Abstract
Nivolumab plus chemotherapy in the first-line setting has demonstrated clinical efficacy in patients with human epidermal growth factor receptor 2–negative advanced or metastatic gastric cancer, and is currently indicated as a standard treatment. Programmed death-ligand 1 (PD-L1) expression is an important biomarker for predicting response to anti–programmed death 1/PD-L1 agents in several solid tumors, including gastric cancer. In the CheckMate-649 trial, significant clinical improvements were observed in patients with PD-L1 combined positive score (CPS) ≥ 5, determined using the 28-8 pharmDx assay. Accordingly, an accurate interpretation of PD-L1 CPS, especially at a cutoff of 5, is important. The CPS method evaluates both immune and tumor cells and provides a comprehensive assessment of PD-L1 expression in the tumor microenvironment of gastric cancer. However, CPS evaluation has several limitations, one of which is poor interobserver concordance among pathologists. Despite these limitations, clinical indications relying on PD-L1 CPS are increasing. In response, Korean gastrointestinal pathologists held a consensus meeting for the interpretation of PD-L1 CPS in gastric cancer. Eleven pathologists reviewed 20 PD-L1 slides with a CPS cutoff close to 5, stained with the 28-8 pharmDx assay, and determined the consensus scores. The issues observed in discrepant cases were discussed. In this review, we present cases of gastric cancer with consensus PD-L1 CPS. In addition, we briefly touch upon current practices and clinical issues associated with assays used for the assessment of PD-L1 expression in gastric cancer.
The interaction between programmed death 1 (PD-1) protein and programmed death-ligand 1 (PD-L1) is a major mechanism of immune modulation that allows for T-cell inactivation and tumor immune evasion [1]. Blocking the PD-1/PD-L1 pathway is a standard therapeutic strategy for the treatment of various solid tumors, including gastric cancer (GC) [2]. Several clinical trials of anti–PD-1/PD-L1 agents have shown clinical efficacy in patients with advanced or metastatic GC [3-10]. Among these, the CheckMate-649 trial demonstrated the efficacy of nivolumab (a PD-1 binding immune checkpoint inhibitor) in combination with chemotherapy as a first-line treatment for human epidermal growth factor receptor 2 (HER2)–negative advanced or metastatic GC [3]. Currently, nivolumab plus chemotherapy is the standard first-line treatment for advanced or metastatic GC [3,9,11].
PD-L1 expression is a biomarker used to predict the therapeutic response to anti–PD-1/PD-L1 agents across several solid tumors, including GC [12]. In GC, PD-L1 expression in both immune and tumor cells provides a comprehensive assessment of PD-L1 expression in the tumor microenvironment, thereby better predicting the therapeutic response to anti–PD-1/PD-L1 agents [13,14]. The combined positive score (CPS) is used to evaluate PD-L1 expression in both immune and tumor cells [13,15,16]. In the CheckMate-649 trial, nivolumab plus chemotherapy significantly improved the overall survival and progression-free survival of patients with PD-L1 CPS ≥5 [3]. Additional results showed significant improvement in overall survival and progression-free survival in patients with PD-L1 CPS ≥1 and all randomly assigned patients [3]. However, approval and recommendations for nivolumab differ according to the country or approval agency [11]. In Korea, nivolumab reimbursement for GC treatment is restricted to patients with PD-L1 CPS ≥5. Therefore, accurate interpretation of PD-L1 CPS, especially at a cutoff of 5, is crucial to identify the patients with GC that are eligible for nivolumab treatment.
Several issues need addressing in PD-L1 tests for GC. First, inter-assay variability needs to be resolved, as various assays are confusing and not all assays are available for laboratories and patients [17]. Efforts are underway to harmonize PD-L1 assays in GC, with one study demonstrating high comparability between the 22C3 and 28-8 pharmDx assays at various CPS cutoffs [17]. Secondly, a more challenging aspect involves interobserver variability among pathologists [18]. Recent studies reported high interobserver variability among pathologists for CPS in GCs [18,19], raising questions on PD-L1 CPS as a biomarker for GC. Nevertheless, given the increasing reliance on specific PD-L1 CPS cutoffs in clinical indications, efforts are necessary to address and overcome this issue. In this context, Korean gastrointestinal pathologists held a consensus meeting to evaluate the PD-L1 CPS in patients with GC. Eleven pathologists reviewed 20 PD-L1 slides near the CPS cutoff 5 and aimed to determine consensus scores. In this article, we briefly introduce the current practice of PD-L1 testing and studies on the interobserver variability of PD-L1 CPS in GC and present cases from the consensus meetings.
Table 1 summarizes the assays used for PD-L1 testing in GC. Currently, three standardized PD-L1 immunohistochemical assays (22C3 pharmDx, 28–8 pharmDx, and SP263) are used to specifically predict responses to pembrolizumab, nivolumab, and tislelizumab.
In GC, PD-L1 expression is scored using the CPS scoring system, which is calculated as the number of PD-L1–stained cells (tumor cells, lymphocytes, and macrophages) divided by the total number of viable tumor cells multiplied by 100 [20]. A tumor cell is PD-L1 positive if the cell membrane is partially or completely stained, irrespective of staining intensity [20]. PD-L1–stained immune cells include only mononuclear inflammatory cells (lymphocytes or macrophages) within tumor nests and adjacent stroma and show membrane and/or cytoplasmic staining [20]. Other stromal cells such as fibroblasts, neutrophils, and plasma cells are excluded [20]. If the calculation result exceeds 100, it is presented as a maximum score of 100 [20]. If the PDL1 staining is heterogeneous, the final CPS is estimated by calculating the CPS results for each area within the entire tumor [20].
Currently, two different PD-L1 assays are approved for clinical evaluation. For each assay, different CPS cutoff values are used. The PD-L1 22C3 pharmDx assay uses CPS ≥1 for PD-L1 positivity [8,10] and the 28-8 pharmDx assay uses CPS ≥5 [3]. For tislelizumab, tumor area positivity score, stained with SP263, is used [7]. Reports should specify the assay type and appropriate cutoff value used for the interpretation of PD-L1 positivity [15].
Table 2 summarizes the studies that evaluated the interobserver concordance of PD-L1 CPS in GC. Two previous studies reported excellent interobserver agreement for PD-L1 evaluation in GC [13,21]. In a study by Kulangara et al. [13], three pathologists evaluated 68 22C3 pharmDx-stained PD-L1 slides (sample type not disclosed); an overall percentage agreement (OPA) of 96.6% at a CPS cutoff of 1 was observed. Nuti et al. [21] evaluated the interobserver concordance among 120 pathologists in their interpretation of 20 22C3 pharmDx-stained surgical samples; an OPA of 90.6% at the CPS cutoff of 1 was observed, with a Fleiss kappa value of 0.828.
In contrast, Park et al. [22] reported poor interobserver concordance among five pathologists who evaluated 55 tissue microarray samples using the 22C3 pharmDx and SP263 assays. The intraclass correlation coefficient (ICC) was 0.387, and the Fleiss kappa value was 0.389 at a 22C3 pharmDx CPS cutoff of 1 [22]. In addition, two recent studies reported poor interobserver agreement regarding the CPS in GC [18,19]. Fernandez et al. [18] evaluated the concordance for PD-L1 CPS among 14 pathologists from 13 institutions in the United States, based on their assessment of 22C3 pharmDx-stained 112 biopsy samples. At a CPS cutoff of 1, the reported OPA and ICC were 31.48% and 0.484 [18], respectively. Higher CPS cutoffs (10 or 20) showed improved agreement than a CPS cutoff 1 [18]. Robert et al. [19] evaluated the interobserver agreement among 12 international pathologists who assessed 100 biopsy samples stained with PD-L1 28-8 and 22C3 pharmDx; interobserver CPS agreement was fair for both pre- (ICC range, 0.45 to 0.55) and post-training (ICC range, 0.56 to 0.57) for both assays [19]. In addition, Robert et al. [19] evaluated CPS interobserver agreement for 35 biopsy fragments. Poor or fair agreement was observed regarding the number of PD-L1–positive immune cells (ICC, 0.19), number of PD-L1–positive tumor cells (ICC, 0.54), total number of viable tumor cells (ICC, 0.09), and the calculated CPS (ICC, 0.14), whereas calculated tumor cell scores showed excellent agreement (ICC, 0.82) [19]. These findings are in agreement with the results for other solid tumors, wherein the interobserver concordance for immune cells was much lower than that for tumor cells [23-25].
Twenty GC samples stained using the PD-L1 28-8 pharmDx assay, in routine clinical pathology practice, with CPS scores ranging from 3 to 7, were collected from seven institutes in Korea; six were resection specimens and 14 were biopsy specimens. The information about samples is summarized in Table 3. PD-L1 slides were scanned using each institute’s scanner. Twelve specimens were scanned using a Philips Digital Pathology Slide SG300 scanner (Amsterdam, Netherlands), four with an Aperio GT 450 DX scanner (Vista, CA, USA), three with an Aperio AT2 scanner (Vista, CA, USA) and one with a 3DHISTECH PANNORAMIC 250 Flash III scanner (Budapest, Hungary). All the participating pathologists specialized in gastrointestinal pathology (the average number of years of experience, 15; range, 6 to 30 years) and routinely assessed PD-L1 28-8 pharmDx in GC in clinical practice. Nine work in university hospitals, and two work in private laboratories.
On the day of the meeting (17 Oct 2023), 11 pathologists evaluated 20 PD-L1 scanned slides using each image viewer, without clinical information, for 30 min and submitted the CPS results. Some pathologists did not complete the survey for all the cases. Subsequently, all the pathologists reviewed the scanned PD-L1 image slides, discussed each case, and determined consensus CPS scores. For cases with poor interobserver agreement (more than 20% of pathologists disagreed with the CPS cutoff of 5), cases were discussed in detail, and a consensus CPS score was determined. Despite discussion, a CPS consensus was not reached for Case 3 (Supplementary Fig. S1).
