This article has been
cited by other articles in ScienceCentral.
Abstract
This study discusses laryngospasm following orthognathic surgery and requiring emergency intubation, followed by systemic complications due to a hypoxic event. A 34-year-old male patient underwent orthognathic surgery due to facial asymmetry. When emerging from general anesthesia, blood pressure elevated suddenly, and severe agitation occurred. After extubation, desaturation occurred immediately, resulting in cyanosis and loss of consciousness. An emergency cricothyroidotomy was performed, and oral intubation was required to maintain the airway. The patient was sent to the intensive care unit on a ventilator. On postoperative day 2, the red blood cell count and hemoglobin level decreased significantly. Electrocardiogram showed abnormalities, although vital signs were stable and no bleeding was observed. Laryngospasm often occurs under general anesthesia, resulting in sustained closure of the vocal cords and obstruction of the airway. This condition can be life-threatening, and awareness of the various precipitating factors is important. Orthognathic surgery is a risk factor for laryngospasm because its surgical site affects the airway, and it involves discharge of blood via intraoral wounds. It is important to understand systemic changes occurring after a hypoxic event, and consultation with specialists from various departments should be actively pursued.
Go to :

Keywords: Orthognathic surgery, Laryngospasm, Cricothyrotomy, Ischemia-reperfusion injury
I. Introduction
Laryngospasm, a responsive reflex to prevent aspiration, may occur while a patient is induced and/or emerges from general anesthesia, resulting in partial or complete sustained closure of the vocal cords. Laryngospasm is self-limiting in most cases, however, when spasm is persistent, it may become a life-threatening situation due to airway complications if not managed properly
1,2.
Laryngospasm is caused by involuntary vocal musculature spasms, and risk factors have been reported widely. Airway irritation is a strong trigger of laryngospasm, and surgeries involving the airway such as tonsillectomy and adenoidectomy or oral debris, blood, and salivation can be risky
3,4.
An acute hypoxic event may result in oxidative stress and systemic complications including cell apoptosis, secondary myocardial injury, and endothelial cell dysfunction
5,6.
Laryngospasm especially is common in pediatrics, otorhinolaryngology, and anesthesiology but had not been reported in oral and maxillofacial surgery. In this case report, we introduce an unusual complication of laryngospasm immediately after extubation following orthognathic surgery, leading to airway obstruction and requiring emergent management.
Go to :

II. Case Report
A 34-year-old male (height, 182 cm; weight, 74 kg) with no specific medical problems visited the Department of Oral and Maxillofacial Surgery at Yongin Severance Hospital with a chief complaint of facial asymmetry. The patient was diagnosed with mandibular prognathism with facial asymmetry, and orthognathic surgery with advancement genioplasty was planned after presurgical orthodontic treatment. During the preoperative anesthetic evaluation, the patient was evaluated as American Society of Anesthesiologists physical status classification I. Other physical results and laboratory values were within normal limits.
On the operation day, the patient was placed in a supine position, and standard monitoring (non-invasive blood pressure, electrocardiogram [ECG]), pulse oximetry, neuromuscular monitor, and capnography) was performed. The patient received preoxygenation with 100% oxygen from a face mask and 0.2 mg of glycopyrrolate as an injection. General anesthesia was induced with inhalational and intravenous agents of 50 µg of fentanyl, 200 mg of 1% lidocaine, 150 mg of 1% propofol, and 50 mg of rocuronium. Nasotracheal intubation was performed with a nasal tube (Nasal RAE; COVIDIEN) of inner diameter 6.5 mm under laryngoscopy. A peripheral intravenous line (IV) of 18G on the right leg and an arterial line of 20G on the left radial artery were placed.
Orthognathic surgery proceeded under hypotensive anesthesia in the usual manner. Le fort I osteotomy was performed and the segment was fixed with a biodegradable system comprised of four L-shaped plates and monocortical 2.0-mm-diameter screws (hydroxyapatite/poly-L-lactide, Osteotrans MX). On the mandible, bilateral intraoral vertical ramus osteotomy was performed with no significant undesired event. Genioplasty involved 6 mm advancement and left mandibular border shaving, and fixture was bicortical using the same system as above, with three 2.0-mm-diameter and 16-mm-length screws. The patient exhibited robust musculoskeletal features, indicated by high bone mineral density and abundant bone marrow. Despite the absence of significant neurovascular injuries during any procedures, mucosal oozing was noted without the presence of a hematological disorder. During surgery, blood loss was greater than expected at 1,700 cc but was well controlled with hemostatic materials, electrocoagulation, and gauze packing. No other anesthetic events occurred during surgery.
