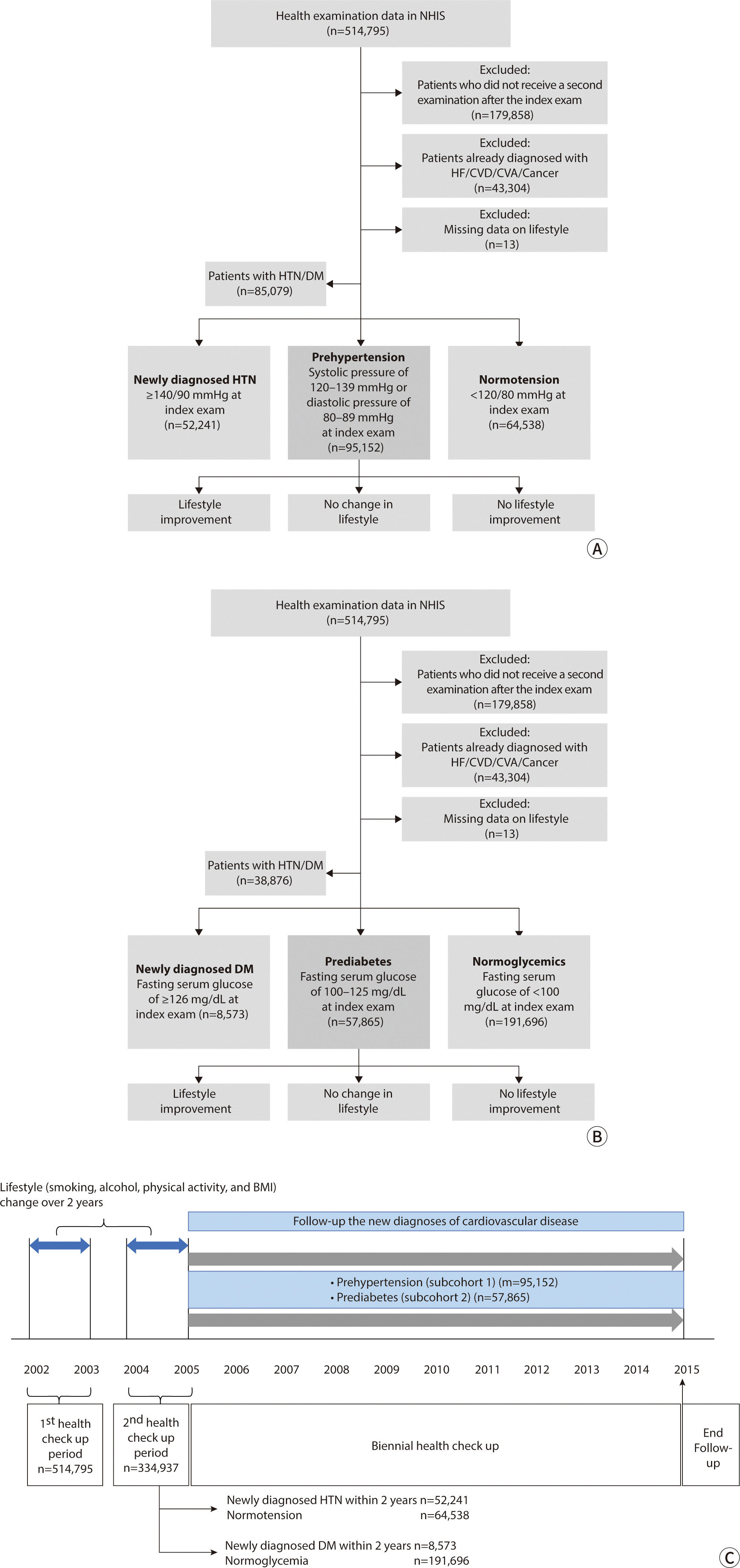
Males constituted 65.2% of the prehypertension cohort (n=60,084). Additionally, women
were found to consume less alcohol (32.0% vs. 80.7% for never drinking) and to
exercise less frequently (46.4% vs. 65.0% for never exercising) compared to men.
Among the men, 44.1% were current smokers (n=25,201), whereas only 1.7% of women
smoked currently (n=577). Similarly, in the prediabetes cohort, men made up 65.4% of
the subjects (n=37,836). The percentage of women who neither exercised (47.2% vs.
67.7%) nor consumed alcohol (29.8% vs. 80.9%) was lower compared to men. In this
cohort, 41.8% of men (n=15,144) were current smokers, while only 2.2% of women
(n=432) were smokers. Furthermore, the prevalence of BMI over 25.0 kg/m
2
was lower in women than in men in both cohorts (32.3% vs. 28.5% in the
prehypertension cohort and 38.1% vs. 36.7% in the prediabetes cohort). The general
characteristics of subjects according to group are summarized in
Table 1.
