Abstract
Atrial fibrillation (AF) is the most common sustained tachyarrhythmia and its increasing prevalence has resulted in a growing healthcare burden. A recent landmark randomized trial, the EAST-AFNET 4 (Early Treatment of Atrial Fibrillation for Stroke Prevention Trial), highlighted the importance of early rhythm control in AF, which was previously underemphasized. Rhythm control therapy includes antiarrhythmic drugs, direct-current cardioversion, and catheter ablation. Currently, catheter ablation is indicated for patients with AF who are either refractory or intolerant to antiarrhythmic drugs or who exhibit decreased left ventricular systolic function. Catheter ablation can be categorized according to the energy source used, including radiofrequency ablation (RFA), cryoablation, laser ablation, and the recently emerging pulsed field ablation (PFA). Catheter ablation techniques can also be divided into the point-by-point ablation method, which ablates the pulmonary vein (PV) antrum one point at a time, and the single-shot technique, which uses a spherical catheter to ablate the PV antrum in a single application. PFA is known to be applicable to both point-by-point and single-shot techniques and is expected to be promising owing to its tissue specificity, resulting in less collateral damage than catheter ablation involving thermal energy, such as RFA and cryoablation. In this review, we aimed to outline catheter ablation for rhythm control in AF by reviewing previous studies.
Atrial fibrillation (AF) is the most common sustained tachyarrhythmia and its global incidence and prevalence are increasing. AF is associated with increased morbidity and mortality related to stroke, congestive heart failure, impaired quality of life, and an increased risk of dementia [1-6]. The comprehensive management of AF, known as the “ABC pathway” suggested by the 2020 European Society of Cardiology Guidelines, includes stroke prevention, rate control, rhythm control, and management of comorbidities [5]. Rhythm control treatments include antiarrhythmic drugs, direct-current cardioversion, catheter ablation, and surgical ablation. Rhythm control therapy for AF has not been emphasized in the past. Many clinical trials, including the AFFIRM (Atrial Fibrillation Follow-up Investigation of Rhythm Management) and the RACE (Rate Control Versus Electrical Cardioversion) trials, which compared rate control and rhythm control treatments, did not demonstrate any clear benefits of the rhythm control strategy and, instead, reported higher rates of adverse events [7-12]. However, since the results of the EAST-AFNET 4 (Early Treatment of Atrial Fibrillation for Stroke Prevention Trial), the benefits of early rhythm control therapy for AF have been highlighted, and real-world data support the advantages of early rhythm control, marking a paradigm shift in AF management [13,14]. Differences in the classification of AF as paroxysmal or persistent, the timing of rhythm control treatment initiation, modalities of rhythm control treatment, extent of left atrial remodeling, and anticoagulation strategies have contributed to the different outcomes observed in recent studies compared with those of past trials. Recently, many studies have focused on early catheter ablation of AF. A recent randomized trial reported no difference in clinical outcomes even after delayed catheter ablation for up to 12 months [15]. However, that study had some limitations, as it utilized the referral date to a tertiary hospital, rather than the diagnosis date of AF, as the starting point for survival analysis. Other observational studies have shown that the shorter the time from the diagnosis of AF to catheter ablation, the better the clinical outcomes [16-19]. This article reviews rhythm control therapy for AF with a focus on catheter ablation.
The estimated prevalence of AF was 50 million cases worldwide in 2020. Using a back-calculation methodology, it is estimated that approximately 11% of the more than 5.6 million AF cases in the United States are undiagnosed [20-22]. According to South Korea’s statistics, the prevalence of AF has gradually increased, reaching 1.53% in 2015, approximately twice that in 2006, using data from the Korean National Health Insurance database. The projected prevalence of AF is expected to increase by approximately 5.81% by 2060 [23]. The risk of AF increases with age. According to the Framingham Heart Study, AF will occur in 26% of Caucasian men and 23% of Caucasian women after 40 years of age [24]. There are various risk factors for AF. These include modifiable risk factors such as hypertension, obesity, endurance exercise, sleep apnea, thyroid disease, and alcohol consumption, as well as non-modifiable risk factors such as age, sex, family history, height, ethnicity, and history of valvular heart disease [25-29].
The concept of AF catheter ablation originated from the maze procedure, which was developed by Cox et al. [30]. This procedure involves the creation of transmural incisions in the left atrium. It has evolved into Cox-Maze III, which has impressive efficacy, achieving >90% freedom from AF recurrence at 12 months [31-33].
The Cox-Maze procedure involves making incisions around the pulmonary veins (PVs); however, the significance of the PVs in AF was not widely recognized until the study by Haïssaguerre et al. [34]. Following that study, which identified PVs as a key source of ectopic beats in AF, catheter ablation has emerged as a treatment option for AF [34,35]. Subsequently, Pappone et al. [36,37] performed PV antral ablation using three-dimensional (3D) electro-anatomic mapping for AF management. Consequently, PV isolation (PVI) is a crucial milestone in catheter ablation for AF, and circumferential PVI has become the standard treatment for AF catheter ablation. In cases of persistent AF, additional ablation procedures, such as linear ablation in the left atrium, ablation of areas with fragmented local electrograms, and ablation of ganglionated plexi, have been attempted to achieve better outcomes. However, the results of these additional ablation procedures have not been satisfactory [38-42], and they have received class IIb recommendation in the clinical guidelines [5,43].