Nine cases were concluded to have consensus CPS of <5. Of these, there was good interobserver agreement for two cases (cases 1 and 15) (Figs. 1, 2). The remaining cases are presented along with a detailed discussion.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 0, n=3; CPS 2, n=2; CPS 3, n=2; CPS 5, n=3). The biopsy specimen consisted of three pieces of signet ring cell carcinoma with normal components. One piece was PD-L1 negative, the second exhibited stained area scores less than 5, and the third one exhibited stained area scores of >5. When the PD-L1 staining revealed a heterogeneous pattern, the final CPS was estimated for each area [20]. By averaging the scores for each region, a consensus CPS score of 2 was determined.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 1, n=1; CPS 2, n=2; CPS 3, n=1; CPS 5, n=4; CPS 8, n=1; CPS 10, n=1). This biopsy specimen consisted of two pieces of moderately differentiated adenocarcinoma tissue without a normal component. One fragment had no PD-L1–positive cells, while the other showed stained area scores of CPS >5. A consensus CPS score of 3 was determined by averaging the scores for each region.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 0, n=2; CPS 3, n=2; CPS 4, n=1; CPS 5, n=1; CPS 7, n=1; CPS 9, n=1). The biopsy specimen comprised four fragments of poorly cohesive carcinoma with normal components. While most pieces were PD-L1–negative, one piece revealed PD-L1–positive immune cells. On an average, a consensus score of 2 was determined.
The biopsy specimen contained a poorly differentiated adenocarcinoma with infiltrating immune cells within and adjacent to the tumor. Most pathologists submitted CPS results of <5 (CPS 0, n=1; CPS 1, n=2; CPS 2, n=1; CPS 3, n=1; CPS 5, n=2); following a detailed discussion, a CPS consensus of 2 was reached. Many pathologists at the meeting mentioned that nonspecific 28-8 pharmDx staining complicates CPS interpretation. Although the tumor cells showed weak staining at a low magnification, only cytoplasmic staining was observed at a high magnification. Tumor cells with cytoplasmic staining only need to be excluded from CPS calculations [20]. In this case, only PD-L1–positive lymphocytes were included in the CPS numerator. Nonspecific cytoplasmic staining in tumor cells is not usually observed in the 22C3 pharmDx assay, and caution is required for interpretation of 28-8 pharmDx assay [17].
This case revealed poor interobserver agreement at a CPS cutoff of 5 (CPS 0, n=1; CPS 3, n=2; CPS, n=4). Six of seven fragments of the biopsy specimen contained signet ring cell carcinoma with the extracellular mucin. Some tumor cells showed partial membrane staining, and lymphocytes showed cytoplasmic staining. Nonspecific staining in the empty space caused by mucin, stained stromal cells, normal glandular cells stained by edge artifacts, and neutrophils were excluded from the CPS calculation. In this case, defining the tumor-associated area was a confounding factor for interpretation, and it was difficult to determine whether the immune cells were within the tumor or distant from the tumor. In general, a 20× field of view rule is applied to define tumor-associated areas [20]. Immune cells are considered tumor-associated and included in the CPS calculation if the cells are present within the tumor nests or adjacent stroma or both within a 20× magnification field of view [20]. At the same time, non-tumor gastritis areas need to be excluded [20]. However, endoscopic specimens inevitably contain non-tumor gastritis areas wherein resident inflammation is common [19]. In this case, when the 20× field of view rule was applied, the surface area presumed to be non-tumor gastritis was included. This confused the pathologists, as there are currently no guidelines for this situation. Considering that mucinous tumors are generally less immunogenic, and immune cells in the specimen are considered mostly gastritis, pathologists at the meeting agreed to a CPS of 3. A correlation with the treatment response would be helpful; however, the patient had not yet received immunotherapy.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 2, n=1; CPS 3, n=1; CPS 4, n=1; CPS 5, n=2; CPS 10, n=1; CPS 20, n=1). The tumor histology was a papillary adenocarcinoma with empty spaces caused by the papillary architecture. At a low magnification, some stained areas were observed. However, at a high magnification, most of the stained areas were revealed as background staining in empty spaces and stromal cells. After excluding nonspecific background staining, the number of positive immune cells appeared to be small. After discussion, the pathologists agreed on a CPS of 2.
The resected specimen showed a poorly cohesive carcinoma with abundant lymphatic tumor emboli in the mucosa and submucosa. The submitted results were CPS 0 (n=1) and CPS 5 (n=2). Tumor cells show only cytoplasmic staining, which must be excluded from the CPS calculation [20]. Normal glandular cells exhibited nonspecific staining. The pathologists agreed on a CPS of 0.
Ten cases were concluded as having consensus CPS ≥5. Among these, good interobserver agreement was noted for five cases (cases 5, 10, 12, 18, and 19) (Figs. 10, 11, 12, 13, and 14, respectively). The remaining cases are presented along with a detailed discussion.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 0, n=3; CPS 1, n=1; CPS 2, n=1; CPS 5, n=2; CPS 8, n =1; CPS 10, n=1; CPS 20, n=1). This resected specimen consisted of poorly cohesive carcinoma with some infiltrating lymphocytes. Owing to the large tumor area, CPS scoring was difficult. Compared to those in the usual diffuse-type GC, relatively abundant tumor-associated lymphocytes were observed in the specimen, and these immune cells were focally PD-L1 positive. Based on the findings, a consensus CPS of 5 was determined.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 1, n=1; CPS 2, n=2; CPS 3, n=1; CPS 5, n=1; CPS 7, n=1). This tumor consisted of four papillary adenocarcinoma fragments and two normal mucosal fragments. Nonspecific staining was observed in the empty space and immune cells in the non-tumor gastritis area. However, even after the exclusion of nonspecific staining, PD-L1–positive immune cells remained around the tumor. A consensus CPS of 5 was determined.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 2, n=1; CPS 3, n=2; CPS 4, n=2; CPS 5, n=3; CPS 6, n=1; CPS 10, n=1). This resection specimen of advanced GC exhibited a small PD-L1–stained area at the front of the invasive tumor. Although the PD-L1–positive area was small, some of the pathologists mentioned that there were abundant immune cells, and PD-L1–positive immune cells at the invasive front were likely the result of a direct interaction between tumor cell antigens and neighboring immune cells. The final consensus was a CPS of 5. The interpretation manual provides guidelines for the calculation of CPS in a small PD-L1–stained area [20]. First, the percentage of the PD-L1–stained area in the entire tumor was evaluated [20]. Next, the CPS of the area was evaluated via staining [20]. Finally, the CPS was calculated considering both the results [20]. By applying this rule, 10% of the entire area was deemed PD-L1 positive, and the CPS was 50. The CPS of the entire tumor area was 10%×50=CPS 5.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 1, n=2; CPS 2, n=1; CPS 3, n=1; CPS 5, n=2; CPS 8, n=1; CPS 10, n=1). The biopsy specimen consisted of tumor cells and abundant tumor-infiltrating lymphocytes without a normal component. A few tumor and immune cells with strong intensity were observed in a small area. Furthermore, a considerable number of weakly stained cells was observed. The staining intensity can vary significantly in a single case [26]. Distinguishing positive staining with weak intensity from nonspecific staining was challenging. Despite background staining, immune cells with membrane and/or cytoplasmic staining at any intensity should be included in CPS calculation. Therefore, by counting the immune cells (not all but some) with cytoplasmic staining, this case was classified as CPS 5.
At a CPS cutoff of 5, there was poor interobserver agreement (CPS 2, n=1; CPS 3, n=1; CPS 5, n=2; CPS 6, n=1; CPS 7, n=1; CPS 8, n=1). Strongly stained areas were observed on mucosal surfaces. The ulcerative area of the mucosa was excluded from CPS calculation. However, the stained area (Fig. 19A box, B) in the mucosa contained tumors admixed with inflammatory cells and focal necrosis, which needed to be counted. In addition, positive intra-glandular macrophages, lymphocytes, and tumor cells were observed at the invasive front of the tumor. If PD-L1 staining shows heterogeneous results, the final CPS must be estimated by calculating the CPS result for each area [20]. Finally, a CPS of 5 was determined for this case.
Poor interobserver agreement among pathologists’ CPS in PD-L1 assessment is a known challenge for the use of PD-L1 as a biomarker in GC. In particular, CPS scoring in biopsy specimens stained with 28-8 pharmDx can be more challenging due to nonspecific staining and various artifacts. To address this, Korean gastrointestinal pathologists had a consensus meeting of PD-L1 in GC to discuss cases near CPS cutoff 5. Our consensus meeting also revealed high variability even among expert pathologists in some cases. In this review, we described cases with consensus CPS including practical considerations and potential pitfalls, which may serve as reference for pathologists.
Supplementary Information
The Data Supplement is available with this article at https://doi.org/10.4132/jptm.2024.03.15.
Notes
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Author Contributions
Conceptualization: SA, HSL, KMK. Participation of consensus meeting: all authors. Supervision: HSL, KMK. Resources: YSP, YK, SA, KL, GYK, MK, HJO. Writing—original draft preparation: SA, HSL. Writing—review & editing: all authors. Approval of final manuscript: all authors.
References
2. Nakamura Y, Kawazoe A, Lordick F, Janjigian YY, Shitara K. Biomarker-targeted therapies for advanced-stage gastric and gastrooesophageal junction cancers: an emerging paradigm. Nat Rev Clin Oncol. 2021; 18:473–87.

