Abstract
Pancreaticobiliary maljunction (PBM) is associated with the development of neoplasms of bile ducts. Cholecystectomy with diversion of the biliary-pancreatic flow is considered the treatment of choice. To describe the surgical treatment employed for a patient with Komi’s type 2 PBM and its long-term results. Laparoscopic common bile duct exploration, intraoperative cholangioscopy, and Roux-en-Y hepatico-jejunostomy were performed. Postoperative evolution was satisfactory. The patient was discharge 72 hours after the surgery. There was no associated morbidity. At 62-month follow-up, clinical examination, laboratory tests, and imaging studies confirmed an adequate patency of bilio-enteric anastomosis. The surgical approach employed was effective and safe, with satisfactory long-term results.
During embryological development of the pancreas and bile ducts, alterations can occur, leading to variations in anatomy. Pancreaticobiliary maljunction (PBM) is a congenital malformation of unclear etiology, characterized by the presence of a common bile-pancreatic duct that exceeds 15 mm in length. In this malformation, the biliopancreatic junction is located outside the duodenal wall, causing the Oddi sphincter to lose control over excretion of biliopancreatic secretions, leading to pancreatobiliary and/or biliopancreatic reflux [1].
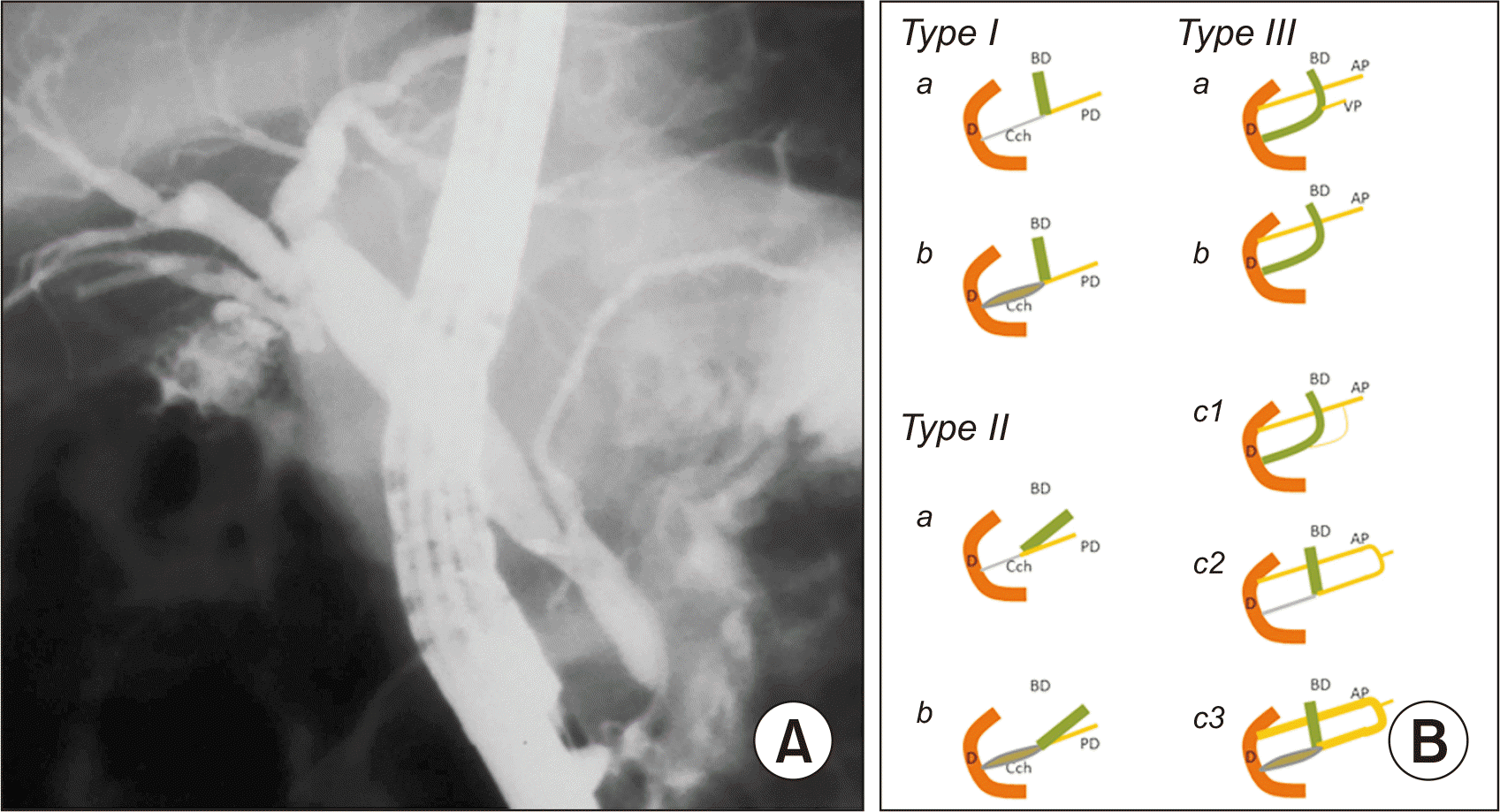
Komi et al. [2] have categorized PBM anomalies into three types based on the angle of confluence of biliary and pancreatic ducts (straight or acute), the dilation or absence of the common duct, and other variations similar to those described by Warshaw et al. [3].
