Abstract
Downstaging of hepatocellular carcinoma (HCC) is typically defined as the reduction in size or number of viable tumors through locoregional therapy (LRT), aiming to meet the established criteria for liver transplantation (LT). According to the Barcelona Clinic Liver Cancer (BCLC) staging system, a subgroup of patients with BCLC-B may benefit most from downstaging therapies. The United Network Organ Sharing downstaging protocol identifies potential candidates for downstaging by setting out ‘inclusion criteria’ and defining ‘successful downstaging.’ Additionally, the protocol considers factors related to tumor biology, such as an alphafetoprotein level <500 ng/mL after LRT. Reports indicate that successful downstaging rates following LRT are about 50%, with post- LT recurrence rates comparable to those of patients within the Milan criteria. A comprehensive multicenter US study on 10-year outcomes post-LT after downstaging showed 10-year post-LT survival and recurrence rates of 52.1% and 20.6%, respectively, for patients whose disease was downstaged; this compares to 61.5% and 13.3% for those consistently within the Milan criteria. Recently, the development of effective systemic treatments for HCC, such as immuno-oncologic agents, has provided additional opportunities for downstaging. Numerous clinical trials are exploring a multidisciplinary approach (MDA) combining LRT and systemic therapy. Although concrete evidence of the superiority of MDA for HCC downstaging is lacking, some retrospective studies and phase I and II trials have shown promising results regarding the efficacy and safety of MDA for this purpose. In this review, we will also discuss the future of MDA protocols in downstaging for improved clinical outcomes.
Go to : 
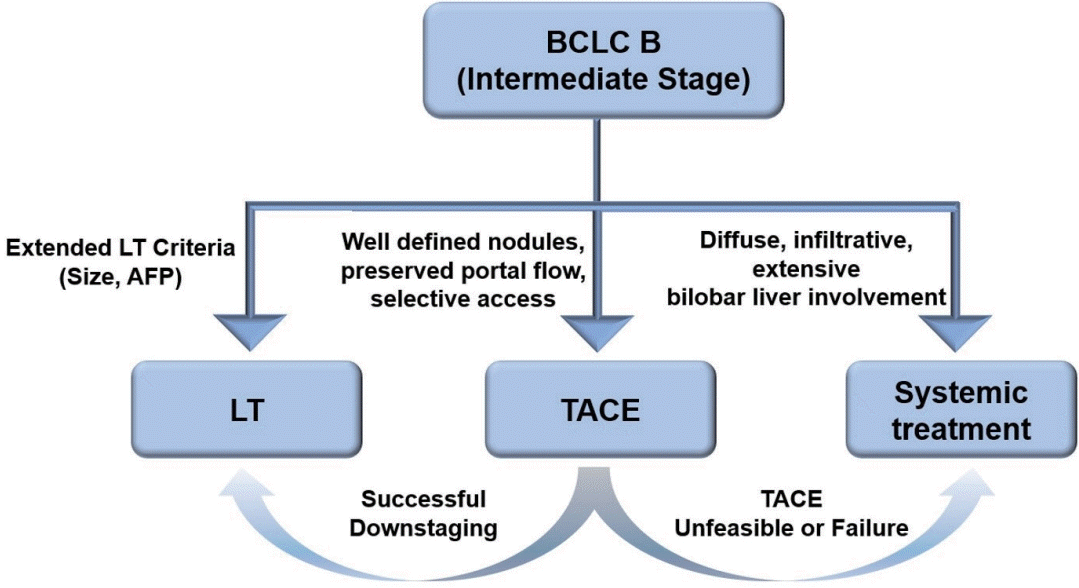
Hepatocellular carcinoma (HCC) is the most common primary malignant tumor of the liver and is the second leading cause of cancer-related deaths worldwide.1,2 Among various staging systems for HCC, the Barcelona Clinic Liver Cancer (BCLC) system is the most widely recognized. The BCLC guidelines recommend several initial treatments, such as ablation, hepatic resection, liver transplantation (LT), trans-arterial chemoembolization (TACE), trans-arterial radioembolization (TARE), and systemic therapy, tailored to each stage of the disease. In 2022, the updated BCLC guidelines further subdivided BCLC-B (intermediate stage) patients into three groups and recommended subsequent LT after successful downstaging in a selected subgroup (Fig. 1).3 Downstaging of HCC is generally defined as a reduction in the size or number of viable tumors using locoregional therapy (LRT) to meet the established criteria for LT. Recent studies have reported that successful downstaging rates after LRT are nearly 50%, and post-LT recurrence rates are comparable to those of patients within the Milan criteria (MC) in highly selected patients.4
Recent developments in systemic treatments for HCC, particularly immuno-oncologic (IO) agents, have been promising,5,6 offering additional opportunities for downstaging. Furthermore, many clinical trials have focused on a multidisciplinary approach (MDA), combining LRT and systemic therapy. Results from retrospective studies and phase I and II trials on the efficacy and safety of MDA for downstaging have been published. In this review, we will also discuss the future protocols of MDA for downstaging to achieve better clinical outcomes.
Go to : 