Table 2 presents the incidence of diseases
during follow-up in subjects with prehypertension and prediabetes, categorized by
gender. In the prehypertension cohort, there were 3,979 MACE cases in men and 2,290
in women. The MACE incidence rates were similar between genders (62.6/10,000 PY for
men vs. 62.1/10,000 PY for women; P=0.724). However, a significant difference was
observed in all-cause mortality rates, with men showing a higher rate than women
(47.1/10,000 PY vs. 24.3/10,000 PY; P<0.001). In the prediabetes cohort,
3,435 MACE cases were recorded in men and 1,904 in women. The incidence rate of MACE
in women was slightly higher than in men, but this difference was not statistically
significant (88.0/10,000 PY for women vs. 92.8/10,000 PY for men; P=0.063). However,
there was a significant difference in all-cause mortality between men and women,
with men experiencing a higher rate (66.7/10,000 PY vs. 41.4/10,000 PY;
P<0.001).
By controlling for lifestyle and clinical factors at the initial health screening, we
evaluated the effects of lifestyle changes on major outcomes using a multivariate
model (
Tables 3,
4). In the prehypertension group, 7,376 patients (7.75%) had
already been diagnosed with T2D, and 2,601 (2.73%) were taking anti-hyperglycemic
medication. Similarly, in the prediabetes group, 11,577 patients (20.01%) had been
diagnosed with HTN, and 8,962 (15.49%) were on antihypertensive medication (
Table 1). To isolate the effects of medication,
the multivariate analysis accounted for the impact of antihypertensive drugs,
anti-hyperglycemic drugs, and aspirin. In men with prehypertension, the risk of MACE
increased if their lifestyle worsened (HR, 1.13; 95% CI, 1.04–1.23, P=0.004),
particularly if they gained weight (HR, 1.15; 95% CI, 1.03–1.28, P=0.010) or
started smoking (HR, 1.34; 95% CI, 1.17–1.55, P<0.001). For women with
prehypertension, the risk of MACE was higher for those who started smoking (HR,
1.69; 95% CI, 1.15–2.49, P=0.008) or reduced their physical activity (HR,
1.25; 95% CI, 1.06–1.47, P=0.010). Conversely, in men with prehypertension,
improving lifestyle factors reduced the risk of MACE (HR, 0.91; 95% CI,
0.84–0.99, P=0.025), particularly through smoking cessation (HR, 0.79; 95%
CI, 0.70–0.89, P<0.001), drinking less (HR, 1.09; 95% CI,
1.00–1.20, P=0.048), or increasing physical activity (HR, 0.91; 95% CI,
0.84–0.99, P=0.027). In men with prediabetes, those whose lifestyle factors
worsened had a 23% higher risk of MACE compared to those with no lifestyle changes
(HR, 1.23; 95% CI, 1.12–1.35, P<0.001). An increased risk of MACE was
also observed in those who gained weight (HR, 1.19; 95% CI, 1.06–1.33,
P=0.003), started smoking (HR, 1.41; 95% CI, 1.22–1.64, P<0.001), or
decreased their physical activity (HR, 1.21; 95% CI, 1.09–1.35,
P<0.001). Additionally, in men with prediabetes, a reduction in alcohol
consumption was linked to a higher risk of MACE (HR, 1.17; 95% CI, 1.07–1.29,
P=0.001). In women with prediabetes, the risk of MACE was 1.24 times higher for
those who gained weight compared to those with no change in BMI levels (HR, 1.24;
95% CI, 1.06–1.45, P=0.006). As weight change can be a consequence of
lifestyle changes, the association between unhealthy lifestyles, excluding BMI, and
MACE was evaluated. Among pre-hypertensive men, those whose lifestyles worsened had
a higher risk of MACE (HR, 1.10; 95% CI, 1.02–1.20, P=0.022). Among
pre-hypertensive women, those whose lifestyles improved tended to have a lower MACE
risk, although this association was not statistically significant (HR, 0.91; 95% CI,
0.81–1.01, P=0.072) (
Table 3,
Supplement 3). In the
prediabetes group, men whose lifestyles worsened showed a significantly higher risk
of MACE (HR, 1.23; 95% CI, 1.12–1.35, P<0.001), while there was no
significant difference in MACE risk among comparative female subjects (
Table 4,
Supplement 4).
