Abstract
The anterior choroidal artery (AChA) supplies the corticospinal tract related to the motor function of the upper and lower limbs. Aneurysms arising at the AChA are not common and exhibit various patterns of involvement of the AChA. Hence, managing an AChA aneurysm poses a high risk of neurological deficits. We report technical issues related to the outcome of coil embolization for unruptured AChA aneurysms. We obtained Institutional Review Board approval for this study. In the past 2 years, 18 consecutive patients (6 males and 12 females, aged 32–68) with unruptured AChA aneurysms were treated using embolization. We present technical details, pre- and post-procedural patient statuses, angiographic outcomes, and recurrence during a mean follow-up period of 12 months (range 3–25). The patients with AChA (n=18) underwent stent-assisted coiling (n=14), coiling (n=2), or stenting only (n=2). The technical strategy for aneurysm embolization included a low-lying approach into the aneurysm, stent-strut abutting (protecting) coil framing to block inflow and avoid compromising AChA flow, and direct intra-aneurysmal angiography via a microcatheter. Angiographic results showed complete occlusion (n=11), neck remnant (n=5), and sac remnant (n=2). During the follow-up, there were 2 recurrences, and 1 of them required a re-procedure. All patients showed no change in clinical status (modified Rankin scale score=0) and did not experience any ischemic or hemorrhagic events during the follow-up period. AChA aneurysms can be managed through embolization using various techniques. Such technical concepts may enhance the safety and improve the outcomes of AChA aneurysm embolization.
Anterior choroidal artery (AChA) aneurysms are relatively rare, accounting for 2–5% of all intracranial aneurysms [1]. The AChA is a small artery that supplies critical territory, including the posterior limb of the internal capsule. Impairment of this artery may produce debilitating postoperative AchoA syndrome, including contralateral hemiparesis [2]. The mortality rate and the treatment-related permanent morbidity rate of surgical clipping of AchoA aneurysms ranged from 6% to 33% and 10% to 28.6%, respectively [3]. The incidence of postoperative infarctions after microsurgical clipping of AChA aneurysms was reported to be between 4.8% and 7% [3].
Coiling of AChA aneurysms has comparable clinical outcomes and rebleeding prevention to clipping, with a significantly lower incidence of AChA infarction than clipping [1]. However, a recent systemic review showed a higher mortality and lower morbidity rate for coiling compared to clipping [4]. These studies were all retrospective and did not compare the initial patient status and morphological characteristics of the aneurysms. Some biases could have been introduced in those studies because the treatment decisions were based on the surgical or neurointerventional capabilities of each institute. These variable situations made the development of the embolization technique challenging, hindering the establishment of a standardized protocol and the proposal of a randomized controlled study to generalize treatment methods.
There have been few technical notes on AChA aneurysm embolization [5]. While the outcomes of aneurysm embolization continue to improve, there is a need to emphasize technical development for specific aneurysms such as AChA aneurysms. Herein, we present a technical concept based on our recent case series in which we were able to enhance both safety and effectiveness.
Eighteen consecutive patients (6 males, 12 females; aged 38–68) with unruptured AChA aneurysms, who underwent endovascular treatment in the past 2 years, were included from a prospectively collected hospital neurovascular database. Inclusion criteria included patients with AChA aneurysms who underwent treatment with stent-assisted coiling, coiling alone, or stenting. Patients with multiple aneurysms were also included if an AChA aneurysm was treated.
Patients diagnosed with an AChA aneurysm but not treated were excluded. One patient who underwent a cerebral angiogram for a small AChA aneurysm at our hospital was scheduled for follow-up, while the remaining 18 patients in this study were evaluated at external hospitals. The cohort in this study has not been included in any other research.
This study cohort did not include any ruptured AChA aneurysms because there were no cases of rupture during the study period. Aneurysm size criteria could not be applied in this study because some patients who initially sought treatment from a neurosurgeon at another hospital were required to undergo clipping, despite having small aneurysms. Subsequently, these patients requested alternative treatments, such as coiling and/or stenting, even after a period of observation.
The embolization procedure was the same as previously described [5-10]. Flow diverters can be utilized as an alternative treatment option. However, they cannot be used in Korea because the Health Insurance Review and Assessment Service has not approved their use for aneurysms smaller than 10 mm in size and those originating in the internal carotid artery.