3. Janjigian YY, Shitara K, Moehler M, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021; 398:27–40.

4. Kang YK, Chen LT, Ryu MH, et al. Nivolumab plus chemotherapy versus placebo plus chemotherapy in patients with HER2-negative, untreated, unresectable advanced or recurrent gastric or gastro-oesophageal junction cancer (ATTRACTION-4): a randomised, multicentre, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2022; 23:234–47.

5. Rha SY, Oh DY, Yanez P, et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for HER2-negative advanced gastric cancer (KEYNOTE-859): a multicentre, randomised, doubleblind, phase 3 trial. Lancet Oncol. 2023; 24:1181–95.
6. Shitara K, Van Cutsem E, Bang YJ, et al. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: the KEYNOTE-062 phase 3 randomized clinical trial. JAMA Oncol. 2020; 6:1571–80.

7. Xu J, Kato K, Raymond E, et al. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first-line treatment for advanced or metastatic oesophageal squamous cell carcinoma (RATIONALE-306): a global, randomised, placebo-controlled, phase 3 study. Lancet Oncol. 2023; 24:483–95.

8. Janjigian YY, Kawazoe A, Bai Y, et al. Pembrolizumab plus trastuzumab and chemotherapy for HER2-positive gastric or gastro-oesophageal junction adenocarcinoma: interim analyses from the phase 3 KEYNOTE-811 randomised placebo-controlled trial. Lancet. 2023; 402:2197–208.

9. Shitara K, Ajani JA, Moehler M, et al. Nivolumab plus chemotherapy or ipilimumab in gastro-oesophageal cancer. Nature. 2022; 603:942–8.

10. Fuchs CS, Doi T, Jang RW, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial. JAMA Oncol. 2018; 4:e180013.
11. Kubota Y, Aoki Y, Kawazoe A, Shitara K. Role of nivolumab in the management of first-Line unresectable advanced or recurrent gastric cancer in combination with chemotherapy: lessons from the Japanese experience. Cancer Manag Res. 2022; 14:3083–94.

12. Yi M, Jiao D, Xu H, et al. Biomarkers for predicting efficacy of PD-1/PD-L1 inhibitors. Mol Cancer. 2018; 17:129.

13. Kulangara K, Zhang N, Corigliano E, et al. Clinical utility of the combined positive score for programmed death ligand-1 expression and the approval of pembrolizumab for treatment of gastric cancer. Arch Pathol Lab Med. 2019; 143:330–7.

14. Lei M, Siemers NO, Pandya D, et al. Analyses of PD-L1 and inflammatory gene expression association with efficacy of nivolumab +/- ipilimumab in gastric cancer/gastroesophageal junction cancer. Clin Cancer Res. 2021; 27:3926–35.

15. Park YS, Kook MC, Kim BH, et al. A standardized pathology report for gastric cancer: 2nd edition. J Pathol Transl Med. 2023; 57:1–27.

16. Kim TH, Kim IH, Kang SJ, et al. Korean Practice Guidelines for Gastric Cancer 2022: an evidence-based, multidisciplinary approach. J Gastric Cancer. 2023; 23:3–106.

17. Ahn S, Kim KM. PD-L1 expression in gastric cancer: interchangeability of 22C3 and 28-8 pharmDx assays for responses to immunotherapy. Mod Pathol. 2021; 34:1719–27.

18. Fernandez AI, Robbins CJ, Gaule P, et al. Multi-institutional study of pathologist reading of the programmed cell death ligand-1 combined positive score immunohistochemistry assay for gastric or gastroesophageal junction cancer. Mod Pathol. 2023; 36:100128.

