Abstract
Tuberculosis (TB) of the genitourinary system is a rare form of extrapulmonary TB. Testicular TB is particularly uncommon among kidney transplantation (KT) recipients. Diagnosing testicular TB is challenging due to the nonspecific nature of clinical presentations and ambiguous imaging results. In this report, we describe a case involving a 36-year-old male KT recipient who presented with left scrotal pain. He had undergone a living donor KT 8 years prior and was receiving tacrolimus, mycophenolate mofetil, and prednisolone. Laboratory tests revealed anemia, leukocytosis, and elevated inflammatory markers. Computed tomography showed left scrotal wall thickening and enlargement, suggestive of a left testicular abscess. We discontinued mycophenolate mofetil and administered intravenous antibiotics. Additionally, we performed an incision and drainage of the abscess. However, there was no improvement in his clinical course. Consequently, we performed a radical left orchiectomy. The biopsy revealed extensive chronic granulomatous inflammation with caseous necrosis, consistent with tuberculous orchiepididymitis. A quadruple anti-TB regimen was administered, leading to an improvement in the patient's condition. To the best of our knowledge, this is the first reported case of testicular TB without other organ involvement in KT recipients. Including testicular TB in the differential diagnosis of testicular infections and masses is necessary to avoid unnecessary surgical procedures.
Tuberculosis (TB) ranks among the most prevalent infectious diseases worldwide. The majority of TB cases are found in Asia, sub-Saharan Africa, and Eastern Europe. In contrast, only 3% of global cases occur in the United States [1]. The Republic of Korea holds the highest TB incidence rate among the Organization for Economic Co-operation and Development (OECD) countries, with 48 cases per 100,000 population, and it ranks second in TB mortality, with a rate of 3.8 deaths per 100,000 population [2,3]. Kidney transplant (KT) recipients, who are in an immunosuppressed state, experience a TB incidence rate that is 20–74 times higher than that of the general population [4]. Furthermore, the incidence of TB varies significantly by geographic area, ranging from 0.3% to 14.6% [4].
TB primarily manifests as pulmonary TB in KT recipients, with extrapulmonary TB occurring in about 35% of cases [5,6]. Within the category of extrapulmonary TB, genitourinary TB is relatively uncommon. Testicular TB, in particular, is rare and often associated with renal TB in the general population [7]. The manifestations of testicular TB are nonspecific and frequently asymptomatic, making it challenging to differentiate and diagnose this condition from other diseases [7].
There have been two reported cases of testicular TB accompanied by disseminated TB in KT recipients. However, to the best of our knowledge, there are no studies reporting cases of TB confined solely to the testis. In this report, we present a case of isolated testicular TB in a KT recipient, which mimicked a testicular abscess.
This study was approved by the Institutional Review Board of Keimyung University Dongsan Hospital (IRB No. DSMC 2024-01-043). The requirement for informed consent was waived by the IRB.
Our case involved a 36-year-old male KT recipient who presented with left scrotal pain. He had been diagnosed with end-stage renal disease resulting from hypertensive nephropathy and had undergone hemodialysis for 4 months. After hemodialysis, he received a living donor KT from his older brother 8 years prior to his presentation. An interferon-gamma release assay was performed at the time of the transplantation, yielding a negative result.
He received induction therapy with basiliximab and was prescribed tacrolimus (4.75 mg twice daily), mycophenolate mofetil (MMF; 500 mg twice daily), and prednisolone (5 mg once daily) as maintenance immunosuppressants. Tacrolimus levels were maintained between 3 and 6 ng/mL in the months leading up to the onset of left scrotal pain. For the first 7 years posttransplant, his creatinine levels consistently ranged from mid-to-high 1 mg/dL. However, in the sixth year posttransplant, donor-specific antibodies (DSAs) against DQ2, with a mean fluorescent intensity (MFI) of 17,225, were detected. In the seventh year posttransplant, he developed cytomegalovirus viremia, with 18,774 copies detected, prompting a reduction in the MMF dosage to 250 mg twice daily. By the eighth year posttransplant, the MFI for DSAs against DQ2 had increased to 22,287, and his creatinine levels had escalated to the high 2 mg/dL range, leading to an increase in the MMF dosage back to 500 mg twice daily. We recommended a kidney allograft biopsy to assess potential antibody-mediated rejection; however, it was not performed due to the patient's refusal. Despite the lack of confirmed rejection, there was a persistent suspicion of antibody-mediated rejection. Subsequently, the kidney allograft function stabilized, with creatinine levels remaining in the mid-to-high 2 mg/dL range. There was no history of other infections, such as TB.