Outcome evaluation was measured by post-embolization angiographic results and final neurological status after the procedure. Angiographic results were assessed using a 3-grade scale on the angiogram after embolization: complete obliteration, presence of a residual neck, or presence of a residual sac. The final neurological status of patients was assessed using the modified Rankin scale (mRS) score, ranging from 0 (normal) to 6 (death), at the time of discharge after the procedure and at the end of the clinical follow-up. Recurrence of the treated aneurysm was assessed using magnetic resonance angiography (MRA). Any progressive occlusion of remnant sac or neck was not evaluated because MRA was the method used for evaluation and it seemed unclear to judge whether sac and/or neck remnant were further occluded or not. We presented technical details, pre-and post-procedural clinical status, angiographic results, and the presence of recurrence during a mean 12-month (3–25) follow-up period.
The combination of embolization techniques included a low-lying approach into the aneurysm, strut-abutting coiling compaction to avoid compromising the flow into the AChA, and intra-aneurysmal angiography (IAA) (Fig. 1A-C). IAA was obtained when the angiogram did not clearly demonstrate a relationship between the aneurysm and the AChA (Fig. 2). Once the microcatheter entered the aneurysm, IAA was performed via the microcatheter to identify the precise origin of the AChA. Aneurysm type was categorized as axillary type, in which the AChA arose proximal to the aneurysm, or shoulder type, in which the AChA arose distal to the aneurysm. Medial to lateral deviation of the AChA origin from the aneurysm was not evaluated due to the limited resolution of the angiogram, especially when the AChA ran parallel to the aneurysm wall.
Achieving a low-lying approach of the microcatheter into the aneurysm involves creating a curve of more than 90 degrees to allow the tip of the microcatheter to enter the aneurysm from below at the origin of the AChA. The advantage of a low-positioned approach is to push coils into the aneurysm sac and prevent the protrusion of coils into the entrance of the AChA [5]. If there is any coil protrusion in front of the AChA with possible flow compromise, perform a pull and push action of the microcatheter and coils repeatedly until the coil frame is properly repositioned into the aneurysm (Fig. 3).
Regarding the repeated pull and push action, it was necessary to create a frame to maintain a small space in front of the AChA origin to ensure that blood flow into the AChA was not compromised (Fig. 1C). Introduction of the coil by smooth pulling and pushing repeatedly is essential to achieve a proper coil position, forming a stable coil frame that creates a triangular space between the stent strut and the adjacent aneurysm wall (typically the anterior aneurysm wall) in front of the AChA origin (Figs. 1, 3). The stent used in this study was the Neuroform Atlas stent (Stryker Neurovascular).
Although we categorized 3 techniques, they were conceptually intertwined and not easily separable. Since this is a procedural concept, it would be challenging to establish any measurable criteria in an objective manner.
Patients with an AChA (n=18) underwent stent-assisted coiling (n=14), coiling (n=2), or stenting only (n=2) (Table 1). All aneurysms were axillary type with a size range of 1.5–5.8 mm (mean 3.3 mm). Six patients had multiple aneurysms. Angiographic results showed complete occlusion (n=11), neck remnant (n=5), and sac remnant (n=2). During a mean 12-month follow-up period (range 3–25 months), there were 2 recurrences.
Among the 2 recurrent patients, 1 exhibited a small remnant sac after coiling at the elongated neck, from which the AChA originated. The remaining sac enlarged but remained stable at the 17-month follow-up MRA, and it was then decided not to retreat it. The other patient underwent stent-assisted coiling for 2 adjacent tandem aneurysms just above the AChA at the same time (Fig. 4). One-year follow-up MRA revealed a recurrent sac had developed within the lower aneurysm, and re-embolization was performed.
Stenting was done in 2 aneurysms to induce a flow-diverting effect [11-13]. One of the aneurysms was a broad-necked aneurysm at the origin of AChA, which was treated with stenting using the same stent that was employed for the stent-assisted coiling of the proximal dorsal wall aneurysm. The other aneurysm showed that the AChA originated from an aneurysmal sac on IAA so stenting of the aneurysm was performed without coil insertion (Fig. 2). The aneurysm remained stable in size and shape during the 3-month follow-up period. There was no significant change in the aneurysms after stenting during the follow-up period.