19. Robert ME, Ruschoff J, Jasani B, et al. High interobserver variability among pathologists using combined positive score to evaluate PDL1 expression in gastric, gastroesophageal junction, and esophageal adenocarcinoma. Mod Pathol. 2023; 36:100154.

20. PD-L1 IHC 28-8 pharmDx interpretation manual: gastric adenocarcinoma, gastroesophageal juction (GEJ) adenocarcinoma, and esophageal adenocarcinoma [Internet]. Santa Clara: DAKO Agilent Technologies, 2021 [cited 2024 Feb 22]. Available from: https://www.agilent.com/cs/library/usermanuals/public/29456-d68866-pd-l1-28-8-gastric-interpretation-manual-en-eu.pdf.
21. Nuti S, Zhang Y, Zerrouki N, et al. High interobserver and intraobserver reproducibility among pathologists assessing PD-L1 CPS across multiple indications. Histopathology. 2022; 81:732–41.

22. Park Y, Koh J, Na HY, et al. PD-L1 testing in gastric cancer by the combined positive score of the 22C3 PharmDx and SP263 assay with clinically relevant cut-offs. Cancer Res Treat. 2020; 52:661–70.

23. Tsao MS, Kerr KM, Kockx M, et al. PD-L1 immunohistochemistry comparability study in real-life clinical samples: results of Blueprint Phase 2 Project. J Thorac Oncol. 2018; 13:1302–11.

24. Prince EA, Sanzari JK, Pandya D, Huron D, Edwards R. Analytical concordance of PD-L1 assays utilizing antibodies from FDA-approved diagnostics in advanced cancers: a systematic literature review. JCO Precis Oncol. 2021; 5:953–73.