Chronic pancreatobiliary reflux is associated with the development of gallstones, cholangitis, acute pancreatitis, and biliary tract neoplasms. Cholecystectomy with diversion of biliary and pancreatic flow is considered the treatment of choice for PBM. It has a prophylactic intent. We describe the surgical treatment used for a patient with Komi type 2 PBM and its long-term results.
A 19-year-old female patient presented with recurrent episodes of “mild acute pancreatitis” since the age of 5. She underwent endoscopic exploration of the bile ducts, sphincterotomy, and extraction of biliary stones, leading to the diagnosis of Komi type 2 pancreaticobiliary junction anomaly (Fig. 1). Over the next 10 years, the patient experienced three self-limiting episodes of mild acute pancreatitis, prompting laparoscopic cholecystectomy. Following a new episode of mild acute pancreatitis, she was referred to our center.
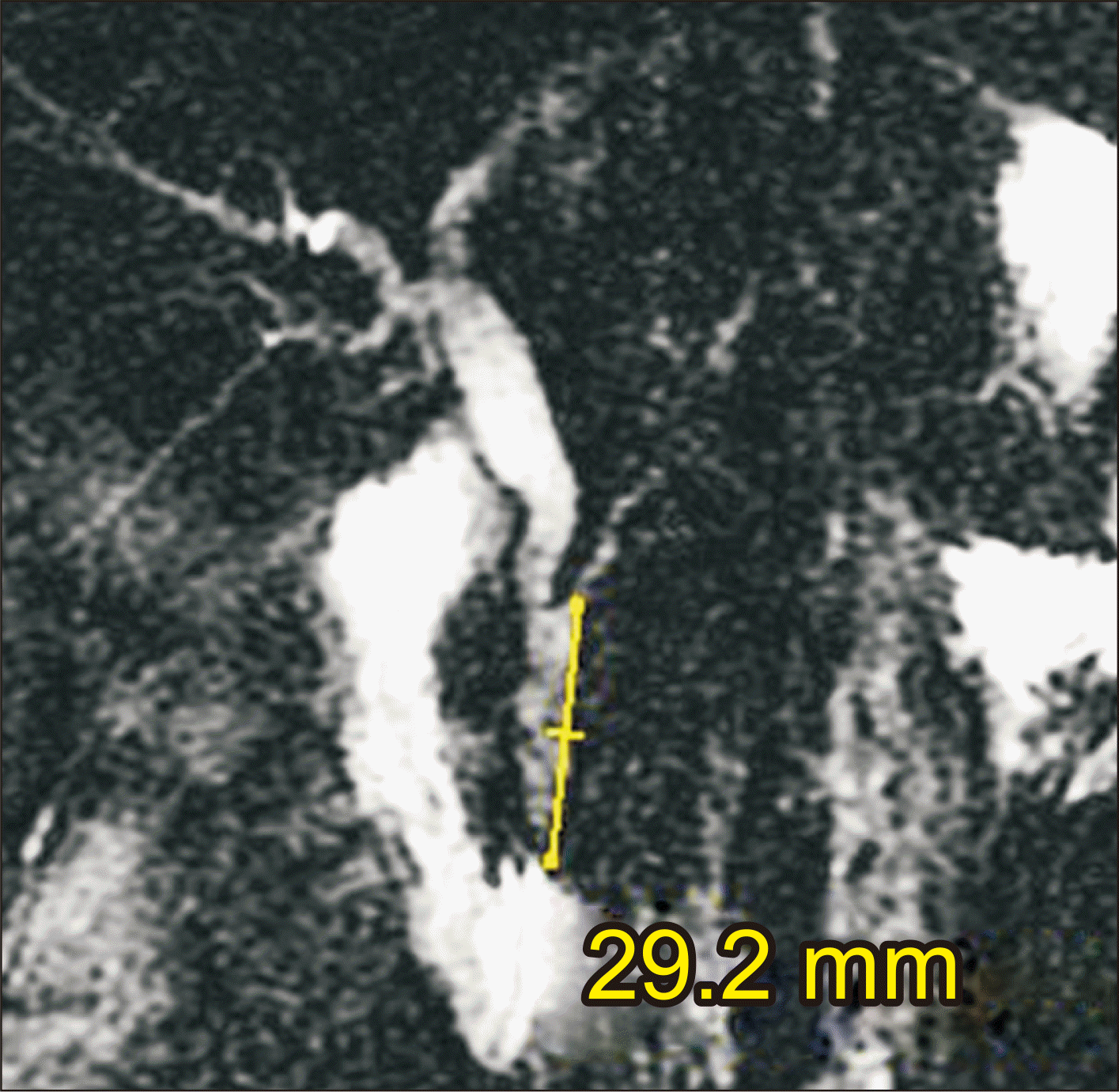
After a thorough diagnostic protocol, including clinical evaluation, laboratory tests, and magnetic resonance cholangiopancreatography (Fig. 2), a plan was formulated and laparoscopic exploration of the bile ducts, transductal flexible choledochoscopy, biliopancreatic brushing, and Roux-en-Y hepaticojejunostomy (RYHJ) were performed (Supplementary Video 1). The postoperative course was satisfactory. The patient was discharged after 72 hours (Fig. 3).
At 62 months of follow-up, the patient remained asymptomatic, with ultrasound findings and pancreaticobiliary function within normal limits The study was approved by the ethical committee of the Dr. Luis Razetti University Hospital. Written informed consent was obtained from all participants.