The angiogram obtained at the end of the procedure demonstrated the patency of the AChA. All patients showed no change in clinical status (mRS=0) after the procedure, and there were no ischemic or hemorrhagic events during the follow-up period.
Treatment-related complication rates for AChA aneurysms varied from 4.5% to 50% [14]. One of the main reasons for such variable outcomes depended on the vague and complex vascular anatomy associated with the AChA. Cerebral angiography may not depict the relationship between an aneurysm and the AChA within the aneurysm sac [15-17].
When the AChA cannot be separated from the aneurysm itself, even 3-dimensional rotational angiography cannot completely demonstrate the relationship of the AChA with the aneurysm. So, the risk of compromised blood flow tended to increase with inadvertent coil protrusion into the origin of the AChA during embolization [14]. Therefore, the procedure would be risky and ineffective unless there is a technical strategy to avoid such risks. Such technical development seems crucial in determining the treatment approach and expanding treatment options for AChA aneurysms.
Our study revealed that embolization of AChA aneurysms can be safe and effective by applying a technical concept to create space in front of the AChA entrance. The packing of coils in the aneurysm sac was combined with several techniques involving a low-lying microcatheter approach into the aneurysm. This resulted in the formation of a rather rigid coil frame in front of the AChA origin and/or for IAA.
The critical point of the technical concept in this study is to establish an intentional coil framework between the anterior wall of the aneurysm and the stent strut (Fig. 1C). To achieve maximum effect, the microcatheter needed to enter the aneurysm through the stent strut after stenting. This way, the stent strut would physically push down into the aneurysmal sac to secure the triangular space in front of the AChA. A coil frame with a triangular space can resist the inflow zone of an aneurysm and improve the packing of the aneurysm sac.
The concept described in this study has evolved from the author’s experience with 80 out of 956 cases of AChA aneurysms [6]. Therefore, it may not be generalized and should be implemented by experienced operators who understand the physical characteristics of coils, stents, and microcatheters, as well as vascular anatomical details. In addition, it would be necessary to go through the decision-making process to determine whether an AChA aneurysm needs to be treated or not. If treatment is required, which option would be better: surgical clipping or endovascular management? Some patients were provided consultation for coiling treatment when they were recommended for clipping to explore non-surgical treatment options.
Surgical clipping still remains an option for treating AChA aneurysms. The incidence of postoperative infarctions after microsurgical clipping of AChA aneurysms was reported to be between 4.8% and 7% and decreased to 2.7% by using intraoperative neuromonitoring and intraoperative indocyanine green videoangiography (ICG-VA) [3,18,19]. However, there were 4 false-negative cases of postoperative infarctions related to the AChA territory that exhibited patent AChA flow on intraoperative ICG-VA due to a changed positional relationship between the clip and the adjacent vessels. Furthermore, atherosclerosis near the aneurysm may lead to postoperative ischemic complications following clipping.
One limitation of our study is that the technical advancements were not adopted by all operators. A randomized controlled study is recommended to compare efficacy, even though the incidence of this aneurysm is not common. The technical concepts recommended by this study may not be accepted by other operators who have experienced different outcomes. If the AChA aneurysm exhibits an anatomical disposition different from the axillary type, as demonstrated in our study, modified methods must be employed beyond those described herein. This study should be subjectively understood, focusing on a descriptive approach to technical concepts. However, this concept can also be applied to other complex aneurysms.
This study cohort did not include any ruptured AChA aneurysms because there were no cases of rupture during the study period. It depends on each patient’s condition whether the same technique can also be used in the case of a ruptured aneurysm, as surgical clipping can be another option in this emergency situation.
For some patients who initially sought treatment from a neurosurgeon at another hospital, clipping was required to eliminate any potential risk of rupture, even in the presence of small aneurysms. Subsequently, these patients requested alternative treatments, such as coiling and/or stenting. In such cases, observation and follow-up studies may also be recommended to assess the feasibility of monitoring the aneurysms and to alleviate anxiety caused by the low risk of rupture, even after a period of observation.
Flow diverters can be used as an alternative treatment option. However, their use is not permitted in Korea because the Health Insurance Review and Assessment Service has not approved them for aneurysms smaller than 10 mm or those originating in the internal carotid artery.