Fig. 1.
Representative images of case 1 with a consensus combined positive score of 0 and good interobserver agreement at 5× (A) and 20× (B) magnification.
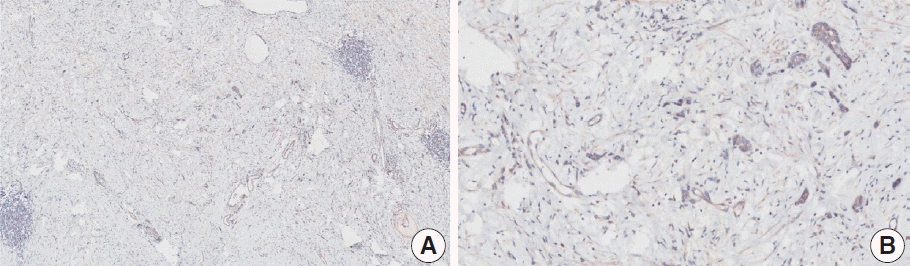
Fig. 2.
Representative images of case 15 with a consensus combined positive score of 2 and good interobserver agreement at 1× (A), 10× (B), 20× (C), and 20× (D) magnification.
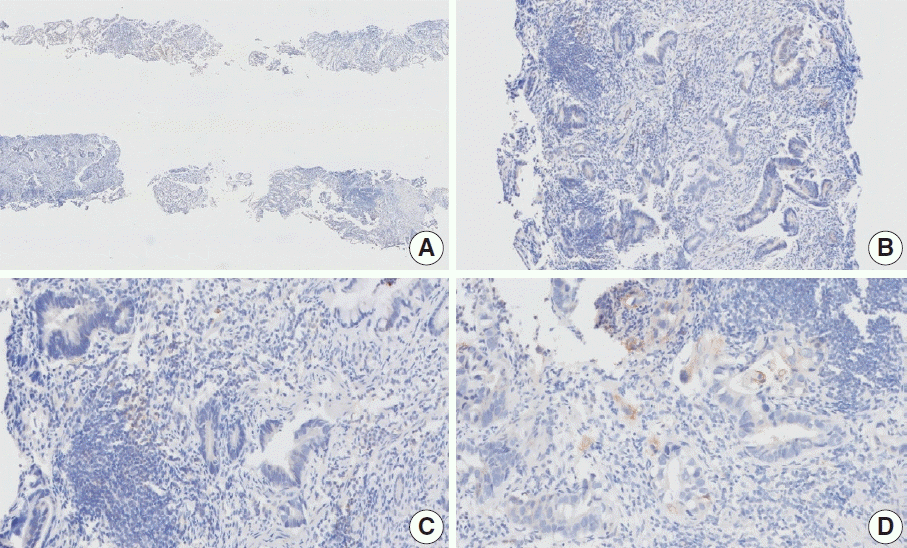
Fig. 3.
Representative images of case 2 with a consensus combined positive score of 2 and poor interobserver agreement at 5× (A), 20× (B), 20× (C), and 40× (D) magnification.
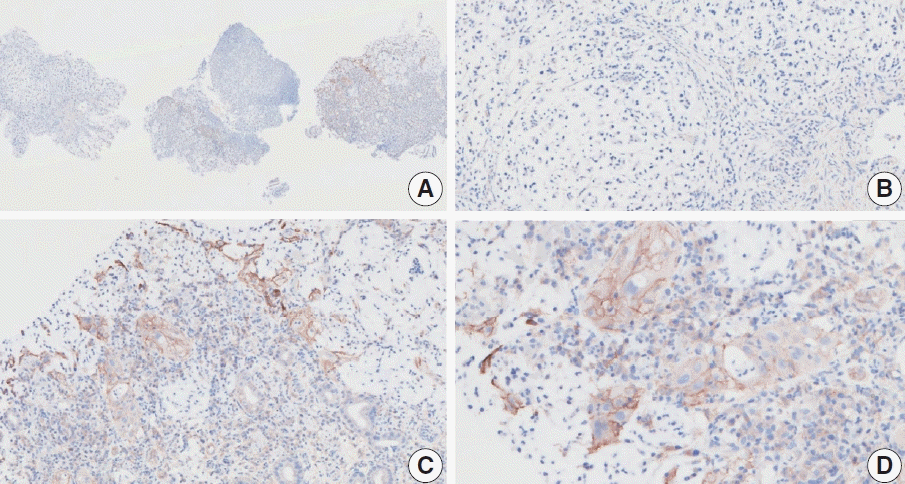
Fig. 4.
Representative images of case 6 with a consensus combined positive score of 3 and poor interobserver agreement at 5× (A), 20× (B), 20× (C), and 40× (D) magnification.
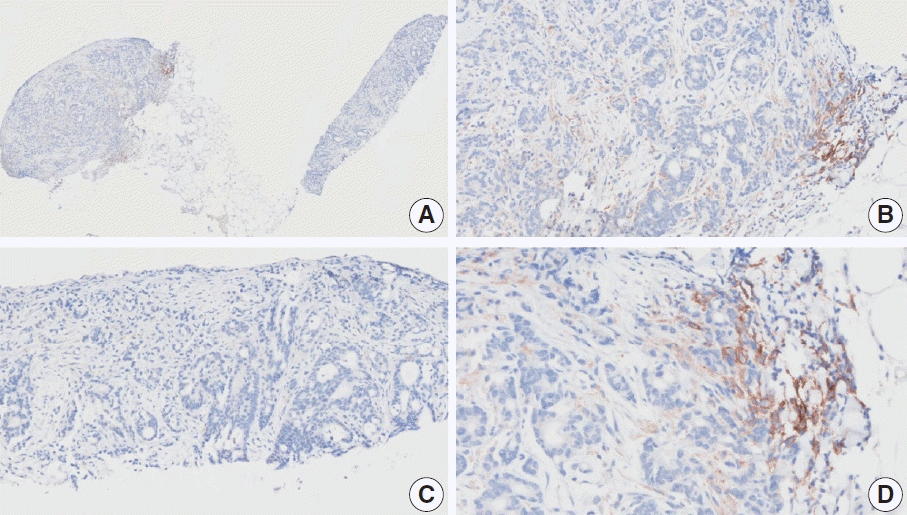
Fig. 5.
Representative images of case 7 with a consensus combined positive score of 2 and poor interobserver agreement at 4× (A), 10× (B), 20× (C), and 20× (D) magnification.
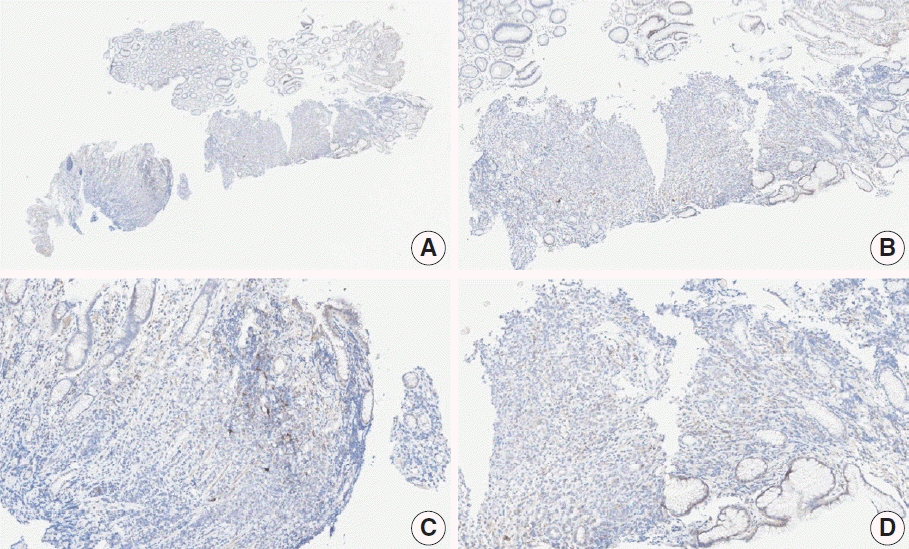
Fig. 6.
Representative images of case 9 with a consensus combined positive score of 2 and poor interobserver agreement at 2× (A), 20× (B), 20× (C), and 40× (D) magnification.
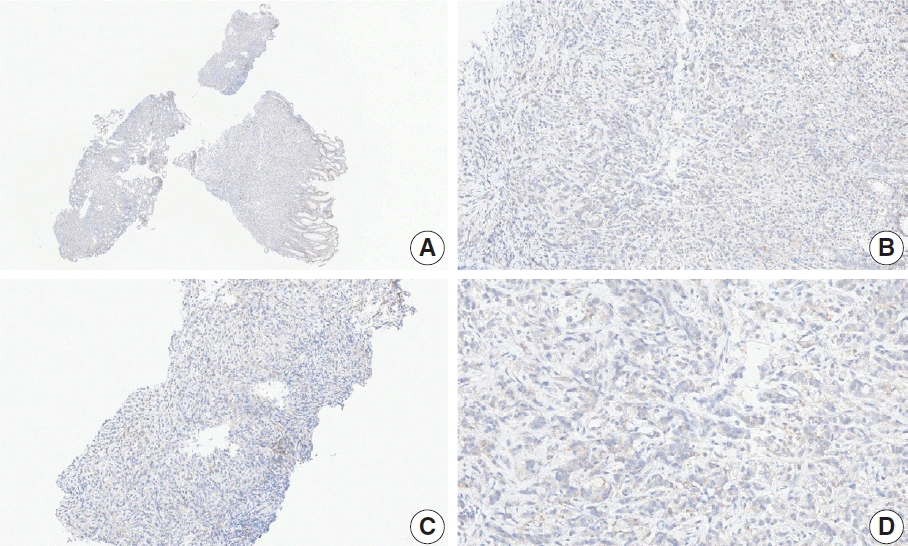
Fig. 7.
Representative images of case 11 with a consensus combined positive score of 3 and poor interobserver agreement at 2× (A), 20× (B), 20× (C), and 20× (D) magnification.
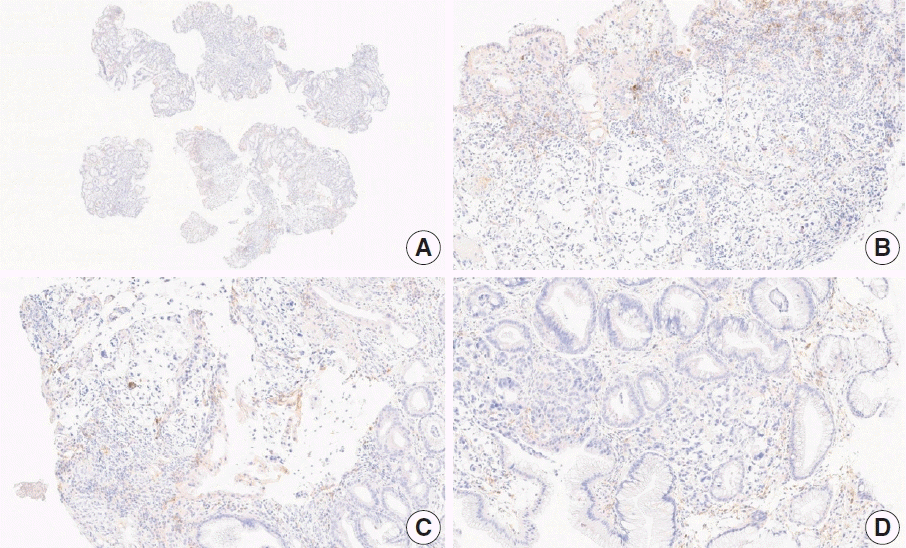
Fig. 8.
Representative images of case 17 with a consensus combined positive score of 2 and poor interobserver agreement at 2× (A), 20× (B), 20× (C), and 40× (D) magnification.
