Abstract
Background
This study aimed to investigate the analgesic and preventive effect of low-level laser therapy (LLLT) on the incisional pain model and spinal nerve ligation (SNL) model in rats and identify the possible mechanisms of action.
Methods
Male Sprague-Dawley rats were used, divided into different treatment groups. The single application group received LLLT before or after skin incision or SNL. The consecutive application group received LLLT for six consecutive days post-incision, three days pre-incision, or three consecutive days pre-SNL. The control group underwent skin incision or SNL without LLLT. The von Frey test was used to quantify the pain associated with mechanical allodynia. Pro-inflammatory cytokine level and alterations in nerve growth factor (NGF) expression were measured by using ELISA and immunohistochemistry, respectively in the skin, muscle of the paw, and spinal cord dorsal horn (SCDH).
Results
In the incisional pain model, LLLT showed significant analgesic and preventive effect. LLLT ameliorated SNL-induced mechanical allodynia but LLLT had no preventive effect. LLLT decreased interleukin-1β (IL-1β) expression levels in the skin, muscle, and SCDH and reduced the optical density of skin and spinal cord NGF in the incisional pain model.
To date, chronic pain, including neuropathic pain and chronic post-surgical pain (CPSP), is challenging to treat and there has been an unmet need for effective treatments. As in the case of neuropathic pain, poorly controlled postoperative pain is associated with a risk factor for CPSP and long term high-dose opioid use, and appropriate prevention and treatment are also required [1].
Non-surgical physiotherapy such as transcutaneous electrical nerve stimulation, acupuncture, and laser therapy are potential management options and are currently used for patients with chronic peripheral neuropathic pain. Low-level laser therapy (LLLT) uses low-level lasers or light-emitting diodes to stimulate tissues non-invasively and has been shown to reduce inflammation and edema, induce analgesia, and improve the healing process in a range of musculoskeletal pathologies [2]. Clinically, LLLT can treat musculoskeletal nociceptive pain in patients with plantar fasciitis [3] and neuropathic pain including trigeminal neuralgia and carpal tunnel syndrome [4,5].
Pro-inflammatory cytokines play a crucial role in the initiation and maintenance of inflammation, such as occurs in arthritis and neurological conditions [6]. Nerve growth factor (NGF) regulates injurious sensation by modulating the release of inflammatory mediators, the activities of ion channel/receptors and genes involved in pain mediation, and local neuronal sprouting [7]. While the exact mechanism is unknown, LLLT is thought to alter cellular signaling pathways, oxidative stress, and the expression of anti-inflammatory mediators [8]. Among the reported effects of LLLT is a reduction in the expression and synthesis of pro-inflammatory cytokines, such as interleukin-1β (IL-1β), tumor necrosis factor-alpha, and IL-6, in addition to regulating the immune response [9,10]. In addition to its effects on the release of inflammatory mediators, LLLT is thought to reduce pain by decreasing NGF expression. The results of these biochemical and cellular changes in animals and patients include such benefits as increased healing in surgical wounds, pain reduction in neuropathies, and amelioration of damage after nerve injury.
According to these above considerations, the aims of this study were to investigate (1) the therapeutic and preventive effects of LLLT on an incisional pain model, (2) the therapeutic and preventive effects of LLLT on a spinal nerve ligation (SNL) model in rats, and (3) the mechanism of action of LLLT in peripheral tissues and the spinal cord dorsal horn (SCDH).
Male Sprague-Dawley rats weighing between 180–220 g (for the SNL model) and 260–300 g (for the incisional pain model) were housed in a 22°C environment with a 12 hr/12 hr light/dark cycle at the Chonnam National University animal facility. Food and water were available without restriction. The animal experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Chonnam National University Hospital (Approved No. CNUHIACUC-21024) and performed in accordance with ethical standards for the investigation of experimental pain in animals. At the end of the study, the rats were euthanized using sevoflurane inhalation anesthesia.
The incision model used in this study was developed by Brennan et al. [11]. After being anesthetized with sevoflurane, a 1-cm longitudinal incision was made in the left paw, approximately 0.5 cm from the edge of the heel and extending toward the middle of the foot heel. The subcutaneous tissue was exposed, and tweezers were used to separate the flexor tendon from its underlying tissue. The flexor tendon was raised and bluntly separated. The skin was sutured to maintain the incision in a constant state of irritation and pain. The von Frey test was performed to measure the mechanical withdrawal threshold and the 50% probability paw withdrawal threshold (PWT) was calculated using the up and down method [12], with measurements taken once. It was assigned a 15 g PWT (the cut-off value). The rats were placed in a mesh cage for 30 minutes after which eight von Frey wires (0.4, 0.6, 1, 2, 4, 6, 8, and 15 g) were applied to the perimeter of the incised hind paw or to the corresponding area on the unincised paw. A positive reaction was shown by a retraction reflex of the paw within 5 seconds. Rats exhibiting a PWT of 5.0 grams or less were considered to have developed significant mechanical allodynia and were included in further assessments to evaluate the efficacy of treatments targeting this condition.
The SNL model was established based on Chung's model for neuropathic rats [13]. The rats were sedated by inhalation of 3% sevoflurane and positioned in a prone position. The skin was cut 1.5 cm above and below the mid-back, exposing the sacral region, the L6 transverse process, and spinal nerves L4 and L5. The L5 and L6 were separated, and each was tightly ligated with a 6-0 black silk thread. The surrounding skin and muscles were then sutured. Mechanical allodynia was measured using von Frey filaments in the same manner as the incisional pain model. Rats with a PWT of 4 grams or less demonstrated significant mechanical allodynia on day 7 and were included in further efficacy evaluations.