He presented at the outpatient clinic with left scrotal pain but no accompanying fever. His height measured 164.7 cm, his weight was 55 kg, and his body mass index was calculated to be 20.28 kg/m2. A physical examination showed testicular swelling, a 5-cm area of hardness, and tenderness. He did not exhibit any cough or sputum production. Additionally, his chest X-ray appeared clear.
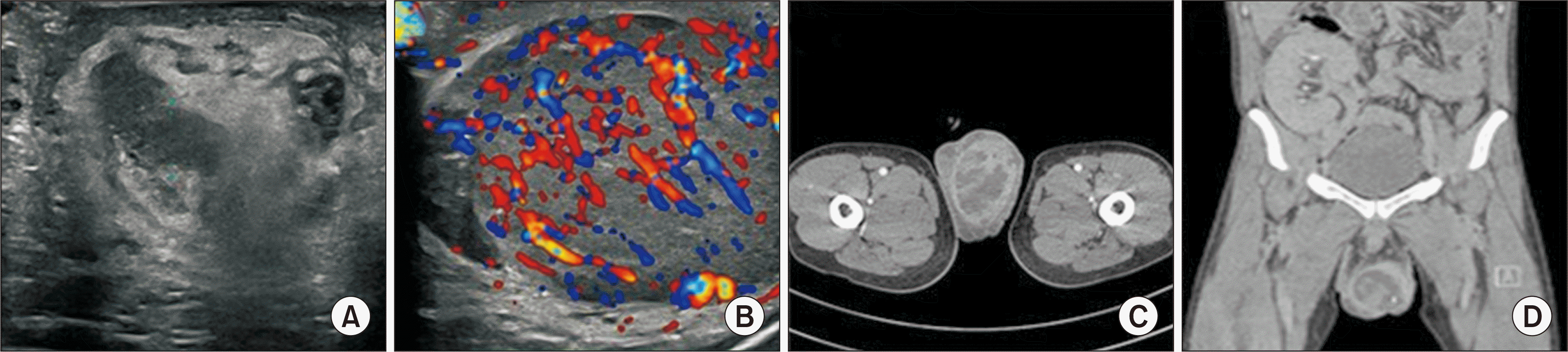
Scrotal ultrasonography (USG) suggested the possibility of testicular inflammation, indicated by an enlarged left testis with heterogeneous echotexture and markedly increased vascular flow. Additionally, there was diffuse thickening of the left epididymis from head to tail, accompanied by increased vascular flow, and diffuse thickening of the vas deferens, along with diffuse echogenic thickening of the scrotal wall (Fig. 1A and B).
Our initial impression was epididymo-orchitis, prompting us to start treatment with oral cefdinir for 3 weeks, followed by oral doxycycline for 7 days. However, the patient's condition deteriorated. He developed a fever, and his laboratory results showed elevated white blood cell counts (13,860 × 103/μL), increased levels of inflammatory markers (C-reactive protein at 4.0 mg/dL and erythrocyte sedimentation rate at 70 mm/hr), and raised creatinine levels (4.1 mg/dL). Computed tomography (CT) with contrast demonstrated heterogeneous swelling and wall thickening in the left scrotum, suggestive of an abscess, and enlarged para-aortic lymph nodes, potentially indicating reactive lymph nodes (Fig. 1C and D).
The patient was admitted for further treatment. We administered intravenous ceftriaxone and metronidazole and discontinued MMF. Additionally, we performed an abscess incision and drainage. Cultures revealed methicillin-resistant coagulase-negative staphylococci and ampicillin-sensitive enterococcus, prompting a change in medication to ampicillin/sulbactam and teicoplanin. Despite these interventions, the clinical course did not improve. Inflammatory marker levels remained elevated, and he continued to experience fever and left scrotal pain.