According to the updated BCLC guidelines, patients classified as BCLC-B (intermediate stage) are subdivided into three groups. The first subgroup includes patients who meet the extended LT criteria (beyond the MC) and for whom LT is recommended as an initial treatment.3 Among the various extended LT criteria, the University of California San Francisco (UCSF) criteria are relatively well-validated. The UCSF criteria define eligibility as either one tumor ≤6.5 cm or ≤3 tumors, with the largest tumor ≤4.5 cm and a total tumor diameter ≤8 cm. A recent multicentric study on the efficacy of living donor LT showed that overall survival (OS) rates were not significantly different between the MC and UCSF groups (1-, 5-, and 10-year survival rates, 90.9%, 78.5%, and 64.1% vs. 88.6%, 73.5%, and 69.4%, respectively; P=0.85).7 The second subgroup consists of patients with diffuse, infiltrative, extensive bi-lobar tumors, for whom systemic treatment is recommended. The third subgroup includes patients with well-defined nodules, preserved portal flow, and selective vascular access; for these patients, TACE is recommended. Importantly, LT is advised for third subgroup patients if they can be successfully downstaged. Therefore, according to the updated BCLC guidelines, the third subgroup is considered the best candidate for downstaging.
The United Network Organ Sharing downstaging (UNOS-DS) protocol, developed based on the UCSF LT criteria (Table 1),8 extends beyond the UCSF criteria in terms of tumor size and number (up to 8 cm in a single tumor, up to 5 cm in 2-3 tumors, and up to 3 cm in 4-5 tumors). Successful downstaging is defined as follows: 1) achievement of tumor control within the MC after treatment, 2) durable tumor control within the MC for more than 6 months, and 3) alpha-fetoprotein (AFP) levels <500 ng/mL if pretreatment AFP >1,000 ng/mL.
UNOS downstaging criteria with definitions of posttreatment success and failure
Several studies demonstrate the clinical efficacy of LT under the UNOS-DS protocol. Mehta et al.9 analyzed 3,698 patients who underwent LT (3,276 within MC, 422 under UNOS-DS) and found no significant difference in 3-year post-LT survival rates between the MC and UNOS-DS groups (83.2% vs. 79.1%; P=0.17). They also noted that post-LT mortality risk was higher in regions with shorter wait times (less than 9 months from successful downstaging to LT) compared to longer wait regions (hazard ratio [HR], 3.1; P=0.005) and in patients with AFP levels ≥100 ng/mL at LT (HR, 2.4; P=0.009). Tabrizian et al.10 analyzed 2,463 patients who underwent LT (2,122 within MC, 341 under UNOS-DS) and reported that 10-year post-LT OS rates were 61.5% for MC and 52.1% for UNOS-DS, respectively (P<0.001), with 10-year post-LT recurrence rates of 13.1% for MC and 20.6% for UNOS-DS, respectively (P<0.001). Based on these findings, they concluded that the UNOS-DS protocol could aid in LT prioritization decisions as the 10-year post-LT outcomes were relatively favorable. They also identified factors associated with poor recurrence-free survival (RFS) in the downstaged group, such as a neutrophil-to-lymphocyte ratio (NLR) >5 at LT (odds ratio [OR], 2.04; 95% confidence interval [CI], 1.34-3.09; P<0.001) and largest viable tumor on explant pathologic findings >5 cm (OR, 2.18; 95% CI, 1.34-3.56; P=0.002), regardless of pretreatment tumor size, number, or tumor marker. Clinical factors associated with downstaging failure included more than three tumors at diagnosis (OR, 2.34; 95% CI, 1.22-4.50; P=0.01), tumor size >7 cm at diagnosis (OR, 2.62; 95% CI, 1.20-5.75; P=0.02), and an AFP response of at least 20 ng/mL with less than 50% improvement (OR, 1.99; 95% CI, 1.14-3.46; P=0.02). Mehta et al.11 further supported the importance of tumor markers in predicting downstaging failure, noting that a pretreatment AFP level >1,000 ng/mL was a better predictor of downstaging failure (HR, 3.3; P<0.001) compared to poor liver function (HR, 1.6; P<0.001). Therefore, more powerful treatment modalities are needed for this high-risk group to achieve successful downstaging, as the improvement