A dual-wavelength laser device (WellsCare Co., Ltd.) was used, delivering red (650 nm) and near-infrared (830 nm) laser light at a 40 mW intensity (± 10%). The rats were rendered unconscious with 2% sevoflurane gas. After creating the incisional pain model, PWT was measured 2 hours later, and the incision was subsequently irradiated using LLLT for either 30 or 60 minutes depending on the experimental group. PWT measurements were taken again at the same time the following day, followed by LLLT according to the group. This procedure was consistently repeated for a duration of 6 days. To ensure consistent laser exposure, the lasers were applied vertically to the left hindpaw, specifically targeting the sole area. For the SNL model, PWT was assessed 7 days post-model establishment to verify successful model creation. On the same day, LLLT was irradiated for 30, 60, or 120 minutes according to the experimental group. PWT was then measured every 30 minutes for 3 hours. Preventive LLLT was administered prior to the procedures in both models.
The expression levels of the cytokines IL-1β, IL-6, and Rat IL-10 were quantified using specific ELISA kits (RLB00, R&D; R6000B, R&D; and R1000, R&D Systems). Absorbance was determined spectrophotometrically at 450 nm. The assays were conducted following the manufacturer’s prescribed protocols. ELISA and immunohistochemistry were only measured in the incisional model. Tissue samples from the skin (approximately 0.35 square centimeters) of the sole, muscle (approximately 0.35 square centimeters) from the sole area, and SCDH (collected from the lumbar enlargement, specifically L2-L4) were collected. Three days post-laser treatment, deep anesthesia was induced with sevoflurane, and the chest was opened to access the heart. Following perfusion with phosphate-buffered saline (PBS), tissues were fixed in 4% paraformaldehyde in PBS (0.01 M, pH 7.4) at 4°C. Tissues were then embedded in O.C.T. compound, frozen, and sectioned at 20 μm thickness. Sections were treated for endogenous peroxidase activity, blocked with milk, and incubated with rabbit anti-NGF antibody (1:200, Abcam, ab52918). Skin and muscle tissues were analyzed using a light microscope (Model: BX53, Olympus) at 100× magnification, while the SCDH sections were analyzed at 40× magnification. Detection was performed using biotinylated secondary antibodies (1:200, Vector Laboratories, PK6101) followed by ABC complex, and DAB substrate, with sections analyzed using ImageJ software.
All data are expressed as the mean ± standard error of the mean. To calculate the percentage of the maximum possible effect (%MPE) in the context of the study, the following formula is used: %MPE = [(Post-treatment threshold−Baseline threshold) / (Maximum possible threshold−Baseline threshold)] × 100. The analysis of the PWTs in the incision and SNL models, as well as the laser intensity results, were evaluated using one-way analysis of variance with Bonferroni comparison for the post-hoc test. The statistical analysis was performed using SPSS 25.0 software (IBM Corp.). P < 0.05 was considered statistically significant. Data for ELISA and immunohistochemistry are presented as mean ± standard error of the mean.
The incision model generates unilateral nociceptive mechanical allodynia. As illustrated in Fig. 1, the PWTs in the 30-minute and 60-minute LLLT groups displayed significantly less sensitivity to incisional pain compared to those in the control group. One group received LLLT for 1 hour on the day before the incision, and another group received LLLT for 1 hour each day for three days before the incision. In this group, a significant attenuation effect was observed on the third day post-incision, compared to one week post-incision in the control group, as shown in Fig. 2.
SNL generates ipsilateral injurious mechanical allodynia. Fig. 3 depicts the mechanical withdrawal thresholds for the ipsilateral hind paw. Notably, 60-minute and 120-minute LLLT sessions significantly alleviated pain in the SNL model compared to the control group. LLLT was administered to the rats for 1 hour each day for three days before the SNL operation. No significant changes were observed in this group, as indicated in Fig. 4.
The study measured levels of the pro-inflammatory cytokines IL-1β, IL-6, and IL-10. While IL-6 and IL-10 levels remained unchanged, a significant increase in IL-1β was observed in the incision model rats. Previous research showed that LLLT could reduce IL-1β levels in the skin and muscle of control rats, these levels were notably lower in the LLLT-treated rats. No significant changes in IL-1β were detected in the SCDH, as demonstrated in Fig. 5.
An immunohistochemistry approach was used to assess the effect of LLLT on NGF levels in the incision model. According to Fig. 6, on day 3 post-incision, NGF levels in the spinal cord and skin tissues were higher in the incision group than in the control group. However, after 60 minutes of LLLT, NGF levels significantly decreased in the spinal cord.
Our study showed that LLLT led to an improvement in mechanical allodynia pain in rats with an incision in the ipsilateral hind paw and also had a preventive effect when administered prior to the incision and 3 days thereafter. In SNL rats, LLLT irradiation reduced the mechanical allodynia in the ipsilateral hind paw but had no preventive effect. LLLT decreased IL-1β expression levels in the skin, muscle, and SCDH and reduced the optical density of skin and spinal cord NGF in the incisional pain model.
LLLT has a long history of use, and clinical and basic scientific evidence supports its role in pain management. It also has few side effects and is well tolerated by the elderly. Because LLLT does not correct situations involving structural deficits or instabilities whether in bone or in soft tissue, LLLT should be used as an adjuvant therapy for pain relief in patients with nociceptive and neuropathic pain [2].