The patient was administered 150 mg of suggamadex after surgery. The anesthesiologist confirmed self-ventilation and verbal response of the patient and removed the nasotracheal tube. During emergence from anesthesia, a sudden elevation in blood pressure to 170/70 mmHg was observed, accompanied by bilateral nostril oozing. Active suctioning was performed using a rubber catheter. Subsequently, the patient exhibited severe agitation, with a rapid decrease in oxygen saturation to 64%, leading to cyanosis and loss of consciousness. An attempt at mask ventilation was ineffective, with the anesthesiologists recognizing clinical signs of laryngospasm. All the doctors in the operating room recognized the emergent situation and called for a backup team. Emergency cricothyrotomy was performed with a needle, and saturation increased to 98% in only a few minutes, after which the spasm spontaneously abated.(
Fig. 1)
 | Fig. 1Time plots during laryngospasm event schematic diagram of event occurrence and management following orthognathic surgery. (Op.: operation, EBL: estimated blood loss, G/A: general anesthesia, BP: blood pressure, ICU: intensive care unit) 
|
Anesthesia was induced again and a Foley catheter was inserted for further systemic management. Estimated blood loss was estimated to be 2,300 cc at the time of reintubation, which was required to maintain airway patency. However, as intraoral swelling was severe, and the vocal cords were stiff, intubation was performed orally with tube of inner diameter 6.5 mm, narrower than average for male patients.(