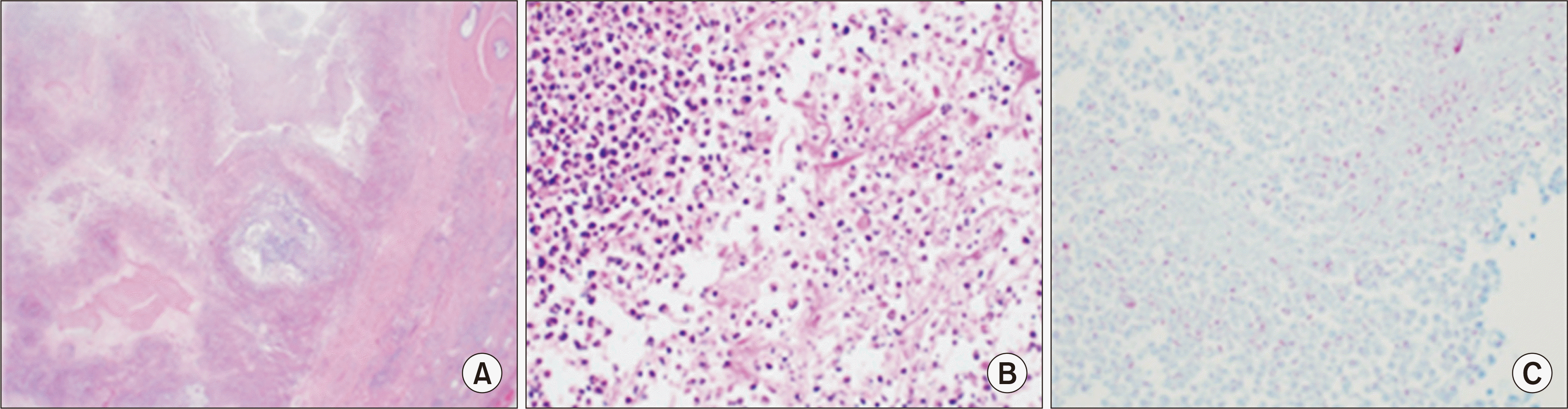
We performed a radical left orchiectomy. The biopsy revealed extensive chronic granulomatous inflammation with caseous necrosis, consistent with tuberculous orchiepididymitis (Fig. 2). The test for Mycobacterium tuberculosis (MTB) was positive, while the test for nontuberculous mycobacteria was negative. We conducted a sputum test and a CT scan to assess for pulmonary TB and systemic TB beyond the testes. Both the sputum acid-fast staining and culture were negative, and the CT scan showed no signs of TB in any locations other than the testes. The patient was treated with a quadruple anti-TB regimen consisting of isoniazid, rifabutin, pyrazinamide, and ethambutol, which led to clinical improvement. The fever subsided, and there were improvements in leukocytosis (6.02 × 103/μL), inflammatory markers (C-reactive protein, 0.2 mg/dL), and creatinine levels (2.9 mg/dL). MMF was withheld throughout the TB treatment. However, 4 months after starting TB therapy, the patient was transferred to another hospital.
KT recipients who are treated with immunosuppressive agents are susceptible to various opportunistic infections. Among these, TB poses significant risks, including graft rejection, loss, and increased mortality [4]. The incidence of TB in KT recipients, who are in an immunosuppressed state, ranges from 0.3% to 14.6%, which is approximately 50 times higher than in the general population. The Republic of Korea, having the highest incidence among OECD countries, warrants a higher level of suspicion for TB compared to other countries [4]. Genitourinary TB is an uncommon type, constituting 8%–15% of extrapulmonary TB [8]. Within this category, testicular TB is exceedingly rare among KT recipients [9].
The blood-testis barrier and the blood-epididymis barrier are essential protective structures within the male reproductive system, located between the blood vessels and the seminiferous tubules in the testes. These barriers also help limit the spread of infections such as TB within the testis. However, MTB can be transmitted through various methods including inhalation, congenital transmission, coitus, and vaccination [1]. Inhalation of droplet aerosols containing MTB is the most common mode of transmission [1]. The exact mechanism by which MTB spreads to the structures within the scrotal sac remains a topic of debate. Typically, tuberculous epididymo-orchitis results from the retrograde movement of MTB from the urinary tract to the prostate via reflux. This is followed by canalicular spread to the seminal vesicle, deferent duct, and epididymis [10,11]. Rarely, it occurs through hematogenous and lymphatic spread. Our case was typical insofar as it involved the epididymis.