Clinically, LLLT contributes to the management of the acute phase of inflammation. In a systematic review and meta-analysis, LLLT has proven to be effective in reducing pain caused by lateral epicondylitis, fracture, and plantar fasciitis [14–16]. In an arthritis rat model, LLLT can also mitigate inflammatory symptoms including swelling and inflammatory pain in the affected knee joint and prevent secondary hyperalgesia [17]. Most of the previous studies have reported the effect of a single application of LLLT on postoperative pain at 24 hours, but the present study showed the preventive effect and the cumulative therapeutic effect of LLLT when it was repeatedly used before or after surgical incision, respectively. To the authors’ knowledge, this is the first report on the preventive effect of LLLT on post-operative pain. Therefore, the authors believe that LLLT may be a potentially beneficial modality for the treatment of acute pain and secondary prevention of chronic pain in post-operative patients.
LLLT was found to be beneficial for the treatment of peripheral neuropathic pain secondary to trigeminal neuralgia and carpal tunnel syndrome [4,5]. In chronic constriction and sciatic nerve crush-type injury, LLLT has demonstrated positive effects in the treatment of peripheral nerve injury [18,19], and LLLT also ameliorated SNL-induced mechanical allodynia in this study. A previous study reported that a 0.3 μg dose of intrathecal morphine resulted in approximately 50% MPE in the SNL model. In this study, LLLT showed a similar antiallodynic effect to the above dose of morphine [20]. With regard to the above considerations, LLLT can be effectively used for pain relief as an adjuvant therapy in patients with neuropathic pain and neurologic deficits caused by peripheral nerve damage. Although LLLT showed the preventive effect in an incisional pain model, there was no preventive effect of LLLT on spinal nerve ligated neuropathic pain in this study. The reason for the lack of preventive effect of LLLT in the SNL model may be related to the following: the effect of LLLT is dependent on the type of laser utilized, as well as the dose and duration of treatment and the pain model.
In this study, LLLT decreased IL-1β expression levels in the skin, muscle, and SCDH, and reduced the optical density of skin and spinal cord NGF in the incisional pain model. Although, the biological mechanisms of LLLT in acute pain remain unclear, the anti-inflammatory action of LLLT may inhibit and/or attenuate the release of inflammatory mediators and pain marker [9]. Cytokines and other inflammatory mediators are produced in large amounts near incisional wounds and in other inflammation-based pain models, such as rheumatoid arthritis and neuropathic pain [21]. IL-1β is a key mediator of inflammation and its intradermal injection into the hind paw skin was shown to cause intense mechanical allodynia [22]. Although the association between LLLT and IL-1β is currently unknown, a previous study showed that LLLT may be effective in reducing inflammation and IL-1β levels under specific settings [23,24]. For example, LLLT reduced IL-1β levels in patients with periodontitis, improving clinical results [23], as well as inflammation, and IL-1β levels in patients with osteoarthritis [24]. In addition to relieving mechanical allodynia, LLLT reduced the increased expression of the pro-inflammatory cytokine IL-1β triggered by an incisional pain model in the peripheral tissue and SCDH. In the present study, LLLT decreased the level of IL-1β expression induced by the incision model, whereas the levels of IL-6 were not significantly altered. These results provide further evidence that IL-1β plays a crucial role in the initiation and maintenance of pain in an incision model.
NGF plays a crucial function in the pain signaling pathway, is associated with the development of postoperative pain, and plays a key role in pain development and maintenance [25]. Inhibiting NGF was shown to reduce postoperative pain in animal models [26]. NGF acts on injury receptors expressed on the main sensory neurons that detect painful stimuli, is an important pain mediator in the skin [27], and is produced in response to tissue injury and inflammation. Increased levels of NGF can lead to the sensitization of injury receptors, resulting in nociceptive hypersensitivity (increased pain sensitivity) and nociceptive abnormalities (response to normally non-painful stimuli) [28]. In peripheral tissues, including skin and muscle, NGF levels increase following tissue injury, so that peripheral injury receptors become sensitized, leading to higher pain sensitivity in the affected area [29]. In this study, incision resulted in increased levels of NGF in the affected paw, which contributed to the development of peripheral sensitization and heightened pain sensitivity. LLLT decreased NGF expression in the incision model rats. Previous studies found that LLLT altered the expression of genes and cellular signaling pathways that regulate NGF synthesis and secretion [30]. The anti-inflammatory properties of LLLT may play a role in reducing NGF expression and in enhancing recovery after injury, as suggested by the results of this study. Some of the limitations of the present study include: 1) This study was performed using an animal model of incisional pain and an SNL model. Thus, further clinical studies are necessary to evaluate LLLT efficacy in human patients. 2) The present study was performed using a small number of rats in each group; and higher numbers of rats are needed to increase reliability.
In conclusion, LLLT alleviated incision pain and spinal nerve ligated-neuropathic pain in rats, as well as reducing the levels of IL-1β and NGF in the peripheral tissue and SCDH in the incisional pain model. The authors conclude that LLLT may be effective for postoperative pain and peripheral nerve injury induced neuropathic pain in patients.
Notes
DATA AVAILABILITY
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
REFERENCES
1. Pirie K, Traer E, Finniss D, Myles PS, Riedel B. 2022; Current approaches to acute postoperative pain management after major abdominal surgery: a narrative review and future directions. Br J Anaesth. 129:378–93. DOI: 10.1016/j.bja.2022.05.029. PMID: 35803751.