Fig. 2)
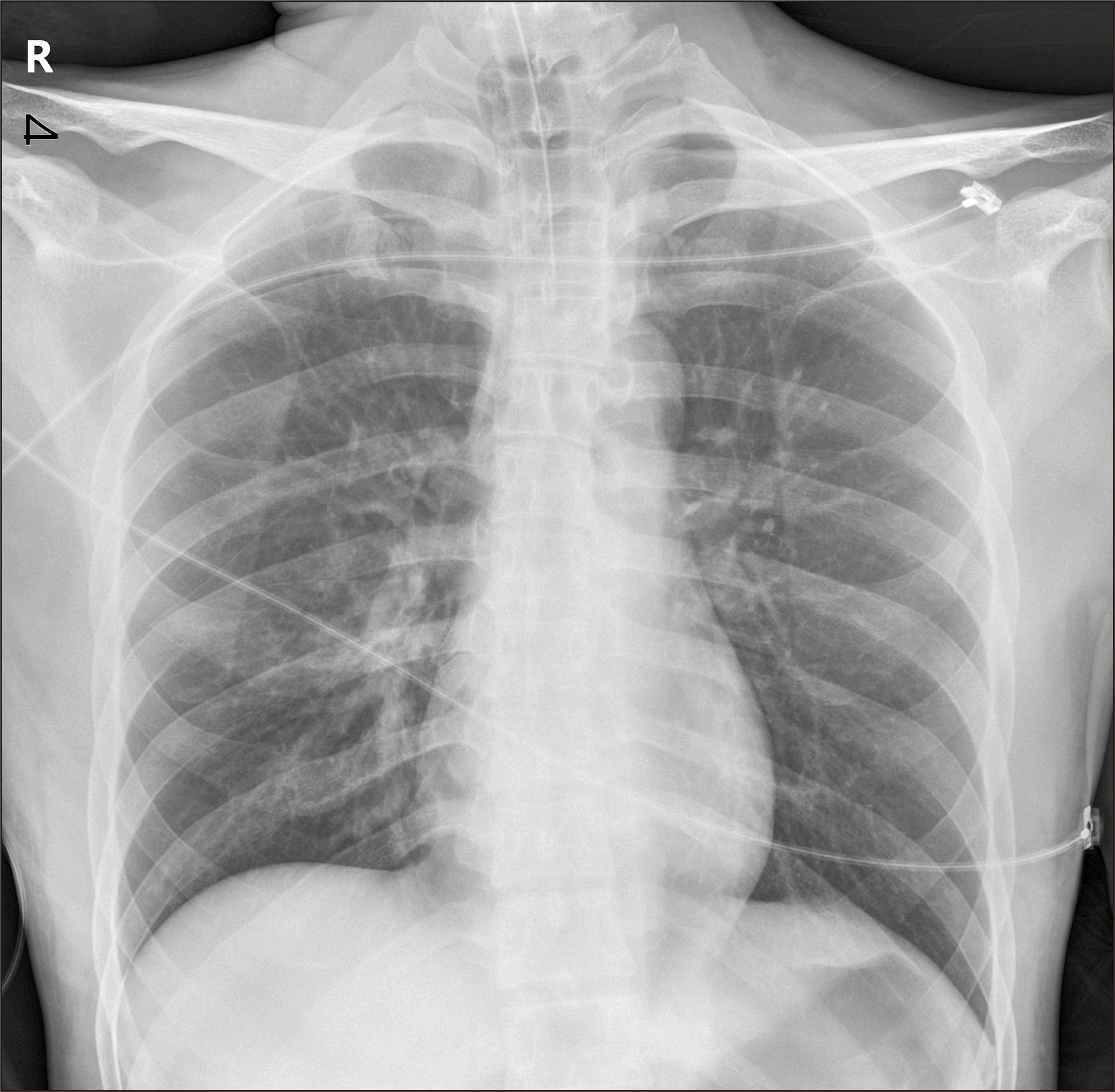 | Fig. 2Chest anterior posterior after oral re-intubation. When considering our patient’s height, this endotracheal tube with inner diameter 6.5 mm was less size, however, it was an inevitable choice in an emergency situation. 
|
To assess neurologic impairment, the patient was awakened for a few minutes. Pupil response was normal, and verbal response was possible. The Otorhinolaryngology team was combined under general anesthesia, and the bleeding was explored with an endoscope, with no obvious source identified in the nasal cavity. The patient was sent to the intensive care unit (ICU) on a ventilator under sedation, and his systemic condition recovered throught the day.
On postoperative day (POD) #2, his hemoglobin (Hb) and hematocrit values decreased significantly on complete blood cell count (CBC) test, although there was no remarkable hemorrhage.(
Table 1) The ECG showed ST & T wave abnormality and T wave inversion (
Fig. 3), and cardiac enzyme markers showed remarkable elevation (CK, 2213 IU/L; CK-MB, 4.1 ng/mL; NT-proBNP, 13 pg/mL; troponin T [TnT], 49 pg/mL). Three packs of red blood cells (RBCs) were transfused, but the Hb value did not increase. Transthoracic echocardiography (TTE) was performed according to the recommendation of the cardiologist, and cardiac function was noted as normal.