Various immunosuppressants are available, including calcineurin inhibitors (tacrolimus, cyclosporine), antimetabolites (MMF, azathioprine), steroids, and T cell-depleting agents (antithymocyte globulin). Although it has not been definitively proven that specific immunosuppressants increase the risk of TB, it is well-established that the use of immunosuppressive therapy in general is associated with an increased risk of TB [4,12]. In our case, the patient was treated with a typical regimen of tacrolimus, MMF, and steroids. Additionally, the escalation of MMF dosage in response to deteriorating kidney allograft function and elevated DSA levels may also contribute to the development of TB.
KT recipients typically develop TB within the first year following transplantation. Predisposing factors for TB include old age, a body mass index below 18.5 kg/m2, diabetes mellitus, chronic kidney disease, and social risk factors such as cigarette smoking, alcoholism, and contact with TB [4,13]. In the case we are discussing, the patient underwent KT 8 years prior and presented no apparent risk factors for TB, apart from the use of immunosuppressants. This situation complicated the diagnosis of TB.
USG is highly valuable for diagnosing testicular TB, though confirming the diagnosis based solely on USG can be challenging. Testicular TB can be categorized into four types according to USG findings: (1) diffusely enlarged, heterogeneously hypoechoic; (2) diffusely enlarged, homogenously hypoechoic; (3) nodular enlargement, heterogeneously hypoechoic; and (4) miliary [14]. Doppler USG typically shows increased vascular flow. In our case, the findings align with the first classification: the left testis is enlarged with a heterogeneous echotexture and markedly increased vascular flow. Additionally, there is diffuse thickening from the head to the tail of the left epididymis with increased vascular flow, along with diffuse thickening of the vas deferens and diffuse echogenic thickening of the scrotal wall.
CT imaging in testicular TB has not been extensively researched. However, the common features of testicular TB include space-occupying lesions that are characterized as ill-defined and heterogeneous, with annular or multilocular enhancement, peripheral enhancement, and hydrocele, which may or may not include calcification [15]. The CT findings of our patient align with the previously described characteristics.
Treating active TB in KT recipients generally follows the same principles as in immunocompetent individuals. However, management becomes more complex due to the patient's immunosuppressed status. Once TB is diagnosed, it is crucial to initiate treatment immediately. It is also essential to consider the epidemiological characteristics of the patient's region and the local patterns of drug resistance. The recommended duration of TB treatment for KT recipients can range from 6 to 24 months, but expert consensus often suggests a minimum treatment period of 9 to 12 months [4]. Despite standard medical therapy, up to 50% of patients may require adjunctive surgical intervention during or after TB treatment [1]. In this patient, early suspicion of testicular TB and prompt diagnostic procedures, such as fine-needle aspiration cytology or epididymal biopsy, might have obviated the need for surgical intervention.
Genitourinary TB has a low prevalence, with only two reported cases of testicular TB accompanied by disseminated TB among KT recipients. In our case, the absence of involvement of other organs and the lack of typical risk factors, apart from the use of immunosuppressants, posed challenges in diagnosis. Additionally, the decision-making process was complicated by the failure to identify the optimal timing for intervention, ultimately necessitating an orchiectomy.
Testicular TB is rare among KT recipients; however, it is crucial to consider it in the differential diagnosis for patients presenting with testicular pain and a mass. Early and accurate diagnosis, followed by appropriate treatment, is essential to prevent unnecessary interventions.
ARTICLE INFORMATION
Conflict of Interest
Seungyeup Han is an editorial board member of the journal but was not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflict of interest relevant to this article was reported.
Author Contributions
Conceptualization: JHP. Data curation: JY, YK, WYP, JHP. Formal analysis: JY, YK, WYP, JHP. Investigation: KJ, SH, BHK, MC. Methodology: KJ, SH, BHK, MC. Software: JY, YJ, WYP, JHP. Validation: JY. Visualization: JY. Writing–original draft: JY. Writing–review & editing: YK, WYP, KJ, BHK, MC, SH, JHP.
REFERENCES
1. Muneer A, Macrae B, Krishnamoorthy S, Zumla A. 2019; Urogenital tuberculosis - epidemiology, pathogenesis and clinical features. Nat Rev Urol. 16:573–98. DOI: 10.1038/s41585-019-0228-9. PMID: 31548730.
2. Min J, Kim HW, Kim JS. 2023; Tuberculosis: Republic of Korea, 2021. Tuberc Respir Dis (Seoul). 86:67–9. DOI: 10.4046/trd.2022.0111. PMID: 36281544. PMCID: PMC9816490.