2. Cotler HB, Chow RT, Hamblin MR, Carroll J. 2015; The use of low level laser therapy (LLLT) for musculoskeletal pain. MOJ Orthop Rheumatol. 2:00068. DOI: 10.15406/mojor.2015.02.00068. PMID: 26858986. PMCID: PMC4743666.
3. Koz G, Kamanli A, Kaban N, Harman H. 2023; Efficacies of extracorporeal shockwave therapy and low-level laser therapy in patients with plantar fasciitis. Foot Ankle Surg. 29:223–7. DOI: 10.1016/j.fas.2023.01.009. PMID: 36737392.
4. Falaki F, Nejat AH, Dalirsani Z. 2014; The effect of low-level laser therapy on trigeminal neuralgia: a review of literature. J Dent Res Dent Clin Dent Prospects. 8:1–5. DOI: 10.5681/joddd.2014.001. PMID: 25024832. PMCID: PMC4091693.
5. Lazovic M, Ilic-Stojanovic O, Kocic M, Zivkovic V, Hrkovic M, Radosavljevic N. 2014; Placebo-controlled investigation of low-level laser therapy to treat carpal tunnel syndrome. Photomed Laser Surg. 32:336–44. DOI: 10.1089/pho.2013.3563. PMID: 24905929.
6. Hung AL, Lim M, Doshi TL. 2017; Targeting cytokines for treatment of neuropathic pain. Scand J Pain. 17:287–93. DOI: 10.1016/j.sjpain.2017.08.002. PMID: 29229214. PMCID: PMC5774983.
7. Reis C, Chambel S, Ferreira A, Cruz CD. 2022; Involvement of nerve growth factor (NGF) in chronic neuropathic pain - a systematic review. Rev Neurosci. 34:75–84. DOI: 10.1515/revneuro-2022-0037. PMID: 35792932.
8. Dos Anjos LMJ, Salvador PA, de Souza ÁC, de Souza da Fonseca A, de Paoli F, Gameiro J. 2019; Modulation of immune response to induced-arthritis by low-level laser therapy. J Biophotonics. 12:e201800120. DOI: 10.1002/jbio.201800120. PMID: 30203577.
9. de Almeida P, Tomazoni SS, Frigo L, de Carvalho Pde T, Vanin AA, Santos LA, et al. 2014; What is the best treatment to decrease pro-inflammatory cytokine release in acute skeletal muscle injury induced by trauma in rats: low-level laser therapy, diclofenac, or cryotherapy? Lasers Med Sci. 29:653–8. DOI: 10.1007/s10103-013-1377-3. PMID: 23812849.
10. de Lima FM, Villaverde AB, Albertini R, Corrêa JC, Carvalho RL, Munin E, et al. 2011; Dual Effect of low-level laser therapy (LLLT) on the acute lung inflammation induced by intestinal ischemia and reperfusion: action on anti- and pro-inflammatory cytokines. Lasers Surg Med. 43:410–20. DOI: 10.1002/lsm.21053. PMID: 21674546.
11. Brennan TJ, Vandermeulen EP, Gebhart GF. 1996; Characterization of a rat model of incisional pain. Pain. 64:493–502. DOI: 10.1016/0304-3959(95)01441-1. PMID: 8783314.
12. Deuis JR, Dvorakova LS, Vetter I. 2017; Methods used to evaluate pain behaviors in rodents. Front Mol Neurosci. 10:284. DOI: 10.3389/fnmol.2017.00284. PMID: 28932184. PMCID: PMC5592204.
13. Ho Kim S, Mo Chung J. 1992; An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain. 50:355–63. DOI: 10.1016/0304-3959(92)90041-9. PMID: 1333581.
14. Bjordal JM, Lopes-Martins RA, Joensen J, Couppe C, Ljunggren AE, Stergioulas A, et al. 2008; A systematic review with procedural assessments and meta-analysis of low level laser therapy in lateral elbow tendinopathy (tennis elbow). BMC Musculoskelet Disord. 9:75. DOI: 10.1186/1471-2474-9-75. PMID: 18510742. PMCID: PMC2442599.
15. Neto FCJ, Martimbianco ALC, de Andrade RP, Bussadori SK, Mesquita-Ferrari RA, Fernandes KPS. 2020; Effects of photobiomodulation in the treatment of fractures: a systematic review and meta-analysis of randomized clinical trials. Lasers Med Sci. 35:513–22. DOI: 10.1007/s10103-019-02779-4. PMID: 30982176.
16. Dos Santos SA, Sampaio LM, Caires JR, Fernandes GHC, Marsico A, Serra AJ, et al. 2019; Parameters and effects of photobiomodulation in plantar fasciitis: a meta-analysis and systematic review. Photobiomodul Photomed Laser Surg. 37:327–35. DOI: 10.1089/photob.2018.4588. PMID: 31107161.
17. Okita S, Sasaki R, Kondo Y, Sakamoto J, Honda Y, Okita M. 2023; Effects of low-level laser therapy on inflammatory symptoms in an arthritis rat model. J Phys Ther Sci. 35:55–9. DOI: 10.1589/jpts.35.55. PMID: 36628144. PMCID: PMC9822828.
18. Dias FJ, Fazan VPS, Cury DP, de Almeida SRY, Borie E, Fuentes R, et al. 2019; Growth factors expression and ultrastructural morphology after application of low-level laser and natural latex protein on a sciatic nerve crush-type injury. PLoS One. 14:e0210211. DOI: 10.1371/journal.pone.0210211. PMID: 30625210. PMCID: PMC6326513.
