Introduction
Materials and Methods
1. Study populations
2. Clinical data and adverse events collection
3. Study variables and statistical analysis
Results
1. Patient characteristics
Table 1.
| Characteristic | Total (n=1,170) | Female (n=493) | Male (n=677) | p-valuea) |
|---|---|---|---|---|
| Age (yr) | ||||
| Median (range) | 61 (17-84) | 57 (17-84) | 63 (29-83) | < 0.001 |
| ≥ 70 yr | 221 (18.9) | 69 (14.0) | 152 (22.5) | < 0.001 |
| ECOG PS | ||||
| 0-1 | 1,155 (98.7) | 486 (98.6) | 669 (98.8) | 0.721 |
| ≥ 2 | 15 (1.3) | 7 (1.4) | 8 (1.2) | |
| BMI (kg/m2), median (range) | 22.7 (14.2-35.6) | 22.6 (14.2-35.6) | 22.7 (15.4-34.4) | 0.314 |
| Past or current medical historyb) | ||||
| Present | 621 (53.1) | 224 (45.4) | 397 (58.6) | < 0.001 |
| None | 547 (46.8) | 269 (54.6) | 278 (41.1) | |
| Unknown | 2 (0.2) | 0 | 2 (0.3) | |
| Marriage | ||||
| Single | 51 (4.4) | 27 (5.5) | 24 (3.5) | 0.100c) |
| Married | 941 (80.4) | 379 (76.9) | 562 (83.0) | |
| Unknown | 178 (15.2) | 87 (17.6) | 91 (13.4) | |
| Cancer type | ||||
| Colon | 366 (31.3) | 178 (36.1) | 188 (27.8) | 0.008 |
| Stomach | 387 (33.1) | 147 (29.8) | 240 (35.5) | |
| Lung | 417 (35.6) | 158 (34.1) | 249 (36.8) | |
| Pathologic stage | ||||
| 1-2 | 621 (53.1) | 261 (52.9) | 360 (53.2) | 0.937 |
| 3-4 | 549 (46.9) | 232 (47.1) | 317 (46.8) | |
| Chemotherapy regimend) | ||||
| Single-agent | 351 (30.0) | 149 (30.2) | 202 (29.8) | 0.887 |
| Platinum-containing | 819 (70.0) | 344 (69.8) | 475 (70.2) | |
| Extensive surgerye) | 44 (3.8) | 14 (2.8) | 30 (4.4) | 0.158 |
| Postoperative weight loss > 10% | 211 (18.0) | 71 (14.4) | 140 (20.7) | 0.006 |
Values are presented as number (%) unless otherwise indicated. BMI, body mass index; ECOG PS, Eastern Cooperative Oncology Group performance status.
a) p-values were calculated using Pearson’s chi-squared test or Fisher’s exact test as appropriate on the percentage difference of female vs. male patients,
b) Medical history included hypertension, diabetes mellitus, tuberculosis, hepatitis, congestive heart failure, coronary artery disease, and chronic obstructive pulmonary disease,
2. Incidence of common adverse events according to sex
Fig. 1.
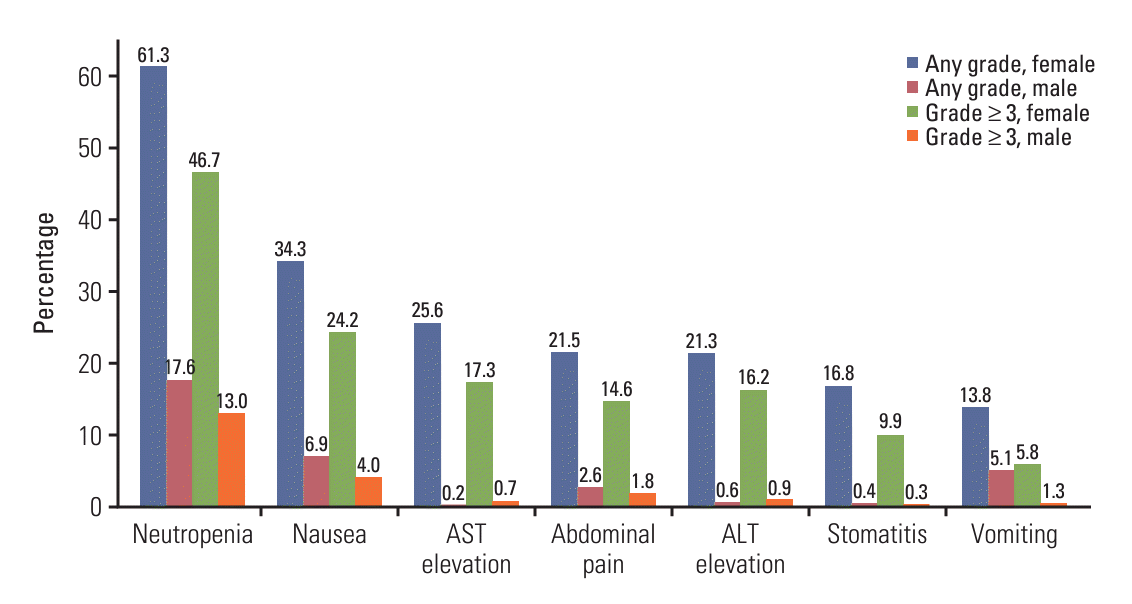
Table 2.