Radiofrequency (RF) is currently the most widely used energy source [5,44]. While the RF spectrum ranges from 30 to 300 GHz, the energy sources used for ablation typically operate at frequencies between 350 and 750 kHz [45]. When an RF current is delivered through the catheter tip electrodes, it generates resistive heating owing to the high resistance of the tissues, converting the RF energy into the thermal energy necessary for ablation [46]. Significant advancements have been made in the RF catheter ablation (RFCA) procedure through the development and refinement of two key technologies: open-irrigated catheters and contact force (CF)-sensing RF technology (Fig. 1A). CF-sensing RF technology has improved long-term procedural effectiveness and enabled operators to avoid excessive force and reduce procedure-related complications such as cardiac tamponade and rupture, thereby enhancing safety and efficiency [47-51]. Open-irrigated catheters decrease thrombus and char formation at the catheter tip, providing more accurate temperature feedback and improving safety [52]. The recently developed TactiFlex catheter (Abbott Inc., Chicago, IL, USA) exhibits enhanced flexibility and delivers irrigation flow directly to the tissue-tip interface, resulting in a lower incidence of steam pops than previous open-irrigated catheters [53].
Until recently, RFCA was performed using conventional power (20–40 W) for long durations (20–40 seconds). However, this method faced several issues: optimal lesion formation was highly dependent on catheter stability, procedure times were relatively long, and the incidence of PV reconnections was high [48]. To address these challenges, efforts have been made to use high power (40–90 W) with relatively short durations (≤15 seconds) for RFCA [54]. High-power short-duration (HPSD) ablation has been attempted based on the theoretical background that it creates immediate and transmural lesions while overcoming catheter instability and minimizing subsequent tissue edema by limiting the application time. Additionally, reduced passive heat conduction may result in less collateral damage to the surrounding structures. However, the safety and efficacy windows are narrow, and the lesion depth is shallow, which may make HPSD ablation less suitable for thick tissues [55]. A human study indicated that while the efficacy and incidence of esophageal injury were similar, the procedure time was significantly shorter in patients with HPSD [56]. Additionally, a meta-analysis showed that although conventional RFCA was superior in terms of freedom from atrial arrhythmia, HPSD was advantageous not only in reducing acute PV reconnection but also in shortening the procedure time [57]. Very high-power, short-duration, temperature-controlled RFCA has also been applied in clinical settings. The QDOT MICRO catheter (Biosense Webster Inc., Diamond Bar, CA, USA), the first and only temperature-controlled CF-sensing catheter, is based on a spring-loaded CF catheter platform. It features two ablation modes: a very high-power, short-duration (vHPSD) mode (90 W, ≤4 seconds) and a conventional-power temperature-controlled (CPTC) mode (≤50 W). The vHPSD mode automatically modulates the power upon reaching maximum temperature, whereas the CPTC mode adjusts the irrigation flow and power based on temperature feedback to maintain a stable tissue temperature. Studies on vHPSD ablation have indicated that it is more efficient and effective than conventional RFCA without compromising safety. vHPSD ablation is now widely used in clinical practice [58,59].
The cryoballoon catheter technique, in which cryoenergy is applied via a balloon catheter introduced into the left atrium through a single transseptal puncture, is also gaining attention owing to its efficacy and safety. Through the application of cryoenergy at mild freezing temperatures ranging from –10°C to –25°C, extracellular ice formation induces cell dehydration, intracellular acidification, and ionic imbalance. These physiological disruptions lead to cell membrane injury and the cessation of cardiac electrical activity. The duration of exposure to freezing temperatures correlates with the extent of permanent cellular damage. Conversely, at extreme freezing temperatures below –50°C, intracellular ice formation occurs, resulting in immediate and permanent tissue injury [60,61]. This technique, specialized for PVI using a single-shot procedure, has a relatively lower complication rate and shorter procedure time than conventional RFCA. Cryoablation is non-inferior to RFCA in reducing clinical recurrence after paroxysmal AF ablation [62]. However, in the case of persistent AF, the effectiveness of cryoablation is limited because of its primary design for creating antral PV lesions, making it less effective in targeting non-PV triggers than RFCA, which uses a point-by-point ablation technique [63-66]. A circular mapping catheter is introduced into the PV via an over-the-wire balloon catheter to confirm PVI after ablation and to monitor PV potentials in real time during the procedure. Phrenic nerve injury (PNI) is the most common complication of cryoablation, occurring in approximately 1.1% to 6.2% of cases [62,67-70]; however, it is usually temporary. According to the recent YETI registry (worldwide survey on outcome after iatrogenic phrenic nerve injury during cryoballoon based pulmonary vein isolation), when using second-generation cryoballoons, PNI occurs in 4.2% of cases, with 53.9% recovering by the end of the procedure, only 0.1% persisting with permanent PNI, and 0.06% with permanent and symptomatic PNI after 12 months (Fig. 1B).
Laser balloon ablation (HeartLight; CardioFocus, Marlborough, MA, USA) is one method that uses laser energy to induce circumferential cell necrosis around the PV and achieve PVI. A compliant balloon adjusts to the PV size irrespective of PV dimensions and then emits a laser source onto the PV antral tissue [71]. The laser energy is titrated from 5.5 to 12 W [72]. PNI is a frequent but temporary complication. A recent meta-analysis showed that this laser balloon technique was comparable to conventional AF ablation techniques (Fig. 1C) [73].
Conventionally, AF catheter ablation involves point-by-point RF ablation with an irrigated catheter. However, this approach can be problematic owing to its complexity, longer procedure time, and the potential for increased procedure-related complications. Additionally, extensive left atrial mapping using multiple catheters is required. Gaps between the ablation lines are also inevitable during point-by-point ablation.