19. Hsieh YL, Chou LW, Chang PL, Yang CC, Kao MJ, Hong CZ. 2012; Low-level laser therapy alleviates neuropathic pain and promotes function recovery in rats with chronic constriction injury: possible involvements in hypoxia-inducible factor 1α (HIF-1α). J Comp Neurol. 520:2903–16. DOI: 10.1002/cne.23072. PMID: 22351621.
20. Lee HG, Park SK, Yoon MH. 2010; Potentiation of morphine antiallodynic efficacy by ACPT-III, a group III metabotropic glutamate receptor agonist, in rat spinal nerve ligation-induced neuropathic pain. Pharmacol Biochem Behav. 96:108–13. DOI: 10.1016/j.pbb.2010.04.014. PMID: 20434481.
21. Carvalho B, Clark DJ, Angst MS. 2008; Local and systemic release of cytokines, nerve growth factor, prostaglandin E2, and substance P in incisional wounds and serum following cesarean delivery. J Pain. 9:650–7. DOI: 10.1016/j.jpain.2008.02.004. PMID: 18394968.
22. Binshtok AM, Wang H, Zimmermann K, Amaya F, Vardeh D, Shi L, et al. 2008; Nociceptors are interleukin-1beta sensors. J Neurosci. 28:14062–73. DOI: 10.1523/JNEUROSCI.3795-08.2008. PMID: 19109489. PMCID: PMC2690713.
23. Kellesarian SV, Malignaggi VR, Majoka HA, Al-Kheraif AA, Kellesarian TV, Romanos GE, et al. 2017; Effect of laser-assisted scaling and root planing on the expression of pro-inflammatory cytokines in the gingival crevicular fluid of patients with chronic periodontitis: a systematic review. Photodiagnosis Photodyn Ther. 18:63–77. DOI: 10.1016/j.pdpdt.2017.02.010. PMID: 28232271.
24. Nambi G. 2021; Does low level laser therapy has effects on inflammatory biomarkers IL-1β, IL-6, TNF-α, and MMP-13 in osteoarthritis of rat models-a systemic review and meta-analysis. Lasers Med Sci. 36:475–84. DOI: 10.1007/s10103-020-03124-w. PMID: 32833088.
25. Barker PA, Mantyh P, Arendt-Nielsen L, Viktrup L, Tive L. 2020; Nerve growth factor signaling and its contribution to pain. J Pain Res. 13:1223–41. DOI: 10.2147/JPR.S247472. PMID: 32547184. PMCID: PMC7266393.
26. Rukwied R, Mayer A, Kluschina O, Obreja O, Schley M, Schmelz M. 2010; NGF induces non-inflammatory localized and lasting mechanical and thermal hypersensitivity in human skin. Pain. 148:407–13. DOI: 10.1016/j.pain.2009.11.022. PMID: 20022698.
27. Nicol GD, Vasko MR. 2007; Unraveling the story of NGF-mediated sensitization of nociceptive sensory neurons: ON or OFF the Trks? Mol Interv. 7:26–41. DOI: 10.1124/mi.7.1.6. PMID: 17339604.
28. Hoheisel U, Unger T, Mense S. 2005; Excitatory and modulatory effects of inflammatory cytokines and neurotrophins on mechanosensitive group IV muscle afferents in the rat. Pain. 114:168–76. DOI: 10.1016/j.pain.2004.12.020. PMID: 15733642.
29. Hayashi K, Shiozawa S, Ozaki N, Mizumura K, Graven-Nielsen T. 2013; Repeated intramuscular injections of nerve growth factor induced progressive muscle hyperalgesia, facilitated temporal summation, and expanded pain areas. Pain. 154:2344–52. DOI: 10.1016/j.pain.2013.07.007. PMID: 23867729.
30. Yazdani SO, Golestaneh AF, Shafiee A, Hafizi M, Omrani HA, Soleimani M. 2012; Effects of low level laser therapy on proliferation and neurotrophic factor gene expression of human schwann cells in vitro. J Photochem Photobiol B. 107:9–13. DOI: 10.1016/j.jphotobiol.2011.11.001. PMID: 22178388.
Fig. 1
The influence of low-level laser treatment (LLLT) on analgesic recovery in a rat incision model over the course of 7 days. (A) Paw withdrawal thresholds (PWTs) to von Frey stimulation; (B) the percentage of the maximum possible effect (%MPE). PWT was measured and LLLT was subsequently irradiated for either 30 or 60 minutes (indicated by upward arrows in the figure). IM: incision model. Tx: treatment. The results are reported as the mean and SEM, with significant improvements in PWTs and %MPE compared to control group: *P < 0.05, **P < 0.01, ***P < 0.001, n = 6 rats/group (one-way ANOVA and Bonferroni post-hoc test).
