Abstract
Purpose
This study examined the effects of duck meats with turmeric powder on blood lipids in 10 female university students.
Methods
The subjects received duck meat with 0%, 0.1%, 0.2%, and 0.4% turmeric powder and glucose, total cholesterol, triglyceride (TG), high-density lipoprotein (HDL)-cholesterol, and low-density lipoprotein (LDL)-cholesterol in their serums after 30, 60, 90, 120, and 180 min were measured.
Results
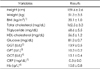
The average height, weight, and body mass index of subjects were 159.6 ± 2.6 cm, 51.3 ± 3.5 kg, and 20.1 ± 1.0, respectively. The fasting glucose, γ-glutamyl transferase (GGT), glutamic pyruvic transferase (GPT), glutamic oxaloacetic transferase (GOT), c-reactive protein (CRP), and hemoglobin were within the normal range. The Δ-AUC (area under the curve) of postprandial glucose, TG did not change, but Δ-AUC of postprandial total cholesterol and LDL were significantly decreased, and HDL was increased by intake of the duck meat with turmeric powder.
Figures and Tables
Fig. 1
Incremental blood glucose concentration and Δ-AUC for 4 hours after consuming each test diet for 10 women. Bars are mean ± SE. Bars with different letters are significantly among the treated concentration of turmeric powder (p < 0.05). 0: 0% turmeric added 0.1: 0.1% turmeric added 0.2: 0.2% turmeric added 0.4: 0.4% turmeric added.

Fig. 2
Incremental lipid profiles and Δ-AUC for 4 hours after consuming each test diet for 10 women. Bars are mean ± SE. Bars with different letters are significantly among the treated concentration of turmeric powder (p < 0.05). 0: 0% turmeric added 0.1: 0.1% turmeric added 0.2: 0.2% turmeric added 0.4: 0.4% turmeric added.

References
1. Ku SK, Hwang SH, Lim SD, Lee KH, Kim YB. Nutritional characteristics and quality changes of duck by-products during frozen storage at -20℃. J Korean Soc Food Sci Anim Resour. 2013; 33(1):109–118.

2. Kim J, Kim WK. Effect of duck extract on lipids in rats. Korean J Nutr. 2003; 36(1):3–8.
3. Song YM, Jin SK, Kim IS, Cho YC, Kim HY, Hah KH, Nam KY. Effects of dietary onion supplementation of on the physicochemical properties of duck meat. J Korean Soc Food Sci Anim Resour. 2004; 24(1):66–72.
4. Yoon WH, Hwang JY, Kim CH. Isolation and purification of antitumar substance from the sulfur fed duck. J Korean Soc Food Sci Anim Resour. 2004; 24(3):293–297.
5. Kim HJ, Liang CY, Ju MK, Lee KH, Cho SH, Lee SK. Effects of dietary germanium supplementaion on the meat quality of duck. J Korean Soc Food Sci Anim Resour. 2003; 23(3):200–208.
6. Lim KT, Lee JC, Cheong JH, Jung WJ, Kim TH. Meat quality mallard by feeding of MS-fermented food waste. Korean J Environ Agric. 2000; 19(4):332–338.
7. Choi HY. Antimicrobial activity of UlGeum(Curcuma longa L.) extract and its microbiological and sensory characteristic effects in processed foods. Korean J Food Cookery Sci. 2009; 25(3):350–356.
8. Yang DS, Yang SJ. Effects of Curcuma longa L. on MDA-MB-231 human breast cancer cells and DMBA-induced breast cancer in rats. J Orient Obstet Gynecol. 2013; 26(3):44–58.
9. Kim CR. Enhancement of liver function by Curcuma extract on acute hepatotoxicity in rat. J Korean Soc Food Sci Anim Resour. 2006; 26(3):386–393.
10. Lee SH, Choi WJ, Lim YS, Kim SH. Antimicrobial effect of ethanol extract from Curcuma aromatica S. Korean J Food Sci Technol. 1997; 9:161–165.
11. Kang JO, Lee SG. Effects of Opuntia ficus-indica pigment and sodium lactate on nitrite-reduced sausages. J Anim Sci Technol. 2008; 50(4):551–560.
12. Lim HS, Baik IK, Lee HS, Lee YJ, Chung NS, Jho SY, Kim SS. Effects of the life style in patients with coronary artery disease on the serum lipid concentrations and atherosclerotic coronary lesion. Korean J Lipidol. 1995; 5(1):71–83.
13. Cho SH, Choi Y. Dietary theraphy of hyperlipidemia. Korean J Lipidol. 1994; 4(2):109–117.
14. Lee JS, Lee MH, Kwon TB, Ju JS. A study on the concentration of serum lipids and its related factors of persons over 40 years old in Whachon area, Kang-won do. Korean J Nutr. 1996; 29(9):1035–1041.
15. Kim JH, Kim OK, Yoon HG, Park J, You Y, Kim K, Lee YH, Choi KC, Lee J, Jun W. Anti-obesity effect of extract from fermented Curcuma longa L. through regulation of adipogenesis and lipolysis pathway in high-fat diet-induced obese rats. Food Nutr Res. 2016; 60:30428.
16. Ho JN, Jang JY, Yoon HG, Kim Y, Kim S, Jun W, Lee J. Anti-obesity effect of a standardised ethanol extract from Curcuma longa L. fermented with Aspergillus oryzae in ob/ob mice and primary mouse adipocytes. J Sci Food Agric. 2012; 92(9):1833–1840.
17. Han JM, Lee JS, Kim HG, Seol IC, Im HJ, Cho JH, Son CG. Synergistic effects of Artemisia iwayomogi and Curcuma longa radix on high-fat diet-induced hyperlipidemia in a mouse model. J Ethnopharmacol. 2015; 173:217–224.