in tumor biology, such as NLR and real pathologic viable tumor size at LT, are crucial for predicting RFS. Mazzaferro et al.12 conducted a randomized, controlled phase 2b/3 trial analyzing 45 patients (23 who underwent LT, 22 who continued follow-up until progression after successful downstaging) with similar conditions under UNOS-DS. They reported that the 5-year OS rate was 77.5% in the LT group versus 31.2% in the control group (HR, 0.32; 95% CI, 0.11-0.92; P=0.035) and concluded that subsequent LT should definitely be considered if HCC patients beyond MC are successfully down-staged. Tan et al.13 conducted a meta-analysis of 25 studies (3,997 patients) on downstaging and reported that the success rate of downstaging and post-LT recurrence rate between two groups (UNOS-DS vs. beyond UNOS-DS) were 83.21% vs. 28.60% (P<0.001) and 9.06% vs. 20.42% (P<0.001), concluding that UNOS-DS criteria are the most reliable, showing the best downstaging success and postLT outcomes.
According to the updated BCLC guideline, TACE and TARE are recommended as treatment modalities for downstaging. Mehta et al.14 analyzed 194 HCC patients who met UNOS-DS criteria and received trans-arterial treatment (TACE, 132 patients; TARE, 62 patients). They reported that 174 patients (83%) were successfully down-staged, with no significant difference in time to or probability of successful downstaging between the TACE and TARE groups.
Go to : 
All comers (AC) are patients whose tumor burden initially exceeds UNOS-DS criteria. Therefore, the inclusion criteria of the AC-DS protocol are defined as patients with HCC beyond UNOS-DS criteria but still classified within BCLC-B. The definition of successful downstaging in the AC-DS protocol is nearly identical to that of the UNOS-DS protocol. However, studies on the clinical outcomes of the AC-DS protocol are less common than those on the UNOS-DS protocol.
In the study conducted by Metha et al.9 the 3-year post-LT OS rate was significantly different between the MC and ACDS (83.2% vs. 71.4%; P=0.04). Natarajan et al.15 analyzed 311 patients who underwent LT (229 in UNOS-DS, 82 in ACDS) and reported a 3-year OS rate from LRT of 69% in the UNOS-DS group and 58% in the AC-DS group (P=0.05). The 3-year post-LT OS rate was also not significantly different between the two groups (91% vs. 81%; P=0.67). They noted that the 3-year OS rate from LRT in AC-DS patients without high-risk factors (AFP >500 ng/mL or Child-Pugh class B/C cirrhosis) was 67.7% compared to 29.9% in AC-DS patients with one or two high-risk factors (P=0.003). They suggested that LT could be considered for highly selected AC-DS patients with favorable tumor biology if successful downstaging is achieved.
Portal vein tumor thrombus (PVTT) has traditionally been considered a contraindication for LT. However, recent studies have reported that both deceased donor LT (DDLT) and living donor LT (LDLT) showed acceptable clinical benefits. Yu et al.16 analyzed 961 patients who underwent LT (489 within MC, 296 beyond MC but without PVTT, 83 with type 1 PVTT, and 93 with type 2 PVTT) and reported that the 5-year OS rate for patients with type 1 PVTT was not significantly different compared to those within MC (78.3% vs. 79.1%; P=0.062) and was superior compared to patients beyond MC but without PVTT (78.3% vs. 50.0%; P=0.012). They mentioned that OS and RFS rates for PVTT patients with AFP ≤100 ng/mL were significantly superior compared to those of patients beyond MC or PVTT patients with AFP >100 ng/mL (P<0.01). Notably, OS rates were not significantly different between PVTT patients with AFP ≤100 ng/mL and those within MC (P=0.065). Soin et al.17 analyzed 405 patients who underwent LT (382 without PVTT, 23 with down-staged PVTT) based on prospectively collected data. They