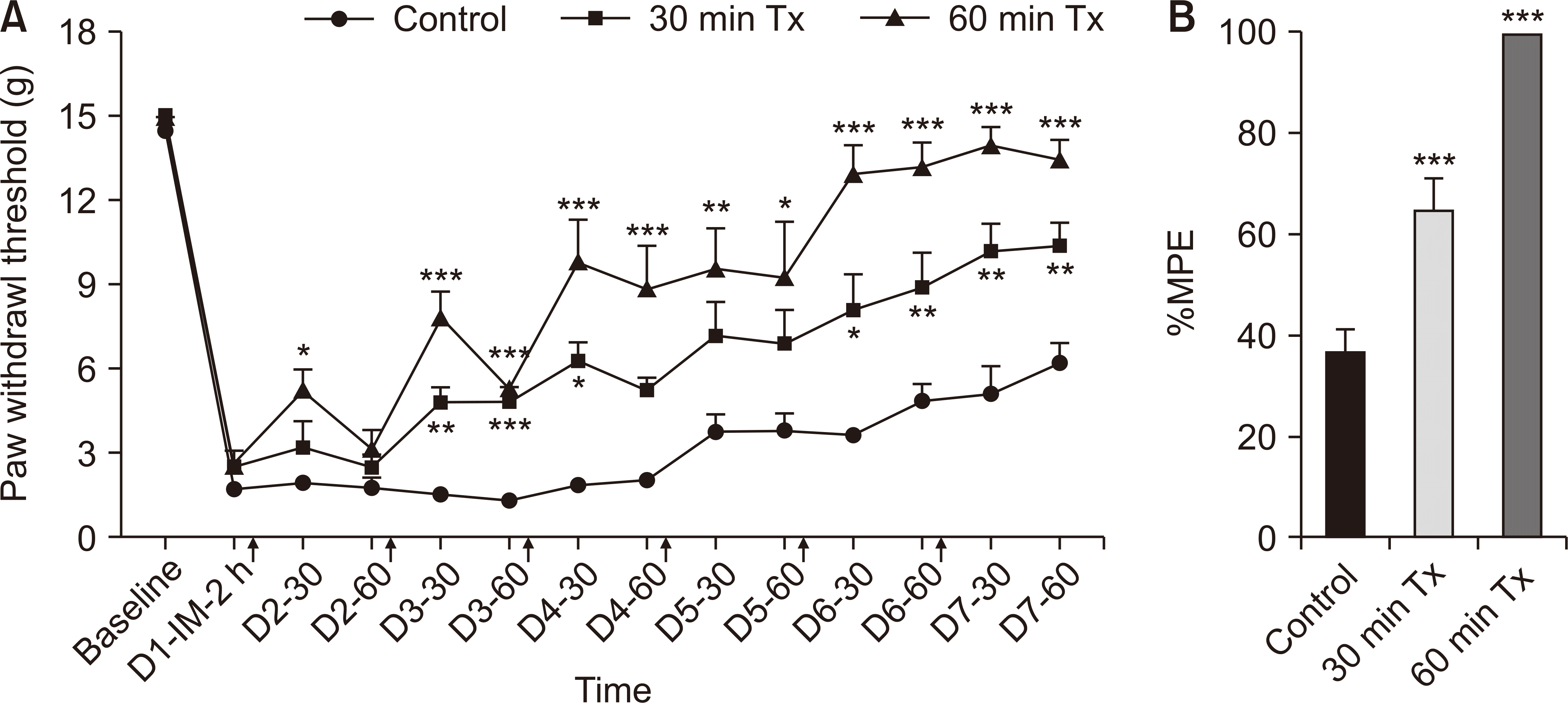
Fig. 2
Preventative effect of low-level laser treatment (LLLT) in an incision model. (A) Paw withdrawal thresholds (PWTs) to von Frey stimulation; (B) the percentage of the maximum possible effect (%MPE). In the pre-treated incision model, Pre-1Tx group received LLLT for 1 hour on the day before the incision, and Pre-3Tx group received LLLT for 1 hour each day for three days before the incision. IM: incision model, Pre Tx: pretreatment. The results are reported as the mean and SEM, with significant differences observed compared to control group: *P < 0.05, **P < 0.01, n = 6 rats/group (one-way ANOVA and Bonferroni post-hoc test).
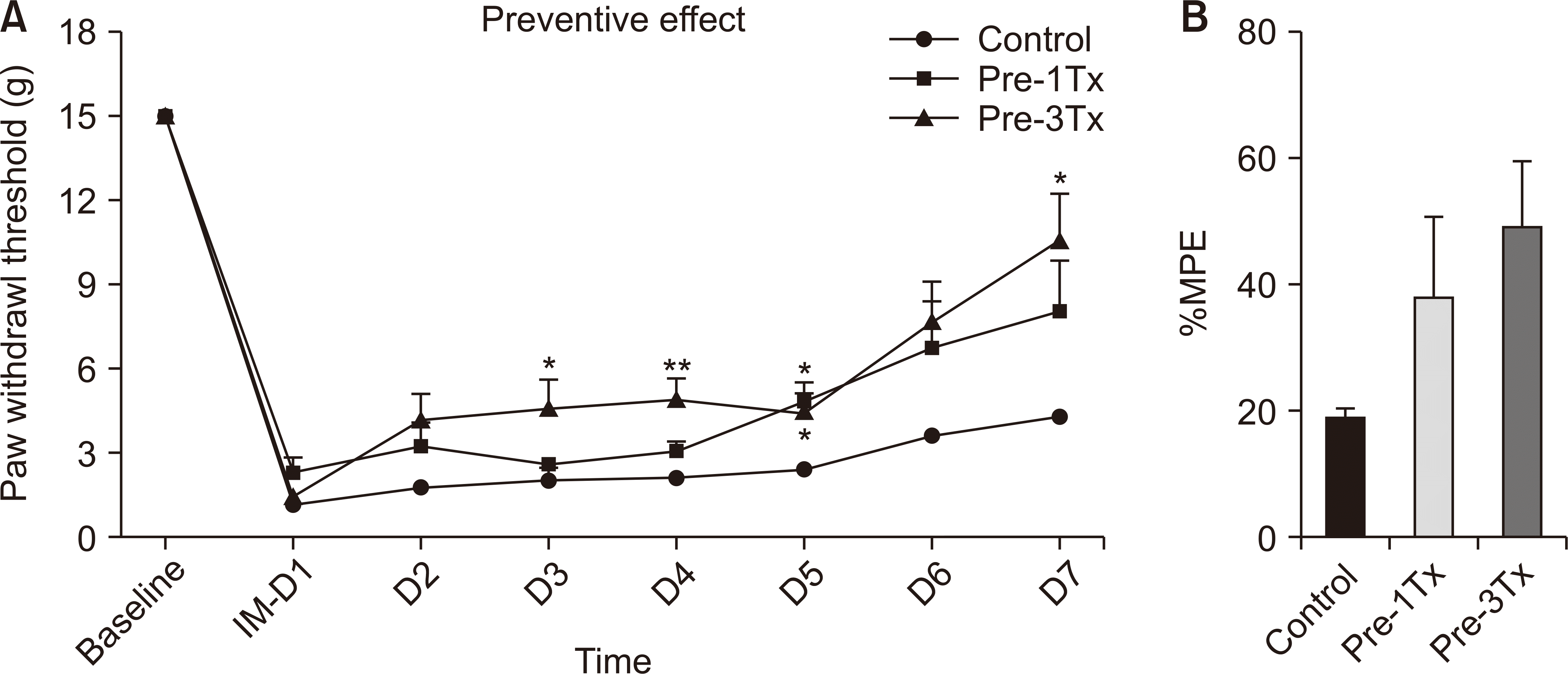
Fig. 3
Effect of low-level laser treatment (LLLT) on analgesic recovery in spinal nerve ligation (SNL) rats over 3 hours. (A) Paw withdrawal threshold (PWT) to von