18. Shin HS, Han JM, Kim HG, Choi MK, Son CG, Yoo HR, Jo HK, Seol IC. Anti-atherosclerosis and hyperlipidemia effects of herbal mixture, Artemisia iwayomogi Kitamura and Curcuma longa Linne, in apolipoprotein E-deficient mice. J Ethnopharmacol. 2014; 153(1):142–150.
19. Purohit A. Antifertility efficacy of Curcuma longa (50% E to H extract) with special referance to serum biochemistry and fertility test. Anc Sci Life. 1999; 18(3-4):192–194.
20. Babu PS, Srinivasan K. Hypolipidemic action of curcumin, the active principle of turmeric (Curcuma longa) in streptozotocin induced diabetic rats. Mol Cell Biochem. 1997; 166(1-2):169–175.
21. Yun S, Yeon JY, Kim MH, Kang MH, Kim TH, Son YK, Kim MH. The Effects of Angelica keiskei Koidzumi and turmeric extract supplementation on the blood lipids, and antioxidant and inflammatory markers in hypercholesterolemic adults in Korea. Korean J Food Nutr. 2009; 22(4):517–525.
22. Soni KB, Kuttan R. Effect of oral curcumin administration on serum peroxides and cholesterol levels in human volunteers. Indian J Physiol Pharmacol. 1992; 36(4):273–275.
23. Baum L, Cheung SK, Mok VC, Lam LC, Leung VP, Hui E, Ng CC, Chow M, Ho PC, Lam S, Woo J, Chiu HF, Goggins W, Zee B, Wong A, Mok H, Cheng WK, Fong C, Lee JS, Chan MH, Szeto SS, Lui VW, Tsoh J, Kwok TC, Chan IH, Lam CW. Curcumin effects on blood lipid profile in a 6-month human study. Pharmacol Res. 2007; 56(6):509–514.

24. Lin XL, Liu MH, Hu HJ, Feng HR, Fan XJ, Zou WW, Pan YQ, Hu XM, Wang Z. Curcumin enhanced cholesterol efflux by upregulating ABCA1 expression through AMPK-SIRT1-LXRα signaling in THP-1 macrophage-derived foam cells. DNA Cell Biol. 2015; 34(9):561–572.

25. Sahebkar A. A systematic review and meta-analysis of randomized controlled trials investigating the effects of curcumin on blood lipid levels. Clin Nutr. 2014; 33(3):406–414.

26. Zhang I, Cui Y, Amiri A, Ding Y, Campbell RE, Maysinger D. Pharmacological inhibition of lipid droplet formation enhances the effectiveness of curcumin in glioblastoma. Eur J Pharm Biopharm. 2016; 100:66–76.

27. Jain SK, Rains J, Jones K. Effect of curcumin on protein glycosylation, lipid peroxidation, and oxygen radical generation in human red blood cells exposed to high glucose levels. Free Radic Biol Med. 2006; 41(1):92–96.

28. Morrone Mda S, Schnorr CE, Behr GA, Gasparotto J, Bortolin RC, da Boit Martinello K, Saldanha Henkin B, Rabello TK, Zanotto-Filho A, Gelain DP, Moreira JC. Curcumin supplementation decreases intestinal adiposity accumulation, serum cholesterol alterations, and oxidative stress in ovariectomized rats. Oxid Med Cell Longev. 2016; 2016:5719291.
29. Ramírez-Tortosa MC, Mesa MD, Aguilera MC, Quiles JL, Baró L, Ramirez-Tortosa CL, Martinez-Victoria E, Gil A. Oral administration of a turmeric extract inhibits LDL oxidation and has hypocholesterolemic effects in rabbits with experimental atherosclerosis. Atherosclerosis. 1999; 147(2):371–378.

30. Arafa HM. Curcumin attenuates diet-induced hypercholesterolemia in rats. Med Sci Monit. 2005; 11(7):BR228–BR234.
31. Ro HK, Choi IS, Oh SH. Effects of high carbohydrate, high fat and protein meal on postprandial thermogenesis in young women. J Korean Soc Food Sci Nutr. 2005; 34(8):1202–1209.
32. Oh HK, Lim HS. Effects of hamburger patties added sea tangle (Laminaria japonica) powder and/or cooked rice on postprandial blood glucose and lipid levels. J Korean Soc Food Sci Nutr. 2011; 40(6):809–817.

33. Kim HH, Lim HS. Effects of sea tangle-added patty on postprandial serum lipid profiles and glucose in borderline hypercholesterolemic adults. J Korean Soc Food Sci Nutr. 2014; 43(4):522–529.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download