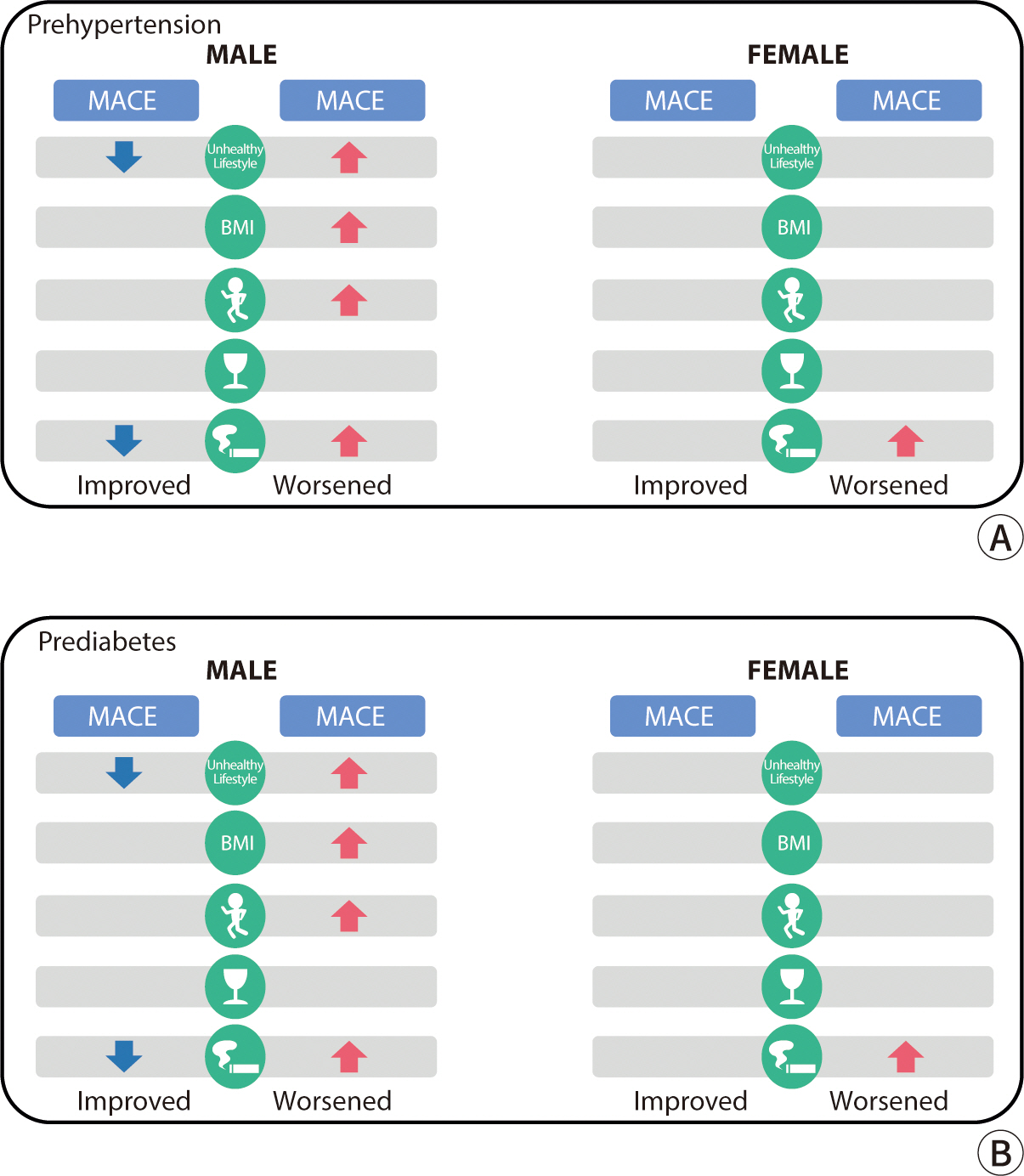
To mitigate the risk of reverse causality, a sensitivity analysis was conducted by
excluding cardiovascular events that occurred within two years following the
observation of lifestyle changes. In the prehypertension male group, the risk of
MACE increased among subjects who experienced a decline in lifestyle quality, gained
weight, decreased their physical activity frequency, or began smoking between
biennial screenings. Conversely, the risk decreased in those who improved their
lifestyles or quit smoking. In the prehypertension female group, an increase in MACE
risk was observed in subjects who started smoking (
Fig. 2A). In the prediabetes group, the MACE risk escalated in men who
worsened their lifestyle, gained weight, reduced their physical activity frequency,
or started smoking between the biennial screenings. In women, the risk increased
among those who gained weight (
Fig. 2B).




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download