Efforts have been made to apply single-shot techniques using RF energy. Multi-electrode radiofrequency ablation catheters such as the PV ablation catheter (PVAC; Medtronic Inc., Minneapolis, MN, USA) and nMARQ (Biosense Webster Inc.) have been developed. PVAC demonstrated high procedural success rates and shorter procedure and fluoroscopy times and did not require a 3D mapping system. However, electrode overheating and tissue contact can lead to a high incidence of silent cerebral lesions. Consequently, despite the development of the improved PVAC Gold system, it was eventually withdrawn from the market [74-77]. During the study period for nMARQ, there were two cases of serious adverse events involving atrioesophageal fistulae, both of which were fatal, leading to its withdrawal from the market owing to safety concerns [78-80]. Subsequently, other catheters such as HELIOSTAR (Biosense Webster Inc.) and LUMINIZE (Apama Medical/Boston Scientific Corp., Marlborough, MA, USA) were introduced, but they are no longer commercially available [81,82]. Therefore, currently, the single-shot techniques available on the market are cryoablation using cryoenergy and, as will be discussed next, pulsed field ablation (PFA).
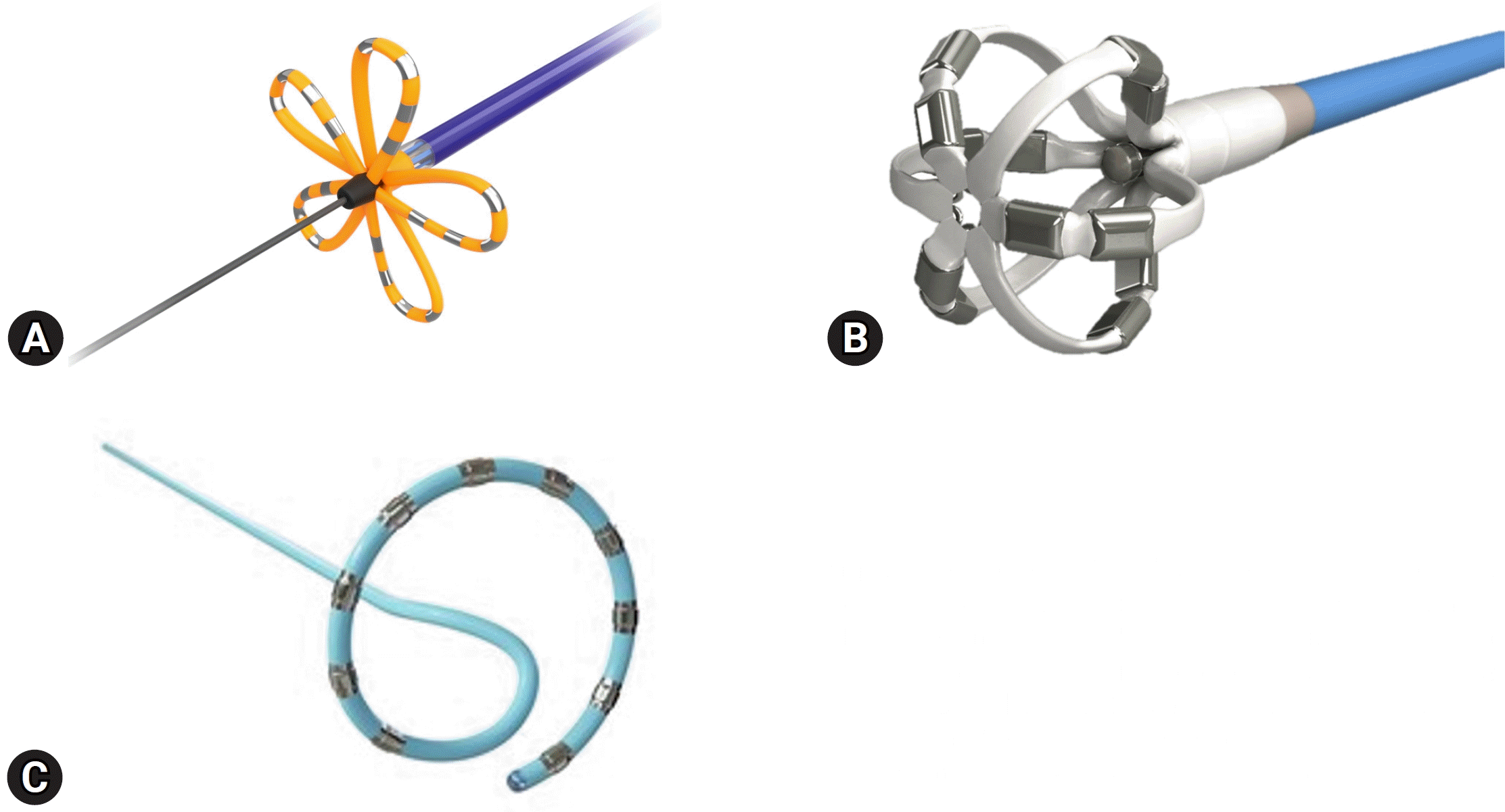
PFA is a novel nonthermal ablation technique that utilizes irreversible electroporation (IRE) by applying an electrical field to the target tissue. When high-voltage currents are applied to the target tissue, cell membrane permeabilization increases owing to the formation of irreversible pores, which leads to the leakage of cell contents and finally, cell death [83,84]. Various tissues have specific thresholds for necrosis induced by PFA, with myocardial tissue having the lowest threshold, making it the most sensitive. Consequently, compared to other energy sources, PFA has the advantage of minimizing collateral damage, including esophageal and nerve injuries, when ablating the myocardial tissue. Additionally, because of the tissue selectivity of IRE and its rapid energy delivery, PFA is expected to have a relatively short procedure time. Experimental data from animal studies have demonstrated the safety of IRE ablation near important structures, including the coronary arteries, right phrenic nerve, and esophagus [85-87]. Observational studies on humans have also been published [88,89]. Recently, the results of a randomized controlled trial (ADVENT [Pulsed Field or Conventional Thermal Ablation for Paroxysmal Atrial Fibrillation] trial) comparing PFA to conventional thermal ablation for paroxysmal AF were published, which demonstrated that PFA is non-inferior to conventional thermal ablation methods such as radiofrequency ablation and cryoablation in terms of procedural efficacy and safety at 1 year [90]. A recent study using a variable-loop circular catheter (VARIPULSE, Biosense Webster Inc.) with a 3D mapping system (CARTO, Biosense Webster Inc.) confirmed the safety and effectiveness of this novel mapping-integrated PFA system [91]. Currently, PFA is used in both point-by-point ablation and single-shot techniques. The FARAWAVE catheter (Boston Scientific Corp.) is a five-spline-shaped PFA catheter that performs ablation by rotating in the PV antrum after positioning and changing its morphology to balloon and flower shapes [89] (Fig. 2A). The OMNYPULSE catheter (Biosense Webster Inc.) is a multi-electrode CF-sensing PFA catheter with a 12-mm-diameter spherical cage [92] (Fig. 2B). It is compatible with the 3D CARTO electroanatomical mapping system (CARTO3, Biosense Webster Inc.). The VARIPULSE multi-electrode variable-loop circular catheter (Biosense Webster Inc.) can also perform short-duration, high-voltage bipolar biphasic PFA using the CARTO3 system [91] (Fig. 2C). Additionally, the TRUPULSE 2 multimodality generator (Biosense Webster Inc.) enables point-by-point PFA using conventional CF-sensing THERMOCOOL SMARTTOUCH catheters (Biosense Webster Inc.) and is compatible with the CARTO3 system [93]. OMNYPULSE, VARIPULSE, and TRUPULSE 2 are currently under development.