reported that 5-year OS and RFS rates for patients with down-staged PVTT were not significantly different compared to patients without PVTT (57% vs. 65%, P=0.06; 66% vs. 51%, P=0.33). They also noted that the 1-year OS rate for patients with down-staged PVTT was better than in PVTT patients receiving sorafenib monotherapy (82% vs. 0%; P<0.001). Serenari et al.18 conducted a pilot prospective study analyzing the clinical outcomes of five HCC patients with PVTT who underwent DDLT after successful downstaging by TARE. They reported that the 5-year OS rate of the DDLT group was significantly better compared to the observation group (60% vs. 0%; P=0.03). Based on these results, we can propose that LT should be carefully considered for highly selected patients with PVTT after successful downstaging.
It is well established that MDA enhances survival in patients with HCC. El Dahan et al.19 conducted a meta-analysis of 12 studies encompassing 15,365 HCC patients and found that MDA was associated with improved overall survival (HR, 0.63; 95% CI, 0.45-0.88). Sinn et al.20 analyzed 6,619 HCC patients (738 receiving MDA care, 5,881 not) and reported significantly higher 5-year survival rates for patients under MDA care compared to those without (71.2% vs. 49.4%; P<0.001). They noted that MDA care was particularly beneficial for patients with BCLC-B or BCLC-C and AFP levels over 200 ng/mL, who could be candidates for downstaging.
As previously mentioned, successful downstaging, including the durable stabilization of tumor biology, can lead to favorable survival and prognosis after LT, even if the initial tumor burden exceeds extended LT criteria (such as UNOS-DS, AC-DS, PVTT). Thus, more robust treatment modalities, such as combination therapies based on MDA, increase the chances of successful downstaging followed by subsequent LT.
Recent advancements in the systemic treatment of HCC, particularly trials on immune checkpoint inhibitor (ICI)-based treatments, have shown promising results. The IMbrave150 trial reported that patients treated with a combination of atezolizumab and bevacizumab had better overall survival (19.2 vs. 13.4 months; HR, 0.66) compared to those treated with sorafenib.5 Similarly, the HIMALAYA trial demonstrated improved overall survival with durvalumab-tremelimumab combination therapy (16.4 vs. 13.8 months; HR, 0.78) compared to sorafenib.6
Combining IO therapy with LRT such as TACE, TARE, and radiation therapy (RT) could be a potent strategy for enhancing the success rate of downstaging. This approach is promising because LRT may induce the release of tumor-associated antigens and activate antigen-presenting cells, potentially converting the tumor microenvironment to become more immunesensitive.21
Kudo et al.22 analyzed 110 patients with unresectable or TACE-unsuitable BCLC-B HCC who were treated with atezolizumab plus bevacizumab, finding that 28 patients (25%) achieved a complete response (CR) and became candidates for curative-intent treatments post-therapy. Ding et al.23 performed a meta-analysis (1,174 patients from three databases and 19 studies) and found that the group treated with a combination of cellular immunotherapy and LRT (predominantly TACE) had significantly improved 1-year OS (OR, 3.52; P<0.0001) compared to those treated with LRT alone.
Yeo et al.24 reported on the clinical outcomes of 1,664 advanced HCC patients, comparing 142 treated with immunotherapy plus TARE and 1,522 treated with immunotherapy alone. They found that the OS of the combination group was significantly higher than that of the immunotherapy-alone group (19.8 vs. 9.5 months; P<0.001). Tai et al.25 conducted a phase 2 trial on TARE followed by nivolumab in 36 advanced HCC patients and reported an objective response rate (ORR) of 41.7%. Lee et al.26 carried out a phase 1/2a trial on TARE followed by durvalumab in 24 BCLC-B and BCLC-C patients and found an ORR of 83.3% (CR, 29.2%; partial response, 54.2%) and an 18-month OS rate of 58.3%.