In conclusion, AChA aneurysms can be managed through embolization with the assistance of various techniques. The technical concept of such tactical development may maximize device characteristics and clarify the anatomical relationship between the AChA and the aneurysm sac. And thus, such techniques may improve outcomes and safety in treating AChA aneurysms. Further follow-up and a larger study may strengthen our study results.
Notes
REFERENCES
1. Kim BM, Kim DI, Shin YS, Chung EC, Kim DJ, Suh SH, et al. Clinical outcome and ischemic complication after treatment of anterior choroidal artery aneurysm: comparison between surgical clipping and endovascular coiling. AJNR Am J Neuroradiol. 2008; 29:286–290.
2. Hendricks BK, Spetzler RF. Surgical challenges associated with anterior choroidal artery aneurysm clipping: 2-dimensional operative video. Oper Neurosurg (Hagerstown). 2020; 19:E289.
3. Eun C, Lee SJ, Park JC, Ahn JS, Kwun BD, Park W. Efficacy of intraoperative neuromonitoring (IONM) and intraoperative indocyanine green videoangiography (ICG-VA) during unruptured anterior choroidal artery aneurysm clipping surgery. J Cerebrovasc Endovasc Neurosurg. 2023; 25:150–159.
4. Al Fauzi A, Rahmatullah MI, Suroto NS, Utomo B, Fahmi A, Bajamal AH, et al. Comparison of outcomes between clipping and endovascular coiling in anterior choroidal artery aneurysm: a systematic review. Neurosurg Rev. 2023; 46:276.
5. Sheen JJ, Suh DC. Low-angled microcatheter approach for coil embolization of the anterior choroidal artery aneurysm. Neuroradiology. 2017; 59:1053–1056.
6. Suh DC, Choi YH, Park SI, Yun S, Jeong SY, Jeong S, et al. Outpatient day-care management of unruptured intracranial aneurysm: a retrospective cohort study. Korean J Radiol. 2022; 23:828–834.
7. Song Y, Kwon B, Al-Abdulwahhab AH, Kurniawan RG, Suh DC. Microcatheter stabilization technique using partially inflated balloon for coil embolization of paraclinoid aneurysms. Neurointervention. 2021; 16:132–140.
8. Kurniawan RG, Song Y, Kwon B, Ahn Y, Suh DC. Tailored antiplatelet agent medication in clopidogrel hyporesponsive patients before stent-assisted coiling: single-center experience. Neuroradiology. 2020; 62:1709–1715.
9. Lee D, Song Y, Shin JH, Suh DC. Low-dose prasugrel in patients with resistance to clopidogrel for the treatment of cerebral aneurysms: follow-up of over 6 months. Neurointervention. 2019; 14:68–70.
10. Jia ZY, Song YS, Sheen JJ, Kim JG, Lee CW, Suh DC. Loop microcatheter technique for coil embolization of paraclinoid aneurysms. Acta Neurochir (Wien). 2018; 160:1755–1760.
11. Song Y, Choe J, Liu H, Park KJ, Yu H, Lim OK, et al. Virtual stenting of intracranial aneurysms: application of hemodynamic modification analysis. Acta Radiol. 2016; 57:992–997.
12. Park W, Song Y, Park KJ, Koo HW, Yang K, Suh DC. Hemodynamic characteristics regarding recanalization of completely coiled aneurysms: computational fluid dynamic analysis using virtual models comparison. Neurointervention. 2016; 11:30–36.
13. Liu H, Choe J, Jung SC, Song Y, Yang KH, Park KJ, et al. Does a low-wall coverage stent have a flow diverting effect in small aneurysms? Neurointervention. 2015; 10:89–93.
14. André A, Boch AL, Di Maria F, Nouet A, Sourour N, Clémenceau S, et al. Complication risk factors in anterior choroidal artery aneurysm treatment. Clin Neuroradiol. 2018; 28:345–356.
15. Wolman DN, Moraff AM, Heit JJ. Anatomy of the intracranial arteries: the internal carotid artery. Neuroimaging Clin N Am. 2022; 32:603–615.
16. Duan Y, Qin X, An Q, Liu Y, Li J, Chen G. A new classification of anterior choroidal artery aneurysms and its clinical application. Front Aging Neurosci. 2021; 13:596829.
17. Park J, Kim JS. Infundibular widening of angiographically invisible duplicate anterior choroidal artery mimicking typical anterior choroidal artery aneurysm. J Korean Neurosurg Soc. 2023; 66:105–110.