In this review, we discussed invasive rhythm control strategies for AF. Conventional RFCA and cryoballoon catheter ablation using cryoenergy are well-established, and recent developments in PFA have either been utilized clinically or prepared for clinical use. In the past, rhythm control for AF was underemphasized; however, with the development of antiarrhythmic drugs and catheter ablation technologies, its importance has been increasingly recognized, particularly the significance of early rhythm control. Therefore, rhythm control therapy, including catheter ablation techniques, should always be considered in comprehensive management strategies for patients with AF.
Notes
Acknowledgments
We thank the Yeungnam University College of Medicine research support team (design director: Hyun Yoon Choi) for providing the illustrations used in this study.
References
1. Marijon E, Le Heuzey JY, Connolly S, Yang S, Pogue J, Brueckmann M, et al. Causes of death and influencing factors in patients with atrial fibrillation: a competing-risk analysis from the randomized evaluation of long-term anticoagulant therapy study. Circulation. 2013; 128:2192–201.
2. Camm AJ, Amarenco P, Haas S, Hess S, Kirchhof P, Kuhls S, et al. XANTUS: a real-world, prospective, observational study of patients treated with rivaroxaban for stroke prevention in atrial fibrillation. Eur Heart J. 2016; 37:1145–53.
3. Kim D, Yang PS, Jang E, Yu HT, Kim TH, Uhm JS, et al. Increasing trends in hospital care burden of atrial fibrillation in Korea, 2006 through 2015. Heart. 2018; 104:2010–7.
4. Bunch TJ. Atrial fibrillation and dementia. Circulation. 2020; 142:618–20.
5. Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C, et al. 2020 ESC guideline for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2021; 42:373–498.
6. Giannone ME, Filippini T, Whelton PK, Chiari A, Vitolo M, Boriani G, et al. Atrial fibrillation and the risk of early-onset dementia: a systematic review and meta-analysis. J Am Heart Assoc. 2022; 11:e025653.
7. Roy D, Talajic M, Nattel S, Wyse DG, Dorian P, Lee KL, et al. Rhythm control versus rate control for atrial fibrillation and heart failure. N Engl J Med. 2008; 358:2667–77.
8. Opolski G, Torbicki A, Kosior DA, Szulc M, Wozakowska-Kaplon B, Kolodziej P, et al. Rate control vs rhythm control in patients with nonvalvular persistent atrial fibrillation: the results of the Polish How to Treat Chronic Atrial Fibrillation (HOT CAFE) Study. Chest. 2004; 126:476–86.
9. Singh SN, Singh BN, Reda DJ, Fye CL, Ezekowitz MD, Fletcher RD, et al. Comparison of sotalol versus amiodarone in maintaining stability of sinus rhythm in patients with atrial fibrillation (Sotalol-Amiodarone Fibrillation Efficacy Trial [Safe-T]). Am J Cardiol. 2003; 92:468–72.
10. Wyse DG, Waldo AL, DiMarco JP, Domanski MJ, Rosenberg Y, Schron EB, et al. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 2002; 347:1825–33.
11. Van Gelder IC, Hagens VE, Bosker HA, Kingma JH, Kamp O, Kingma T, et al. A comparison of rate control and rhythm control in patients with recurrent persistent atrial fibrillation. N Engl J Med. 2002; 347:1834–40.
12. Hohnloser SH, Kuck KH, Lilienthal J. Rhythm or rate control in atrial fibrillation: Pharmacological Intervention in Atrial Fibrillation (PIAF). A randomised trial. Lancet. 2000; 356:1789–94.
