Abstract
Purpose
Materials and Methods
Results
Electronic Supplementary Material
Notes
Ethical Statement
The study was approved by the Institutional Review Board of Seoul National University Hospital (IRB No. 2301-164-1403), and the need for informed consent was waived.
Author Contributions
Conceived and designed the analysis: Kim K, Shin KH.
Collected the data: Chun SJ, Jo JH, Kim YB, Park S, Ahn SJ, Kim SS, Shin KH.
Contributed data or analysis tools: Chun SJ, Kim K, Shin KH.
Performed the analysis: Chun SJ, Kim K.
Wrote the paper: Chun SJ, Jo JH, Kim YB, Park S, Ahn SJ, Kim SS, Kim K, Shin KH.
References
Fig. 1.
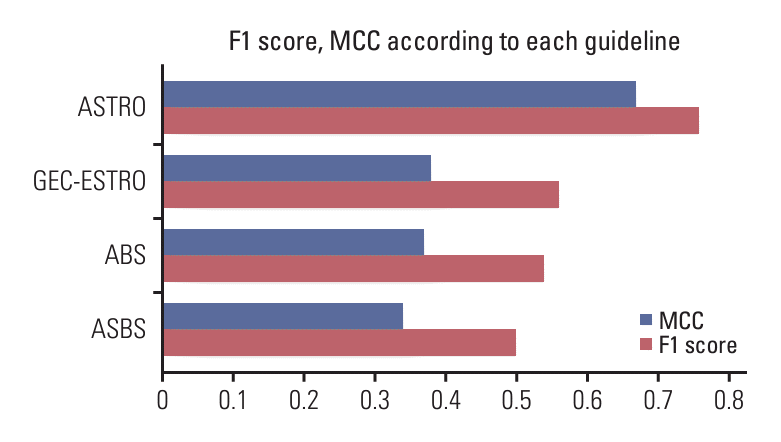
Table 1.
| ABS | ASBS | ASTRO (‘suitable’) | ASTRO (‘cautionary’) | GEC-ESTRO (‘low-risk’) | GEC-ESTRO (‘intermediate-risk’) | |
|---|---|---|---|---|---|---|
| Age (yr) | ≥ 45 | ≥ 45 | ≥ 50 | ≥ 40 | > 50 | > 40, ≤ 50 |
| Histology | All invasive | All invasive | All invasive, but not ILC | All invasive, including ILCa) | IDC, mucinous, tubular, medullary, colloid | IDC, ILC, mucinous, tubular, medullary, colloid |
| Tumor size (cm) | ≤ 3 (total) | ≤ 3 (total) | ≤ 2 (tumor) | 2-3 (tumor)a) | ≤ 3 (tumor size) | ≤ 3 (tumor size) |
| ≤ 3 (total) | ≤ 3 (total) | |||||
| Lymph node | pN0 | pN0 | pN0 | pN0 | pN0 | pN0 |
| Margin | Negative | Negative | ≥ 2 mm | Close (< 2 mm)a) | ≥ 2 mm | Close (< 2 mm) |
| LVSI | No | No | No | Yesa) | No | No |
| ER | Any | Any | Positive | Negativea) | Any | Any |
| HER2 | Negative | Any | Any | Any | Any | Any |
| EIC | Any | Any | No | ≤ 3 cma) | No | No |
| Focality | Unifocal | Any | Unifocal | Unifocalb) | Unifocal | Any |
ABS, American Brachytherapy Society; ASBS, American Society of Breast Surgeons; ASTRO, American Society for Radiation Oncology; EIC, extensive intraductal component; ER, estrogen receptor; GEC-ESTRO, Groupe Européen de Curiethérapie and the European Society for Radiotherapy and Oncology; HER2, human epidermal growth factor receptor 2; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; LVSI, lymphovascular invasion.
Table 2.