Frey stimulation; (B) the percentage of the maximum possible effect (%MPE). Behavioral tests were performed after 30, 60 and 120 minutes of irradiation. The results are reported as the mean and SEM, with statistically significant improvements in PWTs and %MPE compared to control group: *P < 0.05, **P < 0.01, ***P < 0.001, n = 6 rats/group (one-way ANOVA and Bonferroni post-hoc test).
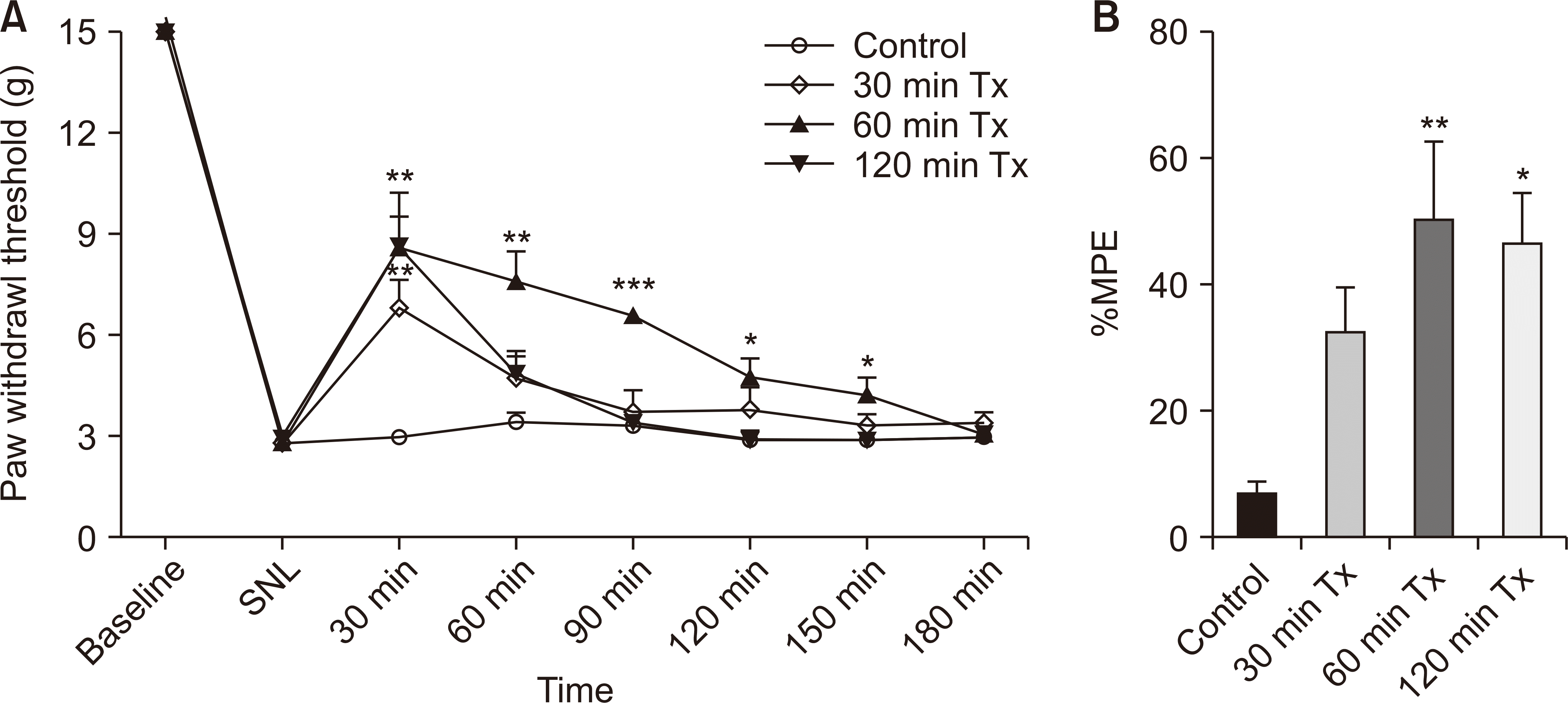
Fig. 4
Preventative effect of low-level laser treatment (LLLT) in the spinal nerve ligation (SNL) model. In the pre-treated SNL model, Pre-3Tx group received LLLT for 1 hour each day for three days before the SNL was made. Pre Tx: pretreatment. The results showed no significant difference between the two groups, are presented with the P values where applicable, n = 6 rats/group (independent sample t-test). The results are reported as the mean and SEM.