13. Kim D, Yang PS, You SC, Sung JH, Jang E, Yu HT, et al. Treatment timing and the effects of rhythm control strategy in patients with atrial fibrillation: nationwide cohort study. BMJ. 2021; 373:n991.
14. Kirchhof P, Camm AJ, Goette A, Brandes A, Eckardt L, Elvan A, et al. Early rhythm-control therapy in patients with atrial fibrillation. N Engl J Med. 2020; 383:1305–16.
15. Kalman JM, Al-Kaisey AM, Parameswaran R, Hawson J, Anderson RD, Lim M, et al. Impact of early vs. delayed atrial fibrillation catheter ablation on atrial arrhythmia recurrences. Eur Heart J. 2023; 44:2447–54.
16. Bunch TJ, May HT, Bair TL, Johnson DL, Weiss JP, Crandall BG, et al. Increasing time between first diagnosis of atrial fibrillation and catheter ablation adversely affects long-term outcomes. Heart Rhythm. 2013; 10:1257–62.
17. De Greef Y, Schwagten B, Chierchia GB, de Asmundis C, Stockman D, Buysschaert I. Diagnosis-to-ablation time as a predictor of success: early choice for pulmonary vein isolation and long-term outcome in atrial fibrillation: results from the Middelheim-PVI Registry. Europace. 2018; 20:589–95.
18. Yu HT, Kim IS, Kim TH, Uhm JS, Kim JY, Joung B, et al. Persistent atrial fibrillation over 3 years is associated with higher recurrence after catheter ablation. J Cardiovasc Electrophysiol. 2020; 31:457–64.
19. De Greef Y, Bogaerts K, Sofianos D, Buysschaert I. Impact of diagnosis-to-ablation time on AF recurrence: pronounced the first 3 years, irrelevant thereafter. JACC Clin Electrophysiol. 2023; 9:2263–72.
20. Turakhia MP, Guo JD, Keshishian A, Delinger R, Sun X, Ferri M, et al. Contemporary prevalence estimates of undiagnosed and diagnosed atrial fibrillation in the United States. Clin Cardiol. 2023; 46:484–93.
21. Tsao CW, Aday AW, Almarzooq ZI, Anderson CA, Arora P, Avery CL, et al. Heart disease and stroke statistics: 2023 update. A report from the American Heart Association. Circulation. 2023; 147:e93–621.
22. Chugh SS, Havmoeller R, Narayanan K, Singh D, Rienstra M, Benjamin EJ, et al. Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation. 2014; 129:837–47.
23. Kim D, Yang PS, Jang E, Yu HT, Kim TH, Uhm JS, et al. 10-year nationwide trends of the incidence, prevalence, and adverse outcomes of non-valvular atrial fibrillation nationwide health insurance data covering the entire Korean population. Am Heart J. 2018; 202:20–6.
24. Lloyd-Jones DM, Wang TJ, Leip EP, Larson MG, Levy D, Vasan RS, et al. Lifetime risk for development of atrial fibrillation: the Framingham Heart Study. Circulation. 2004; 110:1042–6.
25. Gorenek B, Pelliccia A, Benjamin EJ, Boriani G, Crijns HJ, Fogel RI, et al. European Heart Rhythm Association (EHRA)/European Association of Cardiovascular Prevention and Rehabilitation (EACPR) position paper on how to prevent atrial fibrillation endorsed by the Heart Rhythm Society (HRS) and Asia Pacific Heart Rhythm Society (APHRS). Europace. 2017; 19:190–225.
26. Miller JD, Aronis KN, Chrispin J, Patil KD, Marine JE, Martin SS, et al. Obesity, exercise, obstructive sleep apnea, and modifiable atherosclerotic cardiovascular disease risk factors in atrial fibrillation. J Am Coll Cardiol. 2015; 66:2899–906.
27. January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014; 64:e1–76.
28. European Heart Rhythm Association; European Association for Cardio-Thoracic Surgery, Camm AJ, Kirchhof P, Lip GY, Schotten U, et al. Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J. 2010; 31:2369–429.
29. Benjamin EJ, Levy D, Vaziri SM, D’Agostino RB, Belanger AJ, Wolf PA. Independent risk factors for atrial fibrillation in a population-based cohort. The Framingham Heart Study. JAMA. 1994; 271:840–4.
30. Cox JL, Schuessler RB, D’Agostino HJ, Stone CM, Chang BC, Cain ME, et al. The surgical treatment of atrial fibrillation. III. Development of a definitive surgical procedure. J Thorac Cardiovasc Surg. 1991; 101:569–83.
31. Prasad SM, Maniar HS, Camillo CJ, Schuessler RB, Boineau JP, Sundt TM, et al. The Cox maze III procedure for atrial fibrillation: long-term efficacy in patients undergoing lone versus concomitant procedures. J Thorac Cardiovasc Surg. 2003; 126:1822–8.
32. Gaynor SL, Schuessler RB, Bailey MS, Ishii Y, Boineau JP, Gleva MJ, et al. Surgical treatment of atrial fibrillation: predictors of late recurrence. J Thorac Cardiovasc Surg. 2005; 129:104–11.
33. Johansson BI, Väärt O, Edvardsson N, Nyström B, Scherstén H, Karlsson T, et al. Low mortality and low rate of perceived and documented arrhythmias after Cox maze III surgery for atrial fibrillation. Pacing Clin Electrophysiol. 2014; 37:147–56.