| Variable | Total patients (n=1,251) | WBI (n=1,055) | APBI (n=196) | p-value |
|---|---|---|---|---|
| Age (yr) | ||||
| < 45 | 218 (17.4) | 216 (20.5) | 2 (1.0) | < 0.001 |
| 45-50 | 277 (22.1) | 248 (23.5) | 29 (14.8) | |
| > 50 | 756 (60.4) | 591 (56.0) | 165 (84.2) | |
| Laterality | ||||
| Left | 621 (49.6) | 525 (49.8) | 96 (49.0) | 0.902 |
| Right | 630 (50.4) | 530 (50.2) | 100 (51.0) | |
| Axillary surgery | ||||
| SLNBx | 1,222 (97.7) | 1,029 (97.5) | 193 (98.5) | 0.499 |
| ALND | 25 (2.0) | 23 (2.2) | 2 (1.0) | |
| Not done | 4 (0.3) | 3 (0.3) | 1 (0.5) | |
| Histology | ||||
| IDC | 1,155 (92.3) | 982 (93.1) | 173 (88.3) | < 0.001 |
| ILC | 44 (3.5) | 40 (3.8) | 4 (2.0) | |
| Others | 52 (4.2) | 33 (3.1) | 19 (9.7) | |
| Size (invasive tumor only) (cm) | ||||
| ≤ 2 | 973 (77.8) | 794 (75.3) | 179 (91.3) | < 0.001 |
| > 2 and ≤ 3 | 234 (18.7) | 218 (20.7) | 16 (8.2) | |
| > 3 | 44 (3.5) | 43 (4.1) | 1 (0.5) | |
| Size (including DCIS) (cm) | ||||
| ≤ 2 | 788 (63.0) | 620 (58.8) | 168 (85.7) | < 0.001 |
| > 2 and ≤ 3 | 307 (24.5) | 282 (26.7) | 25 (12.8) | |
| > 3 | 156 (12.5) | 153 (14.5) | 3 (1.5) | |
| EIC | ||||
| Positive | 322 (25.7) | 294 (27.9) | 28 (14.3) | < 0.001 |
| Negative | 883 (70.6) | 727 (68.9) | 156 (79.6) | |
| N/A | 46 (3.7) | 34 (3.2) | 12 (6.1) | |
| Focality | ||||
| Unifocal | 1,082 (86.5) | 902 (85.5) | 180 (91.8) | 0.023 |
| Multifocal | 169 (13.5) | 153 (14.5) | 16 (8.2) | |
| Centricity | ||||
| Unicentric | 1,245 (99.5) | 1,049 (99.4) | 196 (100) | 0.620 |
| Multicentric | 6 (0.5) | 6 (0.6) | 0 | |
| Resection margin | ||||
| Positive | 74 (5.9) | 73 (6.9) | 1 (0.5) | < 0.001 |
| Close (< 2 mm) | 436 (34.9) | 404 (38.3) | 32 (16.3) | |
| Negative (≥ 2 mm) | 615 (49.2) | 495 (46.9) | 120 (61.2) | |
| N/Aa) | 126 (10.1) | 83 (7.9) | 43 (21.9) | |
| LVSI | ||||
| Positive | 129 (10.3) | 127 (12.0) | 2 (1.0) | < 0.001 |
| Negative | 1,122 (89.7) | 928 (88.0) | 194 (99.0) | |
| ER status | ||||
| Positive | 1,000 (79.9) | 806 (76.4) | 194 (99.0) | < 0.001 |
| Negative | 251 (20.1) | 249 (23.6) | 2 (1.0) | |
| HER2 status | ||||
| Positive | 153 (12.2) | 151 (14.3) | 2 (1.0) | < 0.001 |
| Negative | 1,097 (87.7) | 903 (85.6) | 194 (99.0) | |
| N/A | 1 (0.1) | 1 (0.1) | 0 | |
| Ki-67 (%) | ||||
| < 20 | 803 (64.2) | 622 (59.0) | 181 (92.3) | < 0.001 |
| ≥ 20 | 445 (35.6) | 430 (40.8) | 15 (7.7) | |
| N/A | 3 (0.2) | 3 (0.3) | 0 | |
| Adjuvant chemotherapy | ||||
| Yes | 426 (34.1) | 409 (38.8) | 17 (8.7) | < 0.001 |
| No | 825 (65.9) | 646 (61.2) | 179 (91.3) | |
| Adjuvant endocrine therapy | ||||
| Yes (TMX) | 535 (42.8) | 481 (45.6) | 54 (27.6) | < 0.001 |
| Yes (AI) | 421 (33.7) | 311 (29.5) | 110 (56.1) | |
| No | 295 (23.6) | 263 (24.9) | 32 (16.3) |
Values are presented as number (%). AI, aromatase inhibitor; ALND, axillary lymph node dissection; APBI, accelerated partial breast irradiation; DCIS, ductal carcinoma in situ; EIC, extensive intraductal component; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; LVSI, lymphovascular space invasion; N/A, not available; SLNBx, sentinel lymph node biopsy; TMX, tamoxifen; WBI, whole breast irradiation.
Table 3.
| Criteria | Total patients (n=1,251) | WBI (n=1,055) | APBI (n=196) | Actual APBI rates |
|---|---|---|---|---|
| ASTROa) | ||||
| Suitable | 158 (13.7) | 72 (7.2) | 86 (55.1) | 86/158 (54.4) |
| Cautionary | 610 (52.9) | 548 (54.9) | 62 (39.7) | 62/610 (10.2) |
| Unsuitable | 386 (33.4) | 378 (37.9) | 8 (5.1) | 8/386 (2.1) |
| GEC-ESTROa) | ||||
| Low-risk | 242 (21.0) | 152 (15.2) | 90 (58.4) | 90/242 (37.2) |
| Intermediate-risk | 385 (33.3) | 352 (35.2) | 33 (21.4) | 33/385 (8.6) |
| High-risk | 527 (45.7) | 496 (49.6) | 31 (20.1) | 31/527 (5.9) |
| ABS | ||||
| Acceptable | 632 (50.5) | 461 (43.7) | 171 (87.2) | 171/632 (27.1) |
| Unacceptable | 619 (49.5) | 594 (56.3) | 25 (12.8) | 25/619 (4.0) |
| ASBS | ||||
| Recommended | 794 (63.5) | 606 (57.4) | 188 (95.9) | 188/794 (23.7) |
| Not recommended | 457 (36.5) | 449 (42.6) | 8 (4.1) | 8/457 (1.8) |
Values are presented as number (%). ABS, American Brachytherapy Society; APBI, accelerated partial breast irradiation; ASBS, American Society of Breast Surgeons; ASTRO, American Society for Radiation Oncology; GEC-ESTRO, Groupe Européen de Curiethérapie and the European Society for Radiotherapy and Oncology; WBI, whole breast irradiation.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download