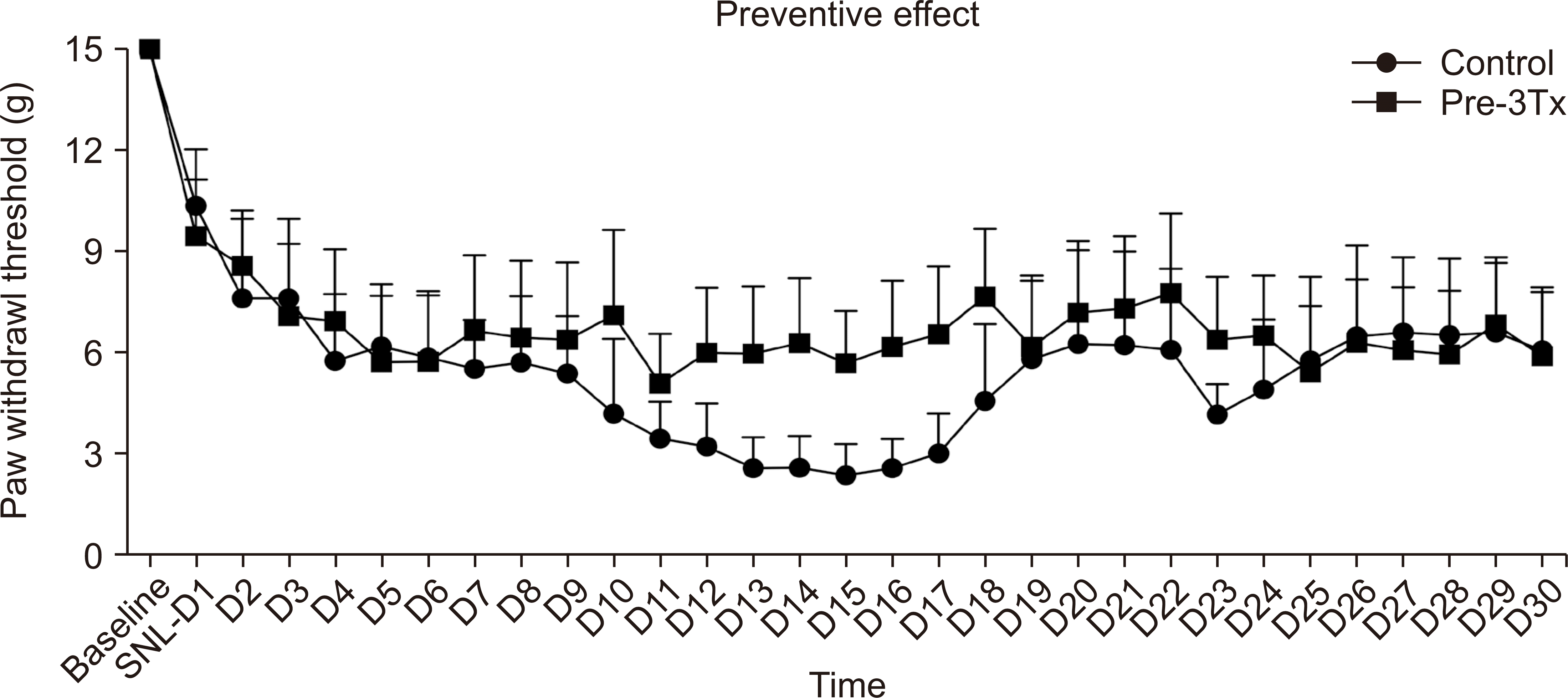
Fig. 5
Cytokine levels in naive groups, control groups vs. low-level laser treatment (LLLT)-treated groups. Interleukin (IL)-1β, IL-6, and IL-10 (pg/mL) in (A, D, G) skin, (B, E, H) muscle, and (C, F, I) spinal cord dorsal horn (SCDH) tissues. Showing significant reductions in IL-1β with LLLT treatment. This indicates LLLT's role in reducing inflammatory responses associated with pain. ***P < 0.001 compared with naive group (A, C, D, F, G, I) n = 6 rats/group; ##P < 0.01, ###P < 0.001 compared with control group (B, E, H) n = 5 rats/group (one-way ANOVA and Bonferroni post-hoc test).
Fig. 6
Immunohistochemical outcomes and optical density (OD) of nerve growth factor (NGF) expression in each group. (A) Representative microphotographs depicting NGF expression in the skin. (B) Relative OD of NGF in the skin of rats in the different groups. (C) Representative microphotographs depicting NGF expression in the spinal cord dorsal horn (SCDH) in each group. (D) Comparison of the relative OD of NGF in the SCDH of the different groups. Immunohistochemical examination revealed significantly higher NGF expression in the control group than in the naive group and significant differences between the control group and LLLT group. Scale bar = 400 μm (A) and 1,000 μm (C). *P < 0.05, **P < 0.01 compared with naive group, #P < 0.05, ##P < 0.01 compared with control group, n = 5 rats/group (one-way ANOVA and Bonferroni post-hoc test).





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download