3. Association between sex and adverse events/chemotherapy regimen-related toxicities
Table 3.
| Female events/total (%) | Male events/total (%) | Odds ratioa) (95% CI) | p-value | |
|---|---|---|---|---|
| AST elevation | ||||
| All regimens | 126/493 (25.6) | 117/677 (17.3) | 1.58 (1.15-2.16) | 0.004 |
| Single-agent | 35/149 (23.5) | 36/202 (17.8) | 1.44 (0.84-2.48) | 0.186 |
| Platinum-containing regimen | 91/344 (26.5) | 81/475 (17.1) | 1.65 (1.13-2.42) | 0.010 |
| ALT elevation | ||||
| All regimens | 105/493 (21.3) | 110/667 (16.2) | 1.28 (0.93-1.75) | 0.127 |
| Single-agent | 31/149 (20.8) | 32/202 (15.8) | 1.38 (0.78-2.42) | 0.269 |
| Platinum-containing regimen | 74/344 (21.5) | 78/475 (16.4) | 1.24 (0.85-1.81) | 0.272 |
| Abdominal pain | ||||
| All regimens | 106/493 (21.5) | 99/677 (14.6) | 1.61 (1.16-2.23) | 0.004 |
| Single-agent | 55/149 (36.9) | 55/202 (27.2) | 1.53 (0.96-2.46) | 0.075 |
| Platinum-containing regimen | 51/344 (14.8) | 44/475 (9.3) | 1.68 (1.06-2.65) | 0.027 |
| Arthralgia | ||||
| All regimens | 20/493 (4.1) | 10/677 (1.5) | 1.84 (1.43-2.38) | < 0.001 |
| Single-agent | 4/149 (2.7) | 1/202 (0.5) | 5.19 (0.55-49.13) | 0.151 |
| Platinum-containing regimen | 16/344 (4.7) | 9/475 (1.9) | 2.37 (1.02-5.47) | 0.044 |
| Nausea | ||||
| All regimens | 169/493 (34.3) | 164/677 (24.2) | 1.80 (1.36-2.36) | < 0.001 |
| Single-agent | 48/149 (32.2) | 51/202 (25.2) | 1.51 (0.93-2.45) | 0.098 |
| Platinum-containing regimen | 121/344 (35.2) | 113/475 (23.8) | 1.94 (1.38-2.71) | < 0.001 |
| Stomatitis | ||||
| All regimens | 83/493 (16.8) | 67/677 (9.9) | 1.83 (1.29-2.62) | 0.001 |
| Single-agent | 27/149 (18.1) | 26/202 (12.9) | 1.37 (0.75-2.51) | 0.305 |
| Platinum-containing regimen | 56/344 (16.3) | 41/475 (8.6) | 2.20 (1.41-3.44) | 0.001 |
| Vomiting | ||||
| All regimens | 68/493 (13.8) | 39/677 (5.8) | 2.80 (1.81-4.35) | < 0.001 |
| Single-agent | 10/149 (6.7) | 9/202 (4.5) | 1.42 (0.54-3.73) | 0.480 |
| Platinum-containing regimen | 58/344 (16.9) | 30/475 (6.3) | 3.29 (2.00-5.42) | < 0.001 |
| Neutropenia | ||||
| All regimens | 302/493 (61.3) | 316/677 (46.7) | 1.84 (1.43-2.38) | < 0.001 |
| Single-agent | 88/149 (59.1) | 78/202 (38.6) | 2.22 (1.42-3.45) | < 0.001 |
| Platinum-containing regimen | 214/344 (62.2) | 238/475 (50.1) | 1.68 (1.23-2.29) | 0.001 |
Table 4.
| Female events/total (%) | Male events/total (%) | Odds ratioa) (95% CI) | p-value | |
|---|---|---|---|---|
| Nausea | ||||
| All regimens | 34/493 (6.9) | 27/677 (4.0) | 1.72 (1.00-2.97) | 0.050 |
| Single-agent | 1/149 (0.7) | 4/202 (2.0) | 0.37 (0.04-3.45) | 0.385 |
| Platinum-containing regimen | 33/344 (9.6) | 23/475 (4.8) | 1.99 (1.12-3.54) | 0.019 |
| Vomiting | ||||
| All regimens | 25/493 (5.1) | 9/677 (1.3) | 3.45 (1.55-7.66) | 0.002 |
| Single-agent | 1/149 (0.7) | 0/202 (0.0) | - | - |
| Platinum-containing regimen | 24/344 (7.0) | 9/475 (1.9) | 3.30 (1.48-7.37) | 0.004 |
| Neutropenia | ||||
| All regimens | 87/493 (17.6) | 88/677 (13.0) | 1.36 (0.97-1.90) | 0.076 |
| Single-agent | 14/149 (9.4) | 9/202 (4.5) | 1.86 (0.76-4.53) | 0.176 |
| Platinum-containing regimen | 73/344 (21.2) | 79/475 (16.6) | 1.28 (0.89-1.84) | 0.186 |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download