34. Haïssaguerre M, Shah DC, Jaïs P, Hocini M, Yamane T, Deisenhofer I, et al. Electrophysiological breakthroughs from the left atrium to the pulmonary veins. Circulation. 2000; 102:2463–5.
35. Haïssaguerre M, Jaïs P, Shah DC, Takahashi A, Hocini M, Quiniou G, et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 1998; 339:659–66.
36. Pappone C, Oreto G, Rosanio S, Vicedomini G, Tocchi M, Gugliotta F, et al. Atrial electroanatomic remodeling after circumferential radiofrequency pulmonary vein ablation: efficacy of an anatomic approach in a large cohort of patients with atrial fibrillation. Circulation. 2001; 104:2539–44.
37. Pappone C, Rosanio S, Oreto G, Tocchi M, Gugliotta F, Vicedomini G, et al. Circumferential radiofrequency ablation of pulmonary vein ostia: a new anatomic approach for curing atrial fibrillation. Circulation. 2000; 102:2619–28.
38. Verma A, Jiang CY, Betts TR, Chen J, Deisenhofer I, Mantovan R, et al. Approaches to catheter ablation for persistent atrial fibrillation. N Engl J Med. 2015; 372:1812–22.
39. He X, Zhou Y, Chen Y, Wu L, Huang Y, He J. Left atrial posterior wall isolation reduces the recurrence of atrial fibrillation: a meta-analysis. J Interv Card Electrophysiol. 2016; 46:267–74.
40. Scott PA, Silberbauer J, Murgatroyd FD. The impact of adjunctive complex fractionated atrial electrogram ablation and linear lesions on outcomes in persistent atrial fibrillation: a meta-analysis. Europace. 2016; 18:359–67.
41. Lee JM, Shim J, Park J, Yu HT, Kim TH, Park JK, et al. The electrical isolation of the left atrial posterior wall in catheter ablation of persistent atrial fibrillation. JACC Clin Electrophysiol. 2019; 5:1253–61.
42. Kistler PM, Chieng D, Sugumar H, Ling LH, Segan L, Azzopardi S, et al. Effect of catheter ablation using pulmonary vein isolation with vs without posterior left atrial wall isolation on atrial arrhythmia recurrence in patients with persistent atrial fibrillation: the CAPLA randomized clinical trial. JAMA. 2023; 329:127–35.
43. Writing Committee Members, Joglar JA, Chung MK, Armbruster AL, Benjamin EJ, Chyou JY, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2024; 83:109–279.
44. Calkins H, Hindricks G, Cappato R, Kim YH, Saad EB, Aguinaga L, et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm. 2017; 14:e275–444.
45. Morady F, Scheinman MM. Transvenous catheter ablation of a posteroseptal accessory pathway in a patient with the Wolff-Parkinson-White syndrome. N Engl J Med. 1984; 310:705–7.
46. Houmsse M, Daoud EG. Biophysics and clinical utility of irrigated-tip radiofrequency catheter ablation. Expert Rev Med Devices. 2012; 9:59–70.
47. Natale A, Reddy VY, Monir G, Wilber DJ, Lindsay BD, McElderry HT, et al. Paroxysmal AF catheter ablation with a contact force sensing catheter: results of the prospective, multicenter SMART-AF trial. J Am Coll Cardiol. 2014; 64:647–56.
48. Reddy VY, Pollak S, Lindsay BD, McElderry HT, Natale A, Kantipudi C, et al. Relationship between catheter stability and 12-month success after pulmonary vein isolation: a subanalysis of the SMART-AF trial. JACC Clin Electrophysiol. 2016; 2:691–9.
49. Macle L, Frame D, Gache LM, Monir G, Pollak SJ, Boo LM. Atrial fibrillation ablation with a spring sensor-irrigated contact force-sensing catheter compared with other ablation catheters: systematic literature review and meta-analysis. BMJ Open. 2019; 9:e023775.
50. Duytschaever M, Vijgen J, De Potter T, Scherr D, Van Herendael H, Knecht S, et al. Standardized pulmonary vein isolation workflow to enclose veins with contiguous lesions: the multicentre VISTAX trial. Europace. 2020; 22:1645–52.
51. Natale A, Monir G, Patel AM, Fishel RS, Marchlinski FE, Delaughter MC, et al. Long-term safety and effectiveness of paroxysmal atrial fibrillation ablation using a porous tip contact force-sensing catheter from the SMART SF trial. J Interv Card Electrophysiol. 2021; 61:63–9.
52. Habibi M, Berger RD, Calkins H. Radiofrequency ablation: technological trends, challenges, and opportunities. Europace. 2021; 23:511–9.
53. Suga K, Kato H, Akita S, Sakurai T, Ota R, Okada T, et al. Optimal ablation settings of TactiFlex SE laser-cut irrigated-tip catheter: comparison with ThermoCool SmartTouch SurroundFlow porous irrigated-tip catheter. J Interv Card Electrophysiol. 2024 Apr 4 [Epub]. https://doi.org/10.1007/s10840-024-01797-9.
54. Naniwadekar A, Dukkipati SR. High-power short-duration ablation of atrial fibrillation: a contemporary review. Pacing Clin Electrophysiol. 2021; 44:528–40.
55. Leshem E, Zilberman I, Tschabrunn CM, Barkagan M, Contreras-Valdes FM, Govari A, et al. High-power and short-duration ablation for pulmonary vein isolation: biophysical characterization. JACC Clin Electrophysiol. 2018; 4:467–79.