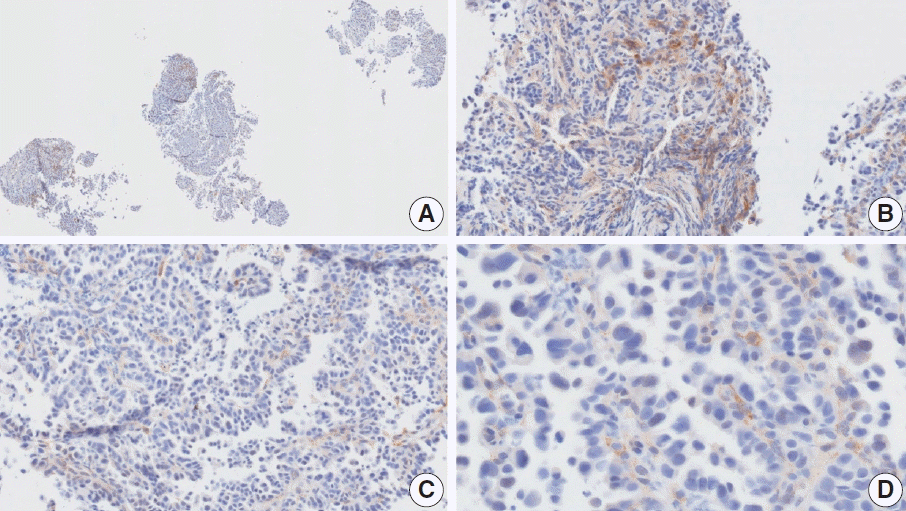
Fig. 9.
Representative images of case 20 with a consensus combined positive score of 0 and poor interobserver agreement at 4× (A), 10× (B), 20× (C), and 40× (D) magnification.
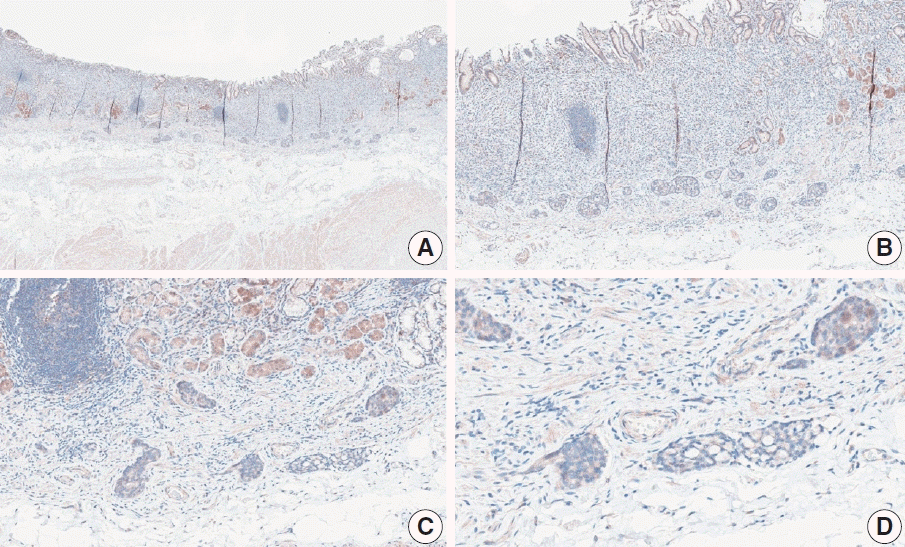
Fig. 10.
Representative images of case 5 with a consensus combined positive score of 5 and good interobserver agreement at 5× (A) and 20× (B) magnification.
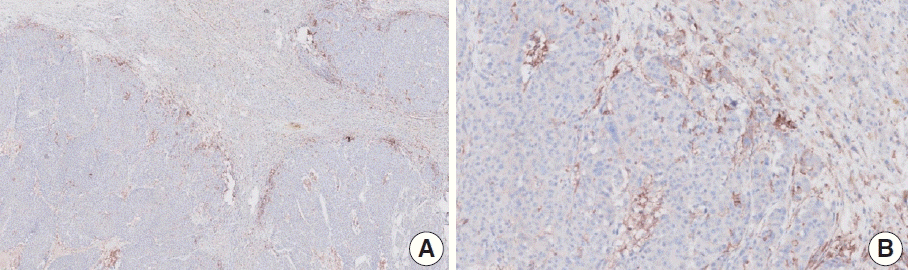
Fig. 11.
Representative images of case 10 with a consensus combined positive score of ≥ 5 and good interobserver agreement at 4× (A), 10× (B), 20× (C), and 20× (D) magnification.
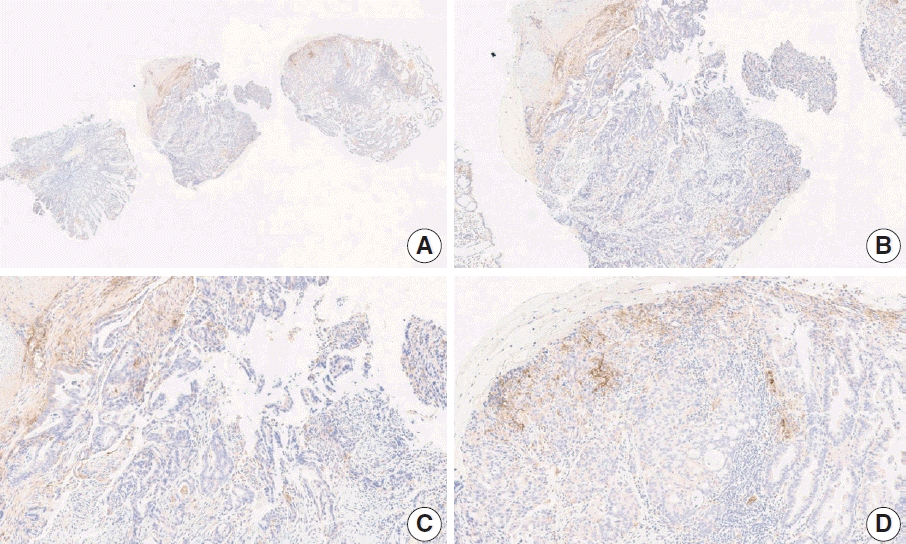
Fig. 12.
Representative images of case 12 with a consensus combined positive score of ≥ 5 and good interobserver agreement at 4× (A), 10× (B), 20× (C), and 20× (D) magnification.
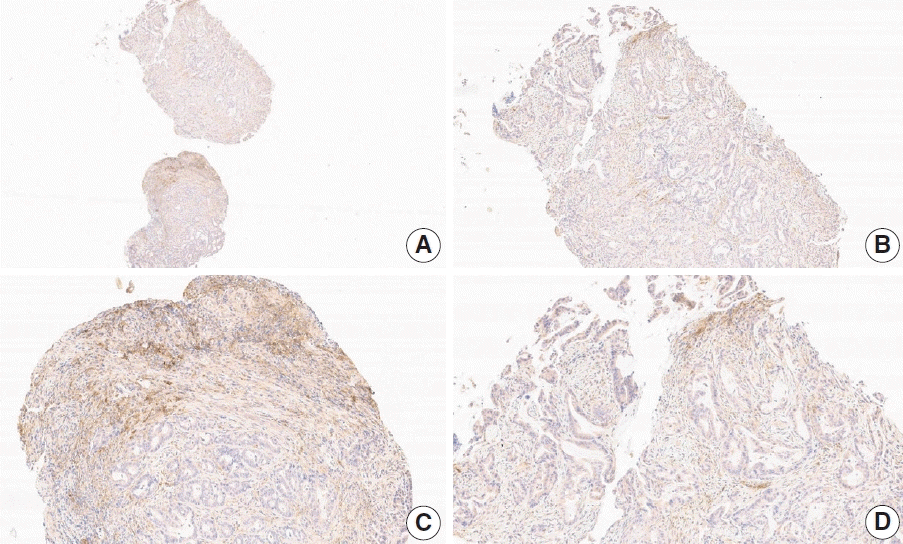
Fig. 13.
Representative images of case 18 with a consensus combined positive score of 5 and good interobserver agreement at 4× (A), 10× (B), 20× (C), and 20× (D) magnification.
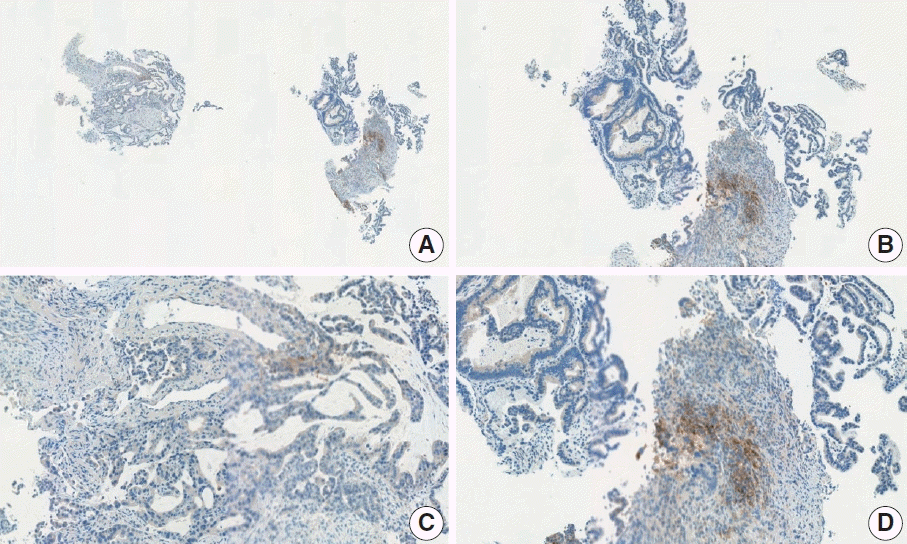
Fig. 14.
Representative images of case 19 with a consensus combined positive score of ≥ 5 and good interobserver agreement at 2× (A), 10× (B), 10× (C), and 20× (D) magnification.
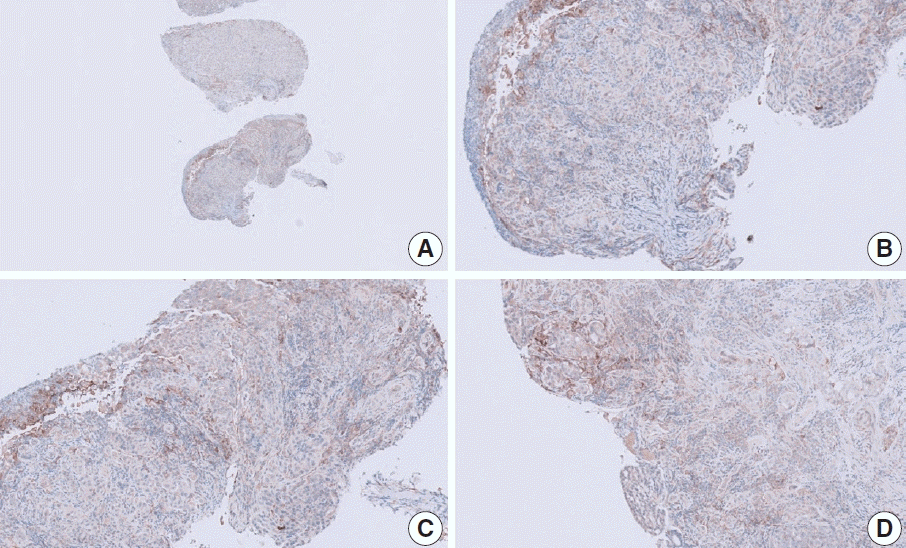
Fig. 15.
Representative images of case 4 with a consensus combined positive score of 5 and poor interobserver agreement at 5× (A), 5× (B), 20× (C), and 20× (D) magnification.
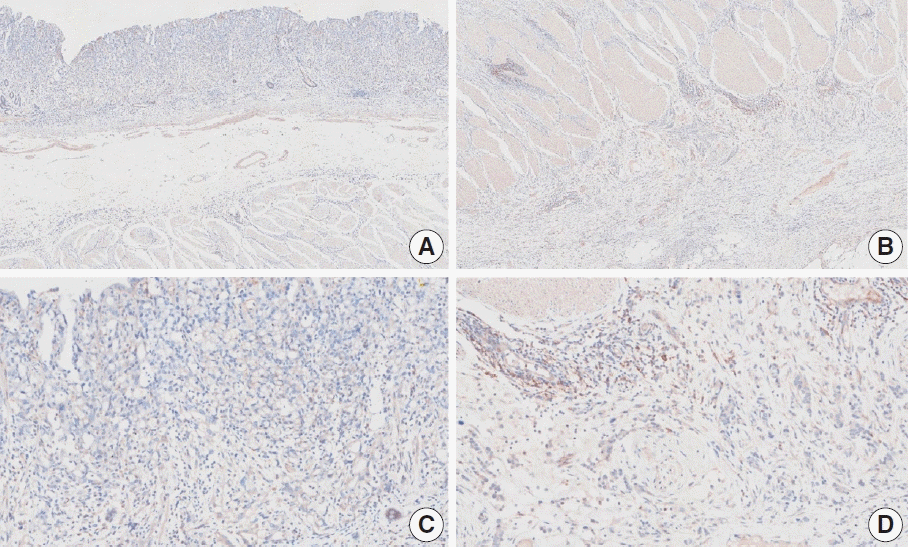
Fig. 16.
Representative images of case 8 with a consensus combined positive score of 5 and poor interobserver agreement at 2× (A), 10× (B), 20× (C), and 20× (D) magnification.