Hwang et al.27 analyzed 98 advanced HCC patients with high-risk factors (bile duct invasion, main portal vein invasion, large tumor) treated with atezolizumab plus bevacizumab, with or without RT. They reported that the OS of the RT combination group was significantly higher than that of the non-combination group (12.00 vs. 5.75 months; P<0.001). Chiang et al.28 conducted a phase 2 trial evaluating the clinical outcomes of sequential TACE and stereotactic body RT followed by avelumab in locally advanced HCC with 64% macrovascular invasion. They enrolled 33 patients, 18 patients (55%) of whom were converted to curative treatment, 14 patients (42%) achieved a CR and did not require further therapy, and the median OS was 30.3 months.
The use of IO agents is generally considered a contraindication in LT patients due to the potential for graft rejection. However, some reports suggest that pre-LT use of IO agents can be safe if there is an appropriate cessation period before LT, known as a washout strategy. Wang et al.29 analyzed 16 HCC patients treated with ICIs before LT and reported an acute rejection rate of 56.3%, though all cases of acute rejection were resolved with immunosuppressive treatment. They also noted that the interval between the last ICI administration and LT in the rejection group was significantly shorter than in the non-rejection group (21.0 [15.5-27.5] vs. 60 [34.0-167.0] days; P=0.01). RezaeeZavareh et al.30 performed a meta-analysis of 30 studies involving 91 HCC patients treated with ICIs before LT and reported an allograft rejection rate of 26.4%. However, overall survival was not different between patients with and without allograft rejection. They also mentioned that the probability of allograft rejection was ≤20% if the median washout period was 94 days.
Go to : 
Currently, LT after successful downstaging in BCLC-B HCC patients using the UNOS-DS protocol is considered a feasible approach with favorable long-term clinical outcomes. Looking ahead, it is crucial to consider expanding the downstaging criteria to include patients beyond UNOS-DS (AC-DS, PVTT). Effective treatment strategies that achieve significant tumor response are essential for broadening these criteria, as the clinical outcomes of subsequent LT depend on successful tumor control and stabilization of tumor biology through treatment. Recently, promising systemic treatments for HCC, such as tyrosine kinase inhibitors and IO agents, have been developed, with many clinical trials focusing on MDA that combine LRT and systemic therapy. Although concrete evidence of MDA’s superiority for HCC downstaging is still lacking, promising results from retrospective studies and phase I/II trials demonstrating the efficacy and safety of MDA for downstaging have been reported. We must, therefore, await the outcomes of ongoing prospective trials that are investigating downstaging based on MDA.
Go to : 
Notes
Ethics Statement
This review article is fully based on articles which have already been published and did not involve additional patient participants. Therefore, IRB approval is not necessary.
Funding Statement
No specific grant was received from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contributions
Conception: SYH, HC, WJ, RGK
Manuscript preparation: SYH, HC, WJ, RGK
Critical revision: SYH, HC, WJ, RGK
All authors reviewed the paper and approved the final version.
Go to : 
References
2. Su GL, Altayar O, O’Shea R, Shah R, Estfan B, Wenzell C, et al. AGA clinical practice guideline on systemic therapy for hepatocellular carcinoma. Gastroenterology. 2022; 162:920–934.