The dorsal pancreatic primordium emerges from the dorsal side of the duodenum toward the end of the fourth week of fetal development. Beginning at the start of the fifth week, the ventral primordium emerges from the hepatic diverticulum’s lateral wall or caudal side. The dorsal and ventral pancreatic buds fuse and come into contact during the sixth and seventh week due to rotation of the stomach and duodenum and rapid expansion of the left side of the duodenum. The distal section of the dorsal pancreatic duct and the ventral pancreatic duct combine to form the Wirsung duct [4].
However, at the end of the fourth week, a bud on the side of the hepatic diverticulum reveals the primordium of the gallbladder and cystic duct. The hepatic diverticulum’s proximal section elongates during the fifth week, transforming the future duct system into a solid cord of cells. Beginning in the sixth week, the common duct lumen gradually reestablishes itself distally [4].
An abnormality in the embryology known as the PBM occurs when the bile duct connects to the pancreatic duct in the ventral pancreas. This results in an abnormally long common duct that is situated outside the muscle layer of the duodenum. If the part of the bile duct that joins the pancreatic duct does not start the process of recanalization, the pancreatic duct becomes the common duct and only the diameter of the latter is preserved. When this section is recanalized the pancreatic and biliary ducts combine to form the common duct, which has a greater diameter as a result [4]. Therefore, PBM can occur with or without congenital dilatation of the bile duct [5,6].