18. Irie T, Yoshitani K, Ohnishi Y, Shinzawa M, Miura N, Kusaka Y, et al. The efficacy of motor-evoked potentials on cerebral aneurysm surgery and new-onset postoperative motor deficits. J Neurosurg Anesthesiol. 2010; 22:247–251.
19. Szelényi A, Langer D, Kothbauer K, De Camargo AB, Flamm ES, Deletis V. Monitoring of muscle motor evoked potentials during cerebral aneurysm surgery: intraoperative changes and postoperative outcome. J Neurosurg. 2006; 105:675–681.
Fig. 1.
Schematic technical concept diagrams of stent-assisted coiling for an anterior choroidal artery (AChA) aneurysm. (A) After a stent is deployed, a microcatheter enters the aneurysm using a low-angle approach technique just above the AChA origin through the stent. (B) Coils were packed by creating a vertical barrier between the stent and the adjacent aneurysm wall to ensure that the flow into the AChA was not compromised. (C) Note the triangular-shaped inflow zone (asterisks in a magnified box figure) designed to protect the inflow zone to the aneurysm and preserve the flow into the AChA.
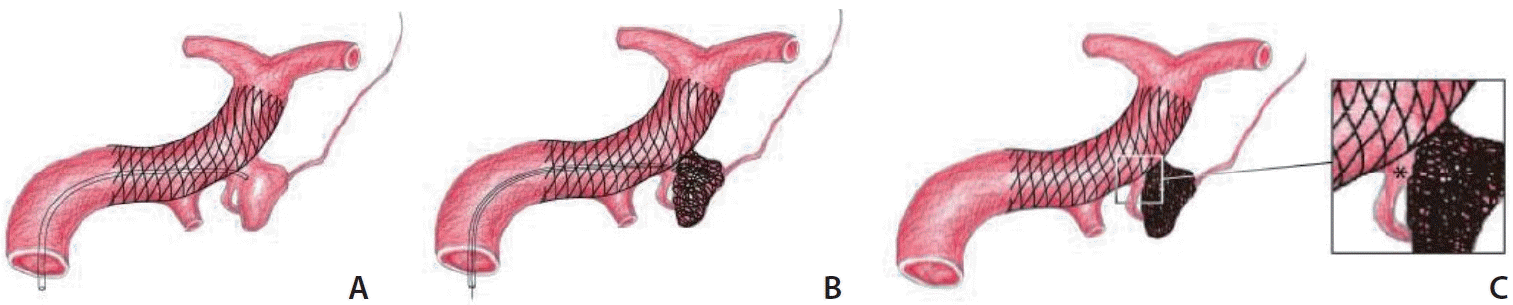
Fig. 2.
To show the anterior choroidal artery (AChA) origin within the aneurysm, (A) intra-aneurysmal injection (B) was performed to demonstrate the exact origin of the AChA, which arose from the aneurysm sac (arrow).
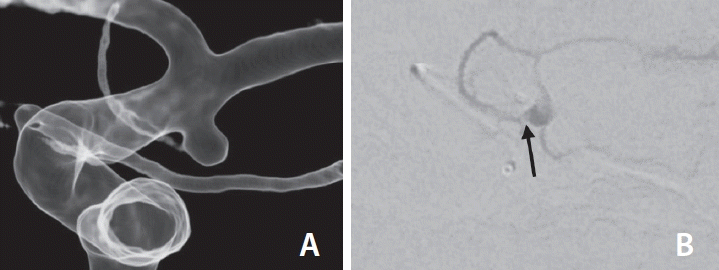
Fig. 3.
An anterior choroidal artery (AChA) aneurysm with a distinctive bleb (Case 12). (A) Note the AChA arises in the sac. (B) A coil was introduced to establish a flow barrier between the stent strut and the aneurysm wall (long arrow) using a low-lying microcatheter (short arrow) positioned in front of the AChA origin. (C) The final angiogram shows a remnant sac diverting the flow into the AChA (arrow). (D) A 15-month follow-up magnetic resonance angiography shows no recurrence of the aneurysm.