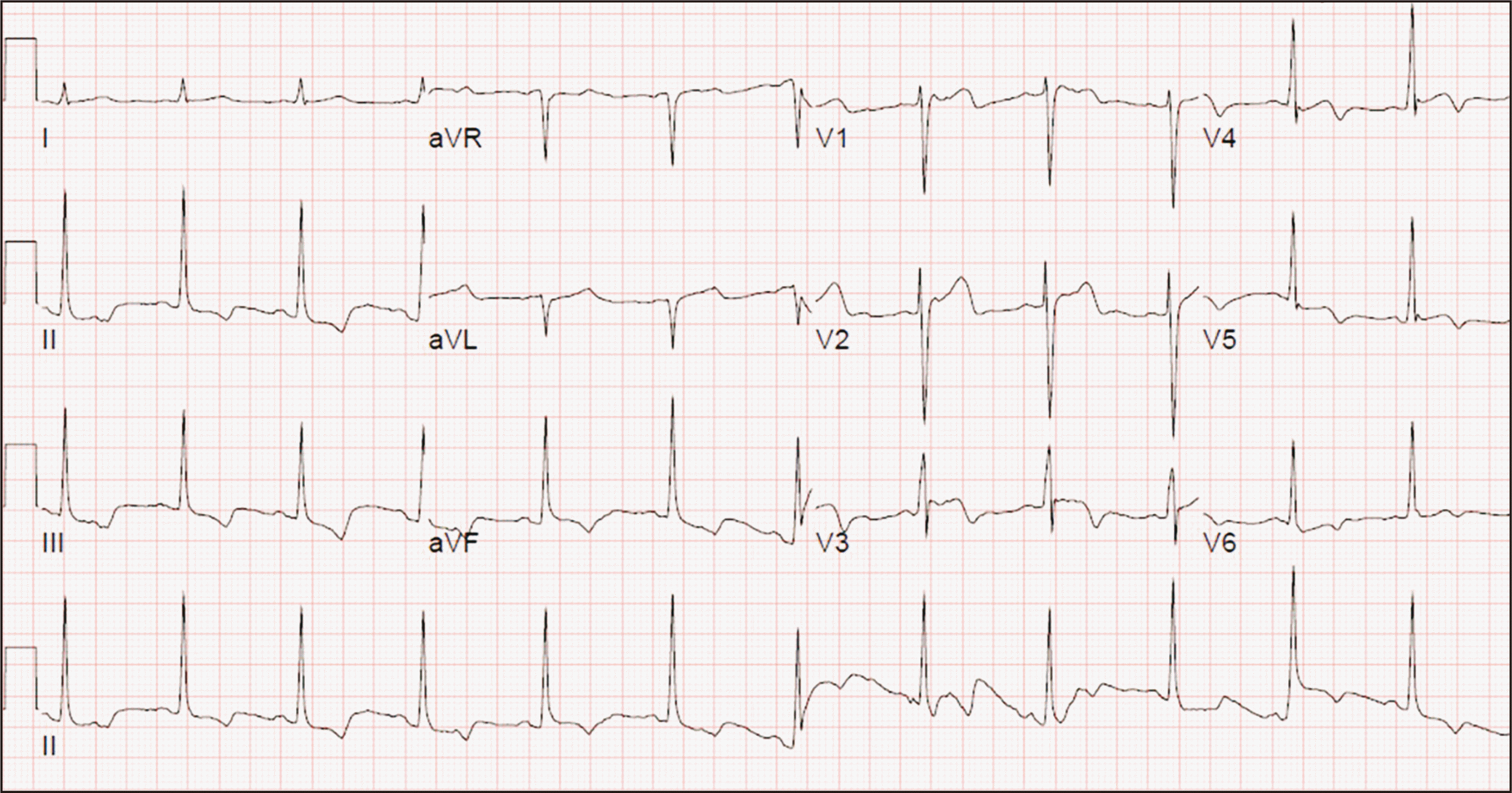 | Fig. 3Electrocardiogram on the postoperative day #2. The results showed that normal sinus rhythm (heart rate; 76 BPM), but ST & T wave abnormality considered with ischemia, and T wave inversion. 
|
Table 1
Results of complete blood cell count test
|
Pre-op. |
Immediately after op. |
POD #1 |
POD #2 |
POD #3 |
POD #4 |
POD #5 |
Discharge |
|
WBC (103/µL) |
5.84 |
21.68 |
18.08 |
10.23
10.10
9.14 |
8.58 |
7.82 |
7.34 |
10.27 |
|
RBC (106/µL) |
5.75 |
3.61 |
3.55 |
2.61
2.61
2.70 |
3.08 |
3.48 |
3.80 |
4.25 |
|
Hb (g/dL) |
16.70 |
10.50 |
10.20 |
7.50
7.70
8.00 |
9.02 |
10.18 |
11.35 |
12.20 |
|
Hematocrit (%) |
50.04 |
30.60 |
30.30 |
22.00
21.90
22.70 |
25.60 |
29.20 |
32.10 |
36.10 |
|
Platelet (103/µL) |
351 |
245 |
251 |
170
159
148 |
142 |
232 |
283 |
387 |

On POD #3, the patient was alert, and vital signs and ventilation were stable. The endotracheal tube was removed and the patient was sent to the general ward. Routine post-operative procedures were performed (dressing, intermaxillary fixation, and follow-up X-rays) on POD #4.(
Fig. 4) Before the patient was discharged, a brain magnetic resonance imaging (MRI) was performed and showed no sign of ischemic injury.