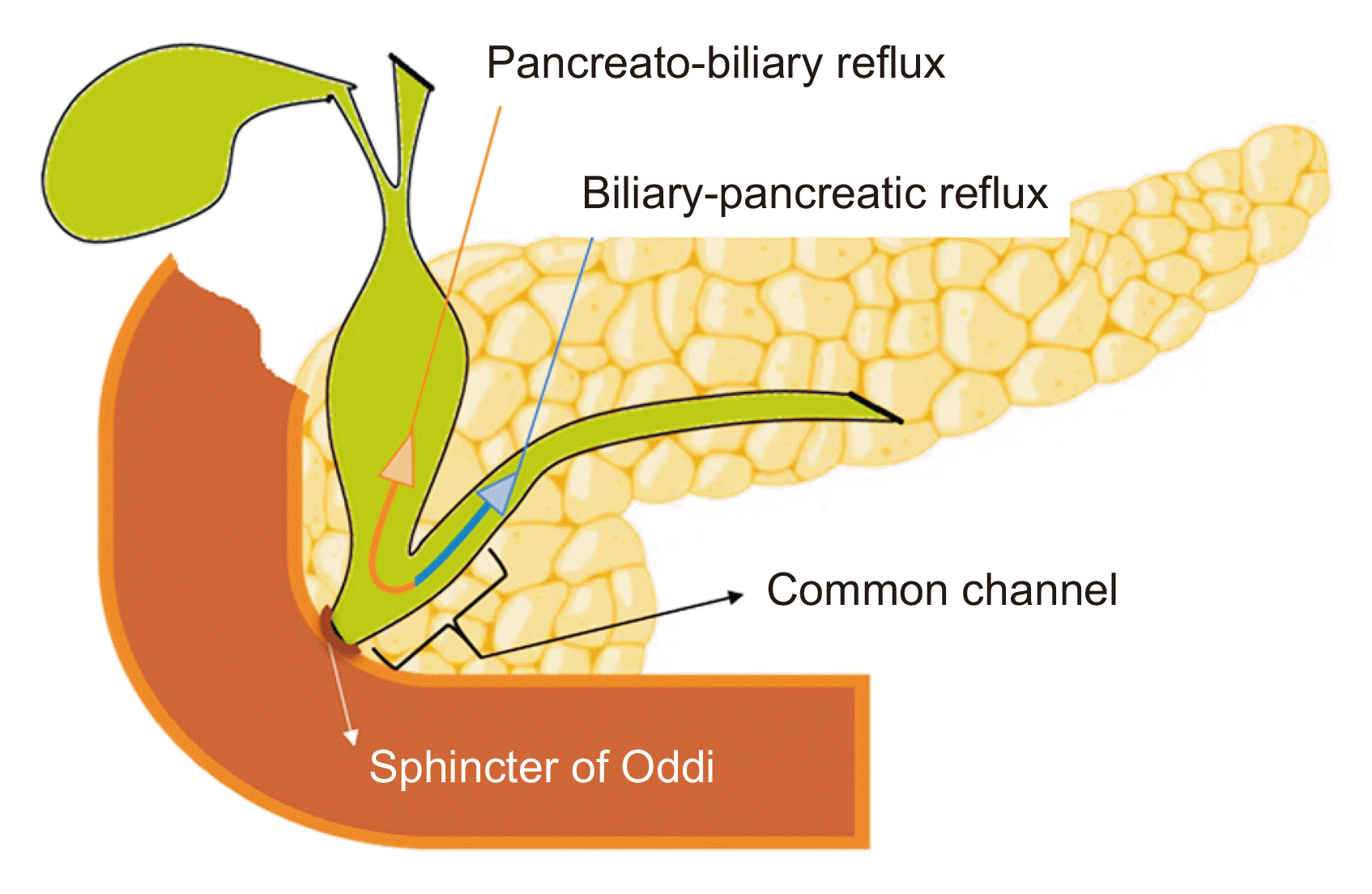
The pancreatic and biliary ducts’ distal end is home to the Oddi sphincter, which controls the flow of bile and pancreatic juice. However, the sphincter is farther away from the point where the pancreatic and biliary channels converge in PBM [7]. Bidirectional regurgitation of fluids may result from this, as illustrated in Fig. 4 [8,9].
The biliary duct receives more of the pancreatic juice reflux because the hydrostatic pressure there is typically higher than that in the pancreatic duct. Because of this, bile typically has an extremely high pancreatic enzyme level (≥ 10,000 UI/L), which is linked to the growth of neoplasms in the biliary network and chronic inflammatory disorders [10].
Due to increased cellular proliferation with random K-ras mutations and, irritation from pancreatic juices, the gallbladder mucosa of patients with PBM should be considered as premalignant [10].
A nationwide study in Japan reported that 21.6% and 42.4% of adult individuals diagnosed with PBM (with and without bile duct dilation, respectively), experienced the development of biliary tract cancers. In addition, simultaneous cancers affecting both the gallbladder and bile duct were identified in 4% of cases involving biliary tract cancers in adults [11]. The incidence of gallbladder cancer can fluctuate, with some series reporting rates as high as 68% [9]. This variability is influenced by the type of PBM, with the non-dilated type showing a higher incidence, as indicated by Kamisawa and Honda [9] in their 2019 series of 145 patients.
Prior to the beginning of malignant alterations, prompt preventative surgical therapy is advised upon diagnosis of PBM. Known as “flow diversion surgery,” cholecystectomy with extrahepatic bile duct resection is a recognized standard surgical therapy approach for these patients. The question of whether patients with PBM who do not have biliary dilatation should have preventive excision of the extrahepatic bile duct remains debatable [12].