56. Baher A, Kheirkhahan M, Rechenmacher SJ, Marashly Q, Kholmovski EG, Siebermair J, et al. High-power radiofrequency catheter ablation of atrial fibrillation: using late gadolinium enhancement magnetic resonance imaging as a novel index of esophageal injury. JACC Clin Electrophysiol. 2018; 4:1583–94.
57. Ravi V, Poudyal A, Abid QU, Larsen T, Krishnan K, Sharma PS, et al. High-power short duration vs. conventional radiofrequency ablation of atrial fibrillation: a systematic review and meta-analysis. Europace. 2021; 23:710–21.
58. Osorio J, Hussein AA, Delaughter MC, Monir G, Natale A, Dukkipati S, et al. Very high-power short-duration, temperature-controlled radiofrequency ablation in paroxysmal atrial fibrillation: the prospective multicenter Q-FFICIENCY trial. JACC Clin Electrophysiol. 2023; 9:468–80.
59. Hussein AA, Delaughter MC, Monir G, Natale A, Dukkipati S, Oza S, et al. Paroxysmal atrial fibrillation ablation with a novel temperature-controlled CF-sensing catheter: Q-FFICIENCY clinical and healthcare utilization benefits. J Cardiovasc Electrophysiol. 2023; 34:2493–503.
60. Andrade JG, Khairy P, Dubuc M. Catheter cryoablation: biology and clinical uses. Circ Arrhythm Electrophysiol. 2013; 6:218–27.
61. Avitall B, Kalinski A. Cryotherapy of cardiac arrhythmia: from basic science to the bedside. Heart Rhythm. 2015; 12:2195–203.
62. Kuck KH, Brugada J, Fürnkranz A, Metzner A, Ouyang F, Chun KR, et al. Cryoballoon or radiofrequency ablation for paroxysmal atrial fibrillation. N Engl J Med. 2016; 374:2235–45.
63. Wazni OM, Dandamudi G, Sood N, Hoyt R, Tyler J, Durrani S, et al. Cryoballoon ablation as initial therapy for atrial fibrillation. N Engl J Med. 2021; 384:316–24.
64. Straube F, Pongratz J, Kosmalla A, Brueck B, Riess L, Hartl S, et al. Cryoballoon ablation strategy in persistent atrial fibrillation. Front Cardiovasc Med. 2021; 8:758408.
65. Andrade JG, Wells GA, Deyell MW, Bennett M, Essebag V, Champagne J, et al. Cryoablation or drug therapy for initial treatment of atrial fibrillation. N Engl J Med. 2021; 384:305–15.
66. Su WW, Reddy VY, Bhasin K, Champagne J, Sangrigoli RM, Braegelmann KM, et al. Cryoballoon ablation of pulmonary veins for persistent atrial fibrillation: results from the multicenter STOP Persistent AF trial. Heart Rhythm. 2020; 17:1841–7.
67. Metzner A, Rausch P, Lemes C, Reissmann B, Bardyszewski A, Tilz R, et al. The incidence of phrenic nerve injury during pulmonary vein isolation using the second-generation 28 mm cryoballoon. J Cardiovasc Electrophysiol. 2014; 25:466–70.
68. Fürnkranz A, Bordignon S, Schmidt B, Perrotta L, Dugo D, De Lazzari M, et al. Incidence and characteristics of phrenic nerve palsy following pulmonary vein isolation with the second-generation as compared with the first-generation cryoballoon in 360 consecutive patients. Europace. 2015; 17:574–8.
69. Abugattas JP, de Asmundis C, Iacopino S, Salghetti F, Takarada K, Coutiño HE, et al. Phrenic nerve injury during right inferior pulmonary vein ablation with the second-generation cryoballoon: clinical, procedural, and anatomical characteristics. Europace. 2018; 20:e156–63.
70. Hoffmann E, Straube F, Wegscheider K, Kuniss M, Andresen D, Wu LQ, et al. Outcomes of cryoballoon or radiofrequency ablation in symptomatic paroxysmal or persistent atrial fibrillation. Europace. 2019; 21:1313–24.
71. Dukkipati SR, Cuoco F, Kutinsky I, Aryana A, Bahnson TD, Lakkireddy D, et al. Pulmonary vein isolation using the visually guided laser balloon: a prospective, multicenter, and randomized comparison to standard radiofrequency ablation. J Am Coll Cardiol. 2015; 66:1350–60.
72. Bordignon S, Chun KR, Gunawardene M, Urban V, Kulikoglu M, Miehm K, et al. Energy titration strategies with the endoscopic ablation system: lessons from the high-dose vs. low-dose laser ablation study. Europace. 2013; 15:685–9.
73. Reynolds MR, Zheng Q, Doros G. Laser balloon ablation for AF: a systematic review and meta-analysis. J Cardiovasc Electrophysiol. 2018; 29:1363–70.
74. Weber S, Höher M, Schultes D. First results and follow-up of a second-generation circular mapping and ablation catheter. J Interv Card Electrophysiol. 2016; 47:213–9.
75. Mönnig G, Eckardt L. Multielectrode Pulmonary Vein Ablation Catheter (PVAC(®)): current data on results and risks. Herzschrittmacherther Elektrophysiol. 2014; 25:236–40.
76. Gaita F, Leclercq JF, Schumacher B, Scaglione M, Toso E, Halimi F, et al. Incidence of silent cerebral thromboembolic lesions after atrial fibrillation ablation may change according to technology used: comparison of irrigated radiofrequency, multipolar nonirrigated catheter and cryoballoon. J Cardiovasc Electrophysiol. 2011; 22:961–8.