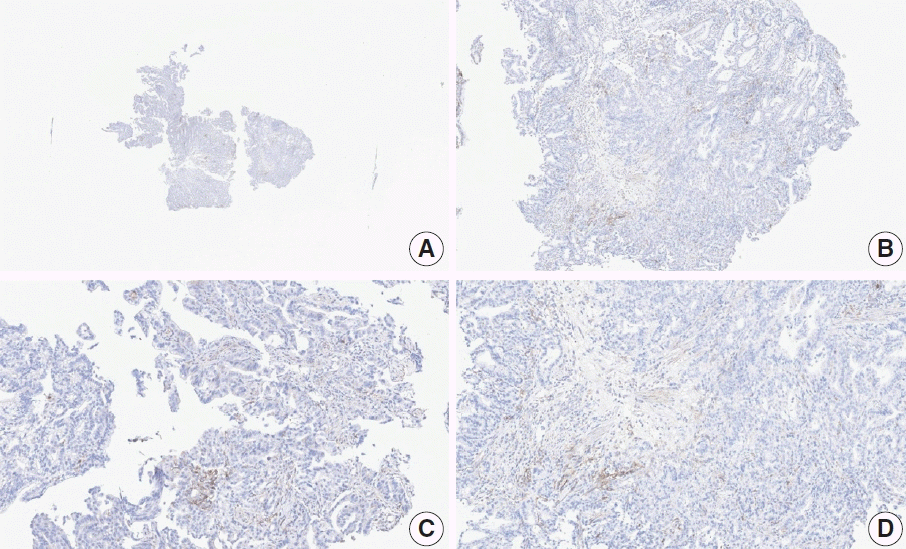
Fig. 17.
Representative images of case 13 with a consensus combined positive score of 5 and poor interobserver agreement at 1× (A), 2× (B), 10× (C), and 20× (D) magnification.
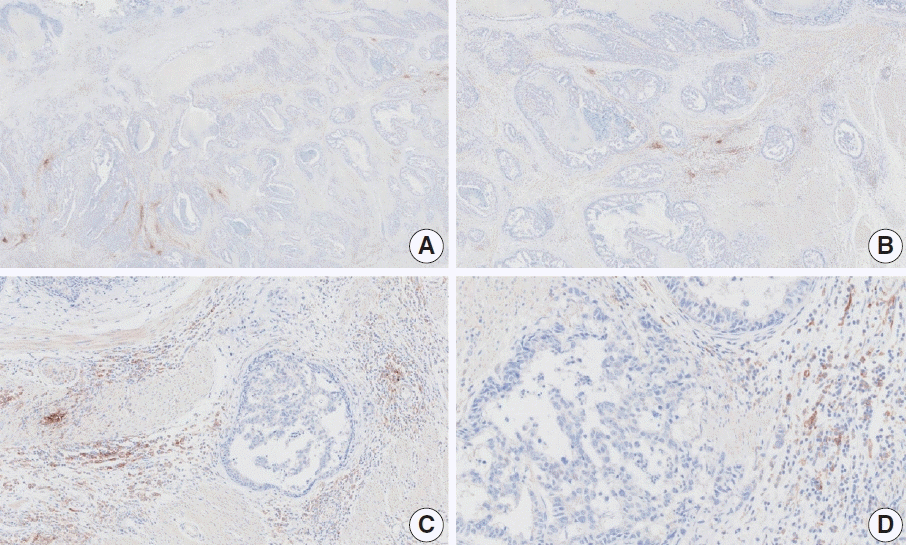
Fig. 18.
Representative images of case 14 with a consensus combined positive score of 5 and poor interobserver agreement at 2× (A), 10× (B), 20× (C), and 20× (D) magnification.
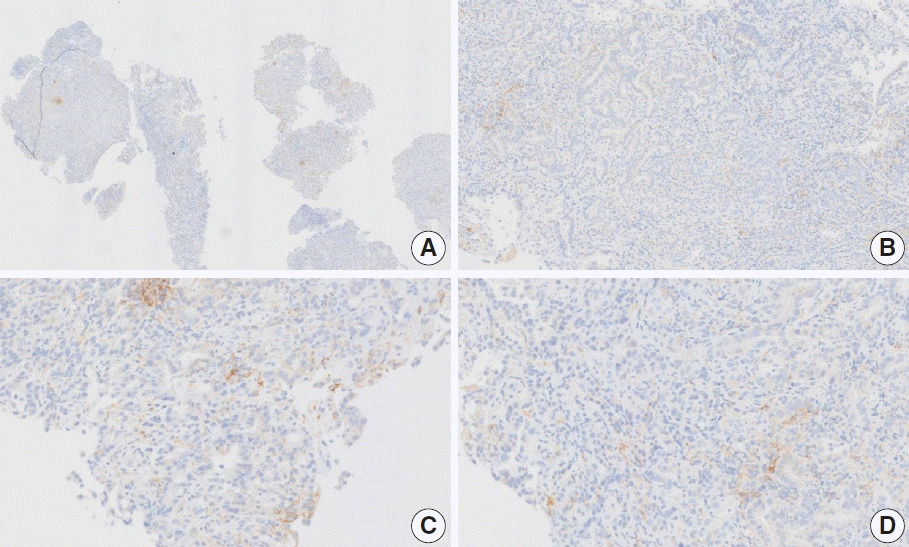
Fig. 19.
Representative images of case 16 with a consensus combined positive score of 5 and poor interobserver agreement at 1× (A), 5× (B) [box in A], 20× (C), and 20× (D) magnification.
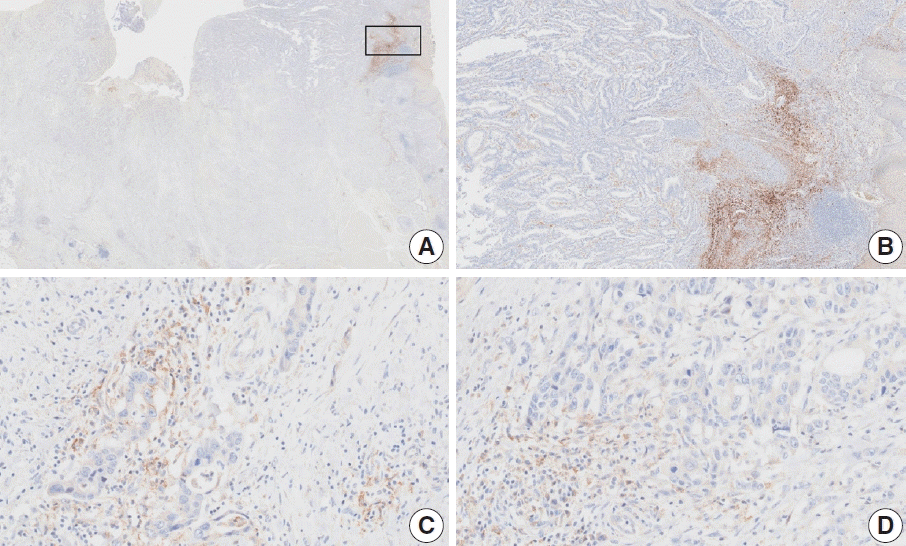
Table 1.
PD-L1 assays in clinical trials for gastric cancer (1st line setting)
Table 2.
Interobserver concordance in PD-L1 assays
| PD-L1 assay | Cutoff | No. of observers | No. of cases | Sample type | Interobserver agreement | Fleiss kappa value | Reference |
|---|---|---|---|---|---|---|---|
| 22C3 PharmDx | CPS ≥ 1 | 3 | 68 | Not mentioned | OPA 96.6% | - | [13] |
| 22C3 PharmDx | CPS ≥ 1 | 120 | 20 (day1), 25 (day2) | Resection | OPA 90.6% | 0.828 | [21] |
| 22C3 PharmDx | CPS value | 5 | 55 | Tissue microarray | ICC 0.387 (lower 95% CI, 20.9%) | CPS ≥ 1 0.389 | [22] |
| SP263 | CPS value | 5 | 55 | Tissue microarray | ICC 0.349 (lower 95% CI, 13.5%) | CPS ≥ 1 0.256 | |
| 22C3 PharmDx | CPS ≥ 1 | 14 | 112 | Biopsy | OPA 31.48% (95% CI, 22.72–40.24) | 0.477 | [18] |
| ICC 0.484 (95% CI, 0.403–0.571) | - | ||||||
| CPS ≥ 10 | 14 | 112 | Biopsy | OPA 67.59% (95% CI, 58.77–76.42) | 0.607 | ||