The use of a laparoscopic approach in addressing PBM, as illustrated in the presented case, presents advantages when compared to open surgery. For instance, Diao et al. [13] in 2011 examined 218 pediatric patients with a specific PBM subtype and, compared outcomes of laparoscopic RYHJ to open surgery. They found that laparoscopy group demonstrated significantly reduced operative blood loss, shorter postoperative hospital stays, quicker resumption of feeding, and shorter drainage duration than the open surgery group. Moreover, only two patients in the laparoscopy group developed bile leaks, a notably lower number than the 11 cases in the open surgery group. The overall incidence of morbidities in the laparoscopy group was significantly lower than that in the open surgery group [13]. Similar positive outcomes with the laparoscopic approach have also been documented in and adult population with PBM [14].
RYHJ, the most commonly utilized procedure, can avoids postoperative gastritis brought on by biliary reflux [15]. Nevertheless, stenosis of the biliary-enteric anastomosis might be linked to delayed onset of acute cholangitis in a limited number of cases [16].
Urushihara et al. [17] in 2017 reported 317 cases of children with PBM in a hospital in Japan, of which 97.8% underwent RYHJ. The median follow-up period was 7.3 years, with good results, as only 1.3% of patients experienced late pancreatic disorders, mostly recurrent pancreatitis [17]. On the other hand, Miyake et al. [18] in 2022 reported results of 7 patients with non-dilated biliary pathways PBM who underwent RYHJ. In all cases, symptoms disappeared after definitive surgery. After an average follow-up of 18 years, only one patient had an intrahepatic calculus due to anastomotic stenosis [18]. Shimotakahara et al. [16] have describe an incidence of 3.5% of acute cholangitis in 28 patients with PBM treated with RYHJ.
In the case described in the present report, no associated morbidity was observed with a follow-up of 62 months.
Based on literature review, the majority of PBM patients are young females whose symptoms begin in childhood [17,18], including recurrent episodes of acute pancreatitis, which can occur in up to 60% of cases in some series [19]. Similarly, the case reported in this article was a young female. Generally, the indication for surgical treatment is broad and often preceded by overuse of both diagnostic procedures and therapeutic interventions.
Similarly, we consider an appropriate postoperative follow-up protocol essential, not only due to the risk of infectious complications, but also because of the potential development of neoplasms arising from the remaining duct responsible for the excretion of pancreatic secretions [15].
Supplementary data related to this article can be found at https://doi.org/10.14701/ahbps.24-063.
ACKNOWLEDGEMENTS
This manuscript was presented at the IXX World Congress of Endoscopic Surgery in Cartagena, Colombia in November 2, 2023.
References
1. Komi N, Tamura T, Miyoshi Y, Kunitomo K, Udaka H, Takehara H. 1984; Nationwide survey of cases of choledochal cyst. Analysis of coexistent anomalies, complications and surgical treatment in 645 cases. Surg Gastroenterol. 3:69–73.
2. Komi N, Takehara H, Kunitomo K, Miyoshi Y, Yagi T. 1992; Does the type of anomalous arrangement of pancreaticobiliary ducts influence the surgery and prognosis of choledochal cyst? J Pediatr Surg. 27:728–731. DOI: 10.1016/S0022-3468(05)80102-2. PMID: 1306647.
3. Warshaw AL, Simeone JF, Schapiro RH, Flavin-Warshaw B. 1990; Evaluation and treatment of the dominant dorsal duct syndrome (pancreas divisum redefined). Am J Surg. 159:59–64. discussion 64–66. DOI: 10.1016/S0002-9610(05)80607-5. PMID: 2403764.
4. Ando H, Kaneko K, Ito F, Seo T, Harada T, Watanabe Y. 1999; Embryogenesis of pancreaticobiliary maljunction inferred from development of duodenal atresia. J Hepatobiliary Pancreat Surg. 6:50–54. DOI: 10.1007/s005340050083. PMID: 10436237.
5. Renard Y, Sommacale D, Avisse C, Palot JP, Kianmanesh R. 2014; [Anatomía quirúrgica de las vías biliares extrahepáticas y de la unión biliopancreática]. EMC Téc Quir Apar Dig. 30:1–21. Spanish. DOI: 10.1016/S1282-9129(14)69151-0.
6. Kamisawa T, Kaneko K, Itoi T, Ando H. 2017; Pancreaticobiliary maljunction and congenital biliary dilatation. Lancet Gastroenterol Hepatol. 2:610–618. DOI: 10.1016/S2468-1253(17)30002-X. PMID: 28691687.