3. Son E, Jeon D. 2021; Current situation of tuberculosis and National Strategic Plan for Tuberculosis Control in Korea. J Korean Med Assoc. 64:316–23. DOI: 10.5124/jkma.2021.64.4.316.
4. Sorohan BM, Ismail G, Tacu D, Obrișcă B, Ciolan G, Gîngu C, et al. 2022; Mycobacterium tuberculosis infection after kidney transplantation: a comprehensive review. Pathogens. 11:1041. DOI: 10.3390/pathogens11091041. PMID: 36145473. PMCID: PMC9505385.
5. Singh N, Paterson DL. 1998; Mycobacterium tuberculosis infection in solid-organ transplant recipients: impact and implications for management. Clin Infect Dis. 27:1266–77. DOI: 10.1086/514993. PMID: 9827281.
6. Canet E, Dantal J, Blancho G, Hourmant M, Coupel S. 2011; Tuberculosis following kidney transplantation: clinical features and outcome. A French multicentre experience in the last 20 years. Nephrol Dial Transplant. 26:3773–8. DOI: 10.1093/ndt/gfr156. PMID: 21467129.
7. Bausch K, Mantica G, Smith EJ, Bartoletti R, Bruyère F, Cai T, et al. 2024; Genitourinary tuberculosis: a brief manual for urologists on diagnosis and treatment from the European Association of Urology Urological Infections Panel. Eur Urol Focus. 10:77–9. DOI: 10.1016/j.euf.2023.07.006. PMID: 37541917.
8. Hadadi A, Pourmand G, Mehdipour-Aghabagher B. 2012; Unilateral testicular tuberculosis: case report. Andrologia. 44:70–2. DOI: 10.1111/j.1439-0272.2010.01097.x. PMID: 21615454.
9. Nayak S, Satish R. 2007; Genitourinary tuberculosis after renal transplantation-a report of three cases with a good clinical outcome. Am J Transplant. 7:1862–4. DOI: 10.1111/j.1600-6143.2007.01853.x. PMID: 17524075.
10. Viswaroop BS, Kekre N, Gopalakrishnan G. 2005; Isolated tuberculous epididymitis: a review of forty cases. J Postgrad Med. 51:109–11. PMID: 16006701.
11. Wise GJ, Shteynshlyuger A. 2008; An update on lower urinary tract tuberculosis. Curr Urol Rep. 9:305–13. DOI: 10.1007/s11934-008-0053-9. PMID: 18765130.
12. Ha YE, Joo EJ, Park SY, Wi YM, Kang CI, Chung DR, et al. 2012; Tacrolimus as a risk factor for tuberculosis and outcome of treatment with rifampicin in solid organ transplant recipients. Transpl Infect Dis. 14:626–34. DOI: 10.1111/j.1399-3062.2012.00721.x. PMID: 22372581.
13. Morris MI, Daly JS, Blumberg E, Kumar D, Sester M, Schluger N, et al. 2012; Diagnosis and management of tuberculosis in transplant donors: a donor-derived infections consensus conference report. Am J Transplant. 12:2288–300. DOI: 10.1111/j.1600-6143.2012.04205.x. PMID: 22883346.
14. Muttarak M, Peh WC, Lojanapiwat B, Chaiwun B. 2001; Tuberculous epididymitis and epididymo-orchitis: sonographic appearances. AJR Am J Roentgenol. 176:1459–66. DOI: 10.2214/ajr.176.6.1761459. PMID: 11373214.
15. Li S, Chen B, Fang X, Wan M, Cao D, Xu H, et al. 2021; A better understanding of testicular and/or epididymal tuberculosis based on clinical, ultrasonic, computed tomography, and magnetic resonance imaging features at a high-volume institute in the modern era. Quant Imaging Med Surg. 11:2465–76. DOI: 10.21037/qims-20-1005. PMID: 34079716. PMCID: PMC8107328.
Fig. 1
Imaging examination of the testis. (A) Ultrasonography: diffusely enlarged, heterogeneously hypoechoic testis lesion. (B) Doppler ultrasonography: increased vascular flow. (C) Computed tomography, transverse view: left scrotal wall thickening and enlargement. (D) Computed tomography, coronal view: left scrotal wall thickening and enlargement.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download