3. Reig M, Forner A, Rimola J, Ferrer-Fàbrega J, Burrel M, Garcia-Criado Á, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022; 76:681–693.

4. Matevish L, Patel MS, Vagefi PA. Downstaging techniques for hepatocellular carcinoma in candidates awaiting liver transplantation. Surg Clin North Am. 2024; 104:145–162.
5. Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020; 382:1894–1905.

6. Abou-Alfa GK, Lau G, Kudo M, Chan SL, Kelley RK, Furuse J, et al. Tremelimumab plus durvalumab in unresectable hepatocellular carcinoma. NEJM Evid. 2022; 1:EVIDoa2100070.

7. Ivanics T, Claasen MPAW, Samstein B, Emond JC, Fox AN, Pomfret E, et al. living donor liver transplantation for hepatocellular carcinoma within and outside traditional selection criteria: a multicentric North American experience. Ann Surg. 2024; 279:104–111.
8. Yao FY, Mehta N, Flemming J, Dodge J, Hameed B, Fix O, et al. Downstaging of hepatocellular cancer before liver transplant: long-term outcome compared to tumors within Milan criteria. Hepatology. 2015; 61:1968–1977.

9. Mehta N, Dodge JL, Grab JD, Yao FY. National experience on downstaging of hepatocellular carcinoma before liver transplant: influence of tumor burden, alpha-fetoprotein, and wait time. Hepatology. 2020; 71:943–954.
10. Tabrizian P, Holzner ML, Mehta N, Halazun K, Agopian VG, Yao F, et al. Ten-year outcomes of liver transplant and downstaging for hepatocellular carcinoma. JAMA Surg. 2022; 157:779–788.

11. Mehta N, Guy J, Frenette CT, Dodge JL, Osorio RW, Minteer WB, et al. Excellent outcomes of liver transplantation following down-staging of hepatocellular carcinoma to within Milan criteria: a multicenter study. Clin Gastroenterol Hepatol. 2018; 16:955–964.

12. Mazzaferro V, Citterio D, Bhoori S, Bongini M, Miceli R, De Carlis L, et al. Liver transplantation in hepatocellular carcinoma after tumour downstaging (XXL): a randomised, controlled, phase 2b/3 trial. Lancet Oncol. 2020; 21:947–956.

13. Tan DJH, Lim WH, Yong JN, Ng CH, Muthiah MD, Tan EX, et al. UNOS down-staging criteria for liver transplantation of hepatocellular carcinoma: systematic review and meta-analysis of 25 studies. Clin Gastroenterol Hepatol. 2023; 21:1475–1484.

14. Mehta N, Frenette C, Tabrizian P, Hoteit M, Guy J, Parikh N, et al. Downstaging outcomes for hepatocellular carcinoma: results from the multicenter evaluation of reduction in tumor size before liver transplantation (MERITS-LT) consortium. Gastroenterology. 2021; 161:1502–1512.
15. Natarajan B, Tabrizian P, Hoteit M, Frenette C, Parikh N, Ghaziani T, et al. Downstaging hepatocellular carcinoma before liver transplantation: a multicenter analysis of the “all-comers” protocol in the multicenter evaluation of reduction in tumor size before liver transplantation (MERITS-LT) consortium. Am J Transplant. 2023; 23:1771–1780.

16. Yu J, Zhuang L, Liu P, Liu Z, Ling S, Deng Y, et al. Long-term outcomes of deceased donor liver transplantation in hepatocellular carcinoma patients with portal vein tumor thrombus: a multicenter study. Eur J Surg Oncol. 2022; 48:121–132.

17. Soin AS, Bhangui P, Kataria T, Baijal SS, Piplani T, Gautam D, et al. Experience with LDLT in patients with hepatocellular carcinoma and portal vein tumor thrombosis postdownstaging. Transplantation. 2020; 104:2334–2345.