77. Bulava A, Haniš J, Sitek D, Ošmera O, Karpianus D, Snorek M, et al. Catheter ablation for paroxysmal atrial fibrillation: a randomized comparison between multielectrode catheter and point-by-point ablation. Pacing Clin Electrophysiol. 2010; 33:1039–46.
78. Vurma M, Dang L, Brunner-La Rocca HP, Sütsch G, Attenhofer-Jost CH, Duru F, et al. Safety and efficacy of the nMARQ catheter for paroxysmal and persistent atrial fibrillation. Europace. 2016; 18:1164–9.
79. Mahida S, Hooks DA, Nentwich K, Ng GA, Grimaldi M, Shin DI, et al. nMARQ ablation for atrial fibrillation: results from a multicenter study. J Cardiovasc Electrophysiol. 2015; 26:724–9.
80. Shin DI, Kirmanoglou K, Eickholt C, Schmidt J, Clasen L, Butzbach B, et al. Initial results of using a novel irrigated multielectrode mapping and ablation catheter for pulmonary vein isolation. Heart Rhythm. 2014; 11:375–83.
81. Reddy VY, Al-Ahmad A, Aidietis A, Daly M, Melton I, Hu Y, et al. A novel visually guided radiofrequency balloon ablation catheter for pulmonary vein isolation: one-year outcomes of the multicenter AF-FICIENT I trial. Circ Arrhythm Electrophysiol. 2021; 14:e009308.
82. Dhillon GS, Honarbakhsh S, Di Monaco A, Coling AE, Lenka K, Pizzamiglio F, et al. Use of a multi-electrode radiofrequency balloon catheter to achieve pulmonary vein isolation in patients with paroxysmal atrial fibrillation: 12-month outcomes of the RADIANCE study. J Cardiovasc Electrophysiol. 2020; 31:1259–69.
83. Boersma L. New energy sources and technologies for atrial fibrillation catheter ablation. Europace. 2022; 24(Suppl 2):ii44–51.
84. Maor E, Sugrue A, Witt C, Vaidya VR, DeSimone CV, Asirvatham SJ, et al. Pulsed electric fields for cardiac ablation and beyond: a state-of-the-art review. Heart Rhythm. 2019; 16:1112–20.
85. Neven K, van Es R, van Driel V, van Wessel H, Fidder H, Vink A, et al. Acute and long-term effects of full-power electroporation ablation directly on the porcine esophagus. Circ Arrhythm Electrophysiol. 2017; 10:e004672.
86. van Driel VJ, Neven K, van Wessel H, Vink A, Doevendans PA, Wittkampf FH. Low vulnerability of the right phrenic nerve to electroporation ablation. Heart Rhythm. 2015; 12:1838–44.
87. du Pré BC, van Driel VJ, van Wessel H, Loh P, Doevendans PA, Goldschmeding R, et al. Minimal coronary artery damage by myocardial electroporation ablation. Europace. 2013; 15:144–9.
88. Ekanem E, Reddy VY, Schmidt B, Reichlin T, Neven K, Metzner A, et al. Multi-national survey on the methods, efficacy, and safety on the post-approval clinical use of pulsed field ablation (MANIFEST-PF). Europace. 2022; 24:1256–66.
89. Reddy VY, Neuzil P, Koruth JS, Petru J, Funosako M, Cochet H, et al. Pulsed field ablation for pulmonary vein isolation in atrial fibrillation. J Am Coll Cardiol. 2019; 74:315–26.
90. Reddy VY, Gerstenfeld EP, Natale A, Whang W, Cuoco FA, Patel C, et al. Pulsed field or conventional thermal ablation for paroxysmal atrial fibrillation. N Engl J Med. 2023; 389:1660–71.
91. Duytschaever M, De Potter T, Grimaldi M, Anic A, Vijgen J, Neuzil P, et al. Paroxysmal atrial fibrillation ablation using a novel variable-loop biphasic pulsed field ablation catheter integrated with a 3-dimensional mapping system: 1-year outcomes of the multicenter inspIRE study. Circ Arrhythm Electrophysiol. 2023; 16:e011780.
92. Di Biase L, Marazzato J, Govari A, Altman A, Beeckler C, Keyes J, et al. Pulsed field ablation index-guided ablation for lesion formation: impact of contact force and number of applications in the ventricular model. Circ Arrhythm Electrophysiol. 2024; 17:e012717.
93. Di Biase L, Marazzato J, Zou F, Lin A, Grupposo V, Chowdhuri N, et al. Point-by-point pulsed field ablation using a multimodality generator and a contact force-sensing ablation catheter: comparison with radiofrequency ablation in a remapped chronic swine heart. Circ Arrhythm Electrophysiol. 2023; 16:663–71.
Fig. 1.
Types of ablation catheters for atrial fibrillation. (A) Open-irrigated contact force-sensing radiofrequency ablation catheter (SMARTTOUCH catheter; Biosense Webster Inc., Diamond Bar, CA, USA). (B) Cryoballoon catheter. (C) Laser balloon catheter (HeartLight; CardioFocus, Marlborough, MA, USA). Illustrated by Yeungnam University College of Medicine research support team.

Fig. 2.
Types of pulsed field ablation catheters for atrial fibrillation. (A) FARAWAVE (Boston Scientific Corp., Marlborough, MA, USA). Images provided courtesy of Boston Scientific Corp. (©2024 Boston Scientific Corp. or its affiliates. All rights reserved). (B) OMNYPULSE (Biosense Webster Inc., Diamond Bar, CA, USA) and (C) VARIPULSE (Biosense Webster Inc.). Adapted with permission from Johnson & Johnson.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download