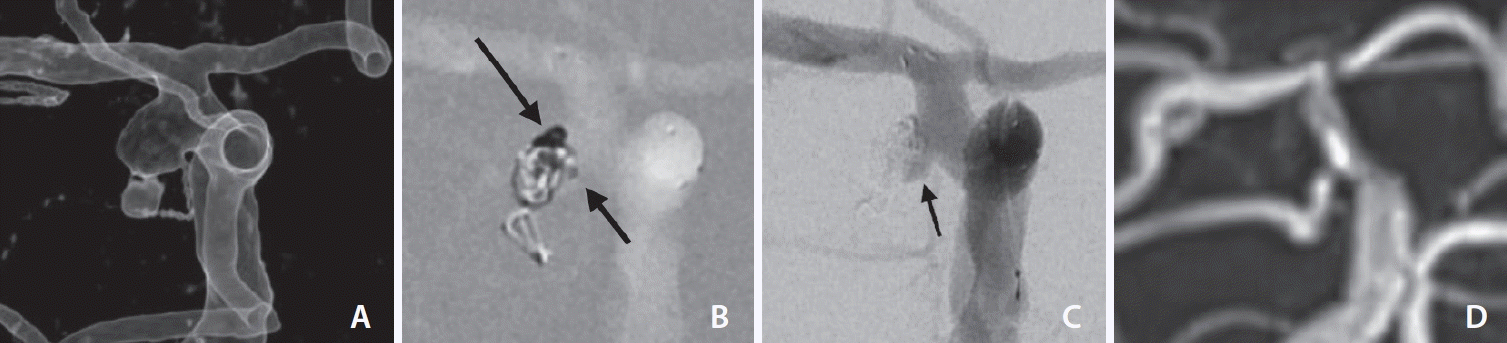
Fig. 4.
A male in his forties (Case 11) with tandem aneurysms (A) at the origin of the right anterior choroidal artery. One year after stent-assisted coiling (B), there was a recurrence (C). Re-embolization was performed by adding more coils within the recurrent sac (not shown).
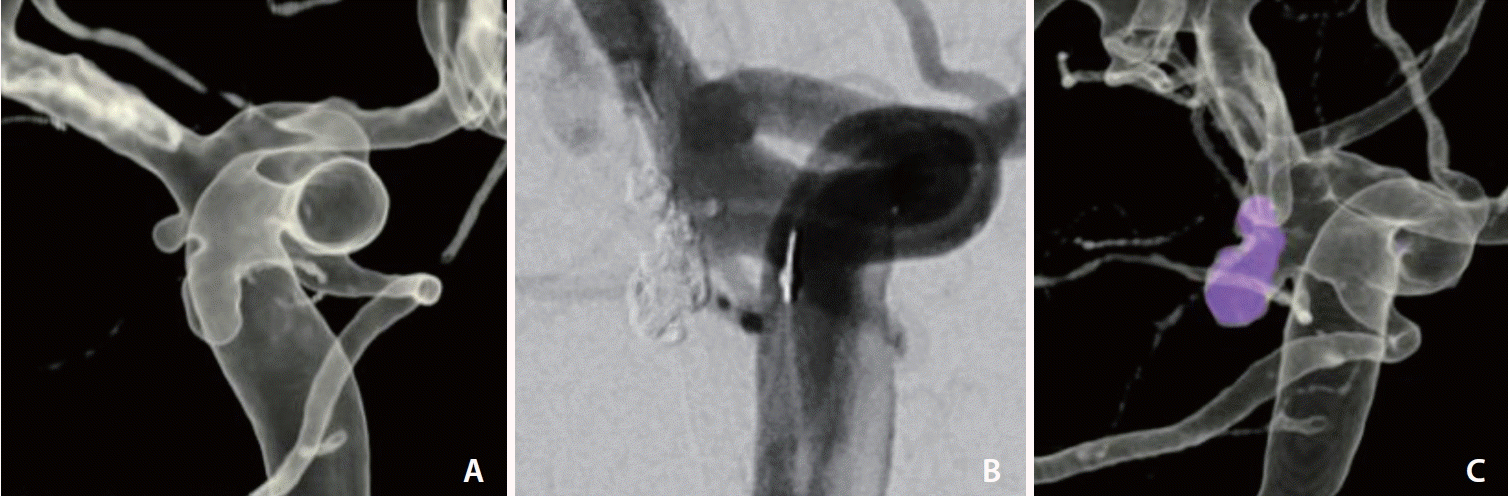
Table 1.
Case summary of 18 aneurysms




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download