18. Serenari M, Cappelli A, Cucchetti A, Mosconi C, Strigari L, Monari F, et al. Deceased donor liver transplantation after radioembolization for hepatocellular carcinoma and portal vein tumoral thrombosis: a pilot study. Liver Transpl. 2021; 27:1758–1766.

19. El Dahan KS, Reczek A, Daher D, Rich NE, Yang JD, Hsiehchen D, et al. Multidisciplinary care for patients with HCC: a systematic review and meta-analysis. Hepatol Commun. 2023; 7:e0143.
20. Sinn DH, Choi GS, Park HC, Kim JM, Kim H, Song KD, et al. Multidisciplinary approach is associated with improved survival of hepatocellular carcinoma patients. PLoS One. 2019; 14:e0210730.

21. Kim DH. Combination of interventional oncology local therapies and immunotherapy for the treatment of hepatocellular carcinoma. J Liver Cancer. 2022; 22:93–102.

22. Kudo M, Aoki T, Ueshima K, Tsuchiya K, Morita M, Chishina H, et al. Achievement of complete response and drug-free status by atezolizumab plus bevacizumab combined with or without curative conversion in patients with transarterial chemoembolization-unsuitable, intermediate-stage hepatocellular carcinoma: a multicenter proof-of-concept study. Liver Cancer. 2023; 12:321–338.

23. Ding M, Wang Y, Chi J, Wang T, Tang X, Cui D, et al. Is adjuvant cellular immunotherapy essential after TACE-predominant minimally-invasive treatment for hepatocellular carcinoma? A systematic meta-analysis of studies including 1774 patients. PLoS One. 2016; 11:e0168798.

24. Yeo YH, Liang J, Lauzon M, Luu M, Noureddin M, Ayoub W, et al. Immunotherapy and transarterial radioembolization combination treatment for advanced hepatocellular carcinoma. Am J Gastroenterol. 2023; 118:2201–2211.
25. Tai D, Loke K, Gogna A, Kaya NA, Tan SH, Hennedige T, et al. Radioembolisation with Y90-resin microspheres followed by nivolumab for advanced hepatocellular carcinoma (CA 209-678): a single arm, single centre, phase 2 trial. Lancet Gastroenterol Hepatol. 2021; 6:1025–1035.

26. Lee YB, Nam JY, Cho EJ, Lee JH, Yu SJ, Kim HC, et al. A phase I/IIa trial of yttrium-90 radioembolization in combination with durvalumab for locally advanced unresectable hepatocellular carcinoma. Clin Cancer Res. 2023; 29:3650–3658.

27. Hwang SY, Woo HY, Heo J, Kim HJ, Park YJ, Yi KY, et al. Outcome of atezolizumab plus bevacizumab combination therapy in high-risk patients with advanced hepatocellular carcinoma. Cancers (Basel). 2024; 16:838.

28. Chiang CL, Chiu KWH, Chan KSK, Lee FAS, Li JCB, Wan CWS, et al. Sequential transarterial chemoembolisation and stereotactic body radiotherapy followed by immunotherapy as conversion therapy for patients with locally advanced, unresectable hepatocellular carcinoma (START-FIT): a single-arm, phase 2 trial. Lancet Gastroenterol Hepatol. 2023; 8:169–178.

29. Wang T, Chen Z, Liu Y, Jia Y, Ju W, Chen M, et al. Neoadjuvant programmed cell death 1 inhibitor before liver transplantation for HCC is not associated with increased graft loss. Liver Transpl. 2023; 29:598–606.
30. Rezaee-Zavareh MS, Yeo YH, Wang T, Guo Z, Tabrizian P, Ward SC, et al. Impact of pre-transplant immune checkpoint inhibitor use on post-transplant outcomes in HCC: a systematic review and individual patient data meta-analysis. J Hepatol. 2024; Jul. 10. doi: 10.1016/j.jhep.2024.06.042 [Epub ahead of print].

Go to : 




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download