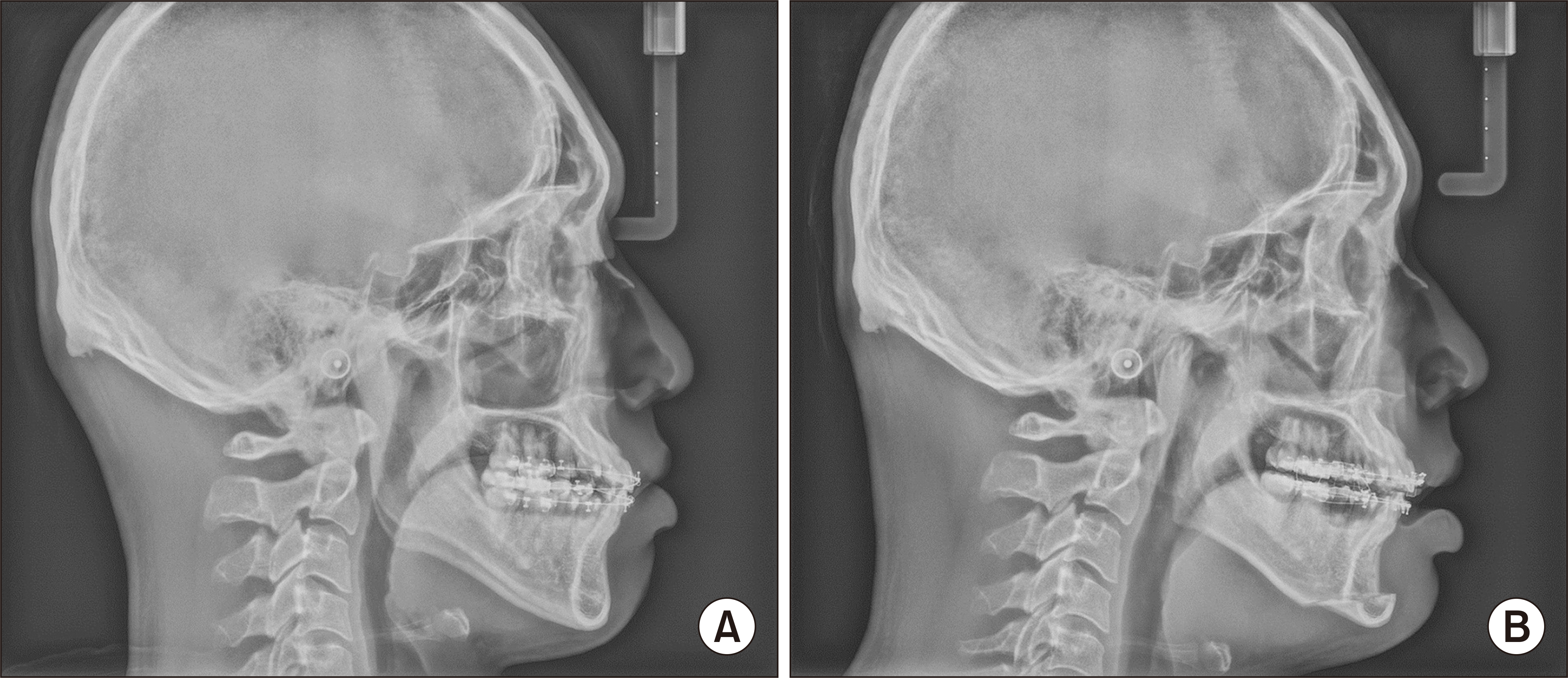 | Fig. 4Lateral cephalometric radiography. A. Pre-operative image. B. Post-operative image after intermaxillary fixation on postoperative day #4. 
|
Go to :

III. Discussion
Although our patient required reintubation and ICU care, he was well managed by a team approach with many specialists from different departments and did not experience significant morbidity. The rarity of such an experience has limited the various consequences of related hypoxia and its management. Additional research into this occurrence is needed.