| ICC 0.604 (95% CI, 0.584–0.624) | - | ||||||
| CPS ≥ 20 | 14 | 112 | Biopsy | OPA 83.33% (95% CI, 76.3–90.36) | 0.626 | ||
| ICC 0.629 (95% CI, 0.562–0.698) | - | ||||||
| 28-8 PharmDx | CPS value | 12 | 100 | Biopsy | ICC 0.45 (95% CI, 0.38–0.53) | - | [19] |
| 22C3 PharmDx | CPS value | 12 | 100 | Biopsy | ICC 0.55 (95% CI, 0.47–0.63) | - |
Table 3.
Case information
| Case No. | Institution | Vendor/file format | Specimen type | Interobserver agreement | Consensus CPS |
|---|---|---|---|---|---|
| 1 | A | Philips/.isyntax | Resection | Gooda | 0 |
| 2 | A | Philips/.isyntax | Biopsy | Poor | 2 |
| 3 | A | Philips/.isyntax | Biopsy | Poor | Not determined |
| 4 | A | Philips/.isyntax | Resection | Poor | 5 |
| 5 | A | Philips/.isyntax | Resection | Good | 5 |
| 6 | A | Philips/.isyntax | Biopsy | Poor | 3 |
| 7 | B | Leica Biosystems/.svs | Biopsy | Poor | 2 |
| 8 | B | Leica Biosystems/.svs | Biopsy | Poor | 5 |
| 9 | B | Leica Biosystems/.svs | Biopsy | Poor | 2 |
| 10 | C | Leica Biosystems/.svs | Biopsy | Good | ≥ 5 |
| 11 | C | Leica Biosystems/.svs | Biopsy | Poor | 3 |
| 12 | C | Leica Biosystems/.svs | Biopsy | Good | ≥ 5 |
| 13 | D | Philips/.isyntax | Resection | Poor | 5 |
| 14 | D | Philips/.isyntax | Biopsy | Poor | 5 |
| 15 | D | Philips/.isyntax | Biopsy | Good | 2 |
| 16 | D | Philips/.isyntax | Resection | Poor | 5 |
| 17 | D | Philips/.isyntax | Biopsy | Poor | 2 |
| 18 | E | 3DHistech/.MRXS | Biopsy | Good | 5 |
| 19 | F | Philips/.isyntax | Biopsy | Good | ≥ 5 |
| 20 | G | Leica Biosystems/.svs | Resection | Poor | 0 |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download