7. Kamisawa T, Egawa N, Nakajima H, Tsuruta K, Okamoto A, Matsukawa M. 2005; Origin of the long common channel based on pancreatographic findings in pancreaticobiliary maljunction. Dig Liver Dis. 37:363–367. DOI: 10.1016/j.dld.2004.11.007. PMID: 15843087.
8. Kwak SW, Kim S, Lee JW, Lee NK, Kim CW, Yi MS, et al. 2009; Evaluation of unusual causes of pancreatitis: role of cross-sectional imaging. Eur J Radiol. 71:296–312. DOI: 10.1016/j.ejrad.2008.04.006. PMID: 18538971.
9. Kamisawa T, Honda G. 2019; Pancreaticobiliary maljunction: markedly high risk for biliary cancer. Digestion. 99:123–125. DOI: 10.1159/000490816. PMID: 30179869.
10. Kamisawa T, Takuma K, Anjiki H, Egawa N, Kurata M, Honda G, et al. 2009; Pancreaticobiliary maljunction. Clin Gastroenterol Hepatol. 7(11 Suppl):S84–S88. DOI: 10.1016/j.cgh.2009.08.024. PMID: 19896105.
11. Morine Y, Shimada M, Takamatsu H, Araida T, Endo I, Kubota M, et al. 2013; Clinical features of pancreaticobiliary maljunction: update analysis of 2nd Japan-nationwide survey. J Hepatobiliary Pancreat Sci. 20:472–480. DOI: 10.1007/s00534-013-0606-2. PMID: 23579999.
12. Kamisawa T, Ando H, Shimada M, Hamada Y, Itoi T, Takayashiki T, et al. 2014; Recent advances and problems in the management of pancreaticobiliary maljunction: feedback from the guidelines committee. J Hepatobiliary Pancreat Sci. 21:87–92. DOI: 10.1002/jhbp.8. PMID: 23798483.
13. Diao M, Li L, Cheng W. 2011; Laparoscopic versus open Roux-en-Y hepatojejunostomy for children with choledochal cysts: intermediate-term follow-up results. Surg Endosc. 25:1567–1573. DOI: 10.1007/s00464-010-1435-x. PMID: 21052722.
14. Liu Y, Yao X, Li S, Liu W, Liu L, Liu J. 2014; Comparison of therapeutic effects of laparoscopic and open operation for congenital choledochal cysts in adults. Gastroenterol Res Pract. 2014:670260. DOI: 10.1155/2014/670260. PMID: 24719612. PMCID: PMC3955616.
15. Ono A, Arizono S, Isoda H, Togashi K. 2020; Imaging of pancreaticobiliary maljunction. Radiographics. 40:378–392. DOI: 10.1148/rg.2020190108. PMID: 31951513.
16. Shimotakahara A, Yamataka A, Yanai T, Kobayashi H, Okazaki T, Lane GJ, et al. 2005; Roux-en-Y hepaticojejunostomy or hepaticoduodenostomy for biliary reconstruction during the surgical treatment of choledochal cyst: which is better? Pediatr Surg Int. 21:5–7. DOI: 10.1007/s00383-004-1252-1. PMID: 15372285.
17. Urushihara N, Hamada Y, Kamisawa T, Fujii H, Koshinaga T, Morotomi Y, et al. 2017; Classification of pancreaticobiliary maljunction and clinical features in children. J Hepatobiliary Pancreat Sci. 24:449–455. DOI: 10.1002/jhbp.485. PMID: 28639336.
18. Miyake H, Fukumoto K, Yamoto M, Nomura A, Yamada S, Kanai R, et al. 2022; Pancreaticobiliary maljunction without biliary dilatation in pediatric patients. Surg Today. 52:207–214. DOI: 10.1007/s00595-021-02322-5. PMID: 34146154.
19. Fujishiro J, Masumoto K, Urita Y, Shinkai T, Gotoh C. 2013; Pancreatic complications in pediatric choledochal cysts. J Pediatr Surg. 48:1897–1902. DOI: 10.1016/j.jpedsurg.2012.12.038. PMID: 24074664.
Fig. 1
Komi type 2 pancreaticobiliary maljunction (PBM). (A) Endoscopic cholangiopancreatography. (B) Komi’s PBM classification scheme. BD, bile duct; PD, pancreatic duct, Cch, common channel; D, duodenum; AP, accessory pancreatic duct; VP, ventral pancreatic duct.




 PDF
PDF Citation
Citation Print
Print




 XML Download
XML Download