1. Considerations of laryngospasm
Laryngospasm is glottis closure caused by reflex constriction of the intrinsic laryngeal muscles following innocuous stimuli including secretions, laryngeal irritation, rapid inflow of cold air, or endotracheal tube insertion. This leads to hypoxia and hypercapnia, which can relieve the spastic reflex and self-limit the event. However, in certain cases, the spasm is sustained and upper airway obstruction results in severe complications such as pulmonary edema, bronchospasm, arrhythmia, and cardiac arrest. The risk factors of laryngospasm are classified into three categories, as anesthesia-related, patient-related, and surgery related factors.(
Table 2)
1-4,7,8
Table 2
Laryngospasm risk factors
|
Known risk factors of laryngospasm |
|
Insufficient anesthesia depth |
|
Airway irritation |
|
Volatile anesthetic agents |
|
Younger age |
|
Asthma |
|
Smoking |
|
Obesity |
|
Intraoral surgery |
|
Airway anomaly |
|
Others |

1) Anesthesia-related factors
Insufficient depth of anesthesia during induction, maintenance, and emergence can contribute to susceptibility to laryngospasm. Volatile anesthetics, mucous, and blood can irritate the airway and trigger laryngospasm, as can laryngoscopy and use of a suction catheter. Among the volatile anesthetic agents, halothane and sevoflurane are the least irritating, whereas desflurane is most likely to cause disruption. On the other hand, the IV anesthetic agent propofol suppresses airway reflexes, limiting laryngospasm. Especially in pediatric patients, inexperienced anesthetists are more likely to cause laryngospasm.
2) Patient-related factors
Age is inversely related to laryngospasm following general anesthesia, and young children show the greatest risk. Asthma or upper respiratory tract infection can irritate the airway and increase laryngospasm risk by approximately 10-fold. Chronic smokers are more prone to laryngospasm because of increased airway sensitivity. Obesity and anatomical anomalies in the airway also contribute to laryngospasm.
3) Surgery-related factors
Both airway-sharing surgery and non-airway-sharing surgery can trigger laryngospasm. Airway-sharing surgeries such as tonsillectomy and adenoidectomy have been reported as the greatest risk factor. Due to stimulation of the superior laryngeal nerve or esophageal nerve, thyroid surgery and esophageal surgeries are reported to cause laryngospasm. Non-airway-sharing surgery such as appendectomy has also been reported to produce laryngospasm9.
Orthognathic surgery also involves risk of laryngospasm due to the surgical environment of proximity to the airway, surgical debris, blood clot of the pharynx, salivation, and postoperative drainage via the surgical wounds.
An effective management plan and team communication are vital to treatment of laryngospasm during emergencies. When oxygen saturation drops below 80%, oxygenation should be re-established immediately. Treatment requires removal of any triggering stimulus, clearance of the oropharynx, supply of 100% oxygen for continuous positive airway pressure, and increased anesthesia with propofol
1,4,7,10.
2. Considerations of ischemic injury
When considering the abnormal CBC and ECG results on POD #2, along with the normal TTE results and stable vital signs, the ischemic event can be explained by laryngospasm after extubation resulted in oxidative stress, followed by eryptosis and acute myocardial injury.
Hypoxia may induce RBC membrane fragility, micro defects, and lytic pore formation. This can result in intravascular hemolysis that can occur as an important source of extracellular adenosine triphosphate (ATP)
5. According to previous animal studies, plasma iron levels decrease 24-48 hours after hypoxia and do not return to baseline until 48-96 hours after erythropoietin concentrations return to baseline
11. Our patient’s pre-existing abnormal RBC and Hb levels were part of a necrotic process in response to hypoxia, and the Hb level was not increased with transfusion of three packs of RBC on POD #2. Oral feeding was started and the patient’s systemic condition recovered in the general ward.
Myocardial injury after non-cardiac surgery is considered with elevation of a postoperative cardiac biomarker (TnT), which occurs in the first 30 days and typically within 72 hours after surgery
6,12,13. Changes in oxygen supply can cause inadequate tissue and organ oxygenation resulting in secondary myocardial injury after a hypoxic event. For consideration of not only myocardial injury, but ischemic effect on skeletal muscles, the patient began limited ambulation with a walker.
Laryngospasm has the potential to result in life-threatening situations and should be managed immediately using a team approach to avoid significant morbidity. Orthognathic surgery may be considered a risk factor for laryngospasm. To manage a patient’s systemic condition following a significant complication, specialists from various departments should be consulted.
Go to :


