | |||||
Values are presented as number (%) unless otherwise indicated.
SD, standard deviation; BMI, body mass index; GERD, gastroesophageal reflux disease; LPRD, laryngopharyngeal reflux disease; RSI, reflux symptom index; RFS, reflux finding score; HAD-A, hospital anxiety and depression scale-anxiety; HAD-D, hospital anxiety and depression scale-depression; PPI, proton pump inhibitor; SOC, system organ class; PT, preferred term; AC, anatomical code; TC, therapeutic code.
Table 4.
| Factor | Estimate | Odds ratio | 95% CI | P-value | |
|---|---|---|---|---|---|
| Predictor of response to 1 month of treatment | |||||
| Sleep habits (reference: [−]) | 0.580 | 1.785 | 1.06–3.007 | 0.029* | |
| Initial alcohol consumption (reference: [−]) | −0.533 | 0.587 | 0.355–0.969 | 0.037* | |
| Presence of past medication history (reference: [−]) | −0.826 | 0.438 | 0.215–0.891 | 0.005* | |
| Current medication history, TC, sialogogue (reference: [−]) | −14.790 | <0.001 | <0.001–>999.999 | 0.983 | |
| Predictors of response to 3 months of treatment | |||||
| Male sex (reference: female) | −0.663 | 0.516 | 0.269–0.987 | 0.046* | |
| HAD-D total | −0.143 | 0.867 | 0.784–0.958 | 0.005* | |
| Pre-treatment RFS | 0.172 | 1.187 | 1.049–1.344 | 0.007* | |
| Current medical history, SOC, thyroid (reference: [−]) | −1.038 | 0.354 | 0.109–1.153 | 0.085 | |
| Past medication history, TC, thyroid hormone (reference: [−]) | −1.827 | 0.161 | 0.033–0.788 | 0.024* | |
| Predictors of early responders compared to late responders | |||||
| Presence of past medication history (reference : [−]) | −0.826 | 0.438 | 0.215–0.891 | 0.023* | |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download