1. Pirillo A, Casula M, Olmastroni E, Norata GD, Catapano AL. Global epidemiology of dyslipidaemias. Nat Rev Cardiol. 2021; 18:689–700.

2. Bazarbashi N, Miller M. Triglycerides: how to manage patients with elevated triglycerides and when to refer? Med Clin North Am. 2022; 106:299–312.
3. Wolska A, Yang ZH, Remaley AT. Hypertriglyceridemia: new approaches in management and treatment. Curr Opin Lipidol. 2020; 31:331–9.

4. Rygiel K. Hypertriglyceridemia: common causes, prevention and treatment strategies. Curr Cardiol Rev. 2018; 14:67–76.
5. Reiner Z. Hypertriglyceridaemia and risk of coronary artery disease. Nat Rev Cardiol. 2017; 14:401–11.

6. Patel A, Barzi F, Jamrozik K, Lam TH, Ueshima H, Whitlock G, et al. Serum triglycerides as a risk factor for cardiovascular diseases in the Asia-Pacific region. Circulation. 2004; 110:2678–86.

7. Lin CF, Chang YH, Chien SC, Lin YH, Yeh HY. Epidemiology of dyslipidemia in the Asia Pacific region. Int J Gerontol. 2018; 12:2–6.

8. Mohamed-Yassin MS, Baharudin N, Daher AM, Abu Bakar N, Ramli AS, Abdul-Razak S, et al. High prevalence of dyslipidaemia subtypes and their associated personal and clinical attributes in Malaysian adults: the REDISCOVER study. BMC Cardiovasc Disord. 2021; 21:149.

9. Cho SM, Lee H, Lee HH, Baek J, Heo JE, Joo HJ, et al. Dyslipidemia fact sheets in Korea 2020: an analysis of nationwide population-based data. J Lipid Atheroscler. 2021; 10:202–9.

10. Ihm SH, Chung WB, Lee JM, Hwang BH, Yoo KD, Her SH, et al. Efficacy and tolerability of pitavastatin versus pitavastatin/fenofibrate in high-risk Korean patients with mixed dyslipidemia: a multicenter, randomized, double-blinded, parallel, therapeutic confirmatory clinical trial. Clin Ther. 2020; 42:2021–35.

11. Kim NH, Kim SG. Fibrates revisited: potential role in cardiovascular risk reduction. Diabetes Metab J. 2020; 44:213–21.

12. Sando KR, Knight M. Nonstatin therapies for management of dyslipidemia: a review. Clin Ther. 2015; 37:2153–79.

13. Lian X, Wang G, Zhou H, Zheng Z, Fu Y, Cai L. Anticancer properties of fenofibrate: a repurposing use. J Cancer. 2018; 9:1527–37.

14. Moon JH, Kim K, Choi SH. Lipoprotein lipase: is it a magic target for the treatment of hypertriglyceridemia. Endocrinol Metab (Seoul). 2022; 37:575–86.

15. Jiang S, Uddin MJ, Yu X, Piao L, Dorotea D, Oh GT, et al. Peroxisomal fitness: a potential protective mechanism of fenofibrate against high fat diet-induced non-alcoholic fatty liver disease in mice. Diabetes Metab J. 2022; 46:829–42.

16. Rhee EJ, Kim HC, Kim JH, Lee EY, Kim BJ, Kim EM, et al. 2018 Guidelines for the management of dyslipidemia in Korea. J Lipid Atheroscler. 2019; 8:78–131.

17. Li YH, Ueng KC, Jeng JS, Charng MJ, Lin TH, Chien KL, et al. 2017 Taiwan lipid guidelines for high risk patients. J Formos Med Assoc. 2017; 116:217–48.

19. Wang G, He L, Liu J, Yu J, Feng X, Li F, et al. Coronary flow velocity reserve is improved by PPAR-α agonist fenofibrate in patients with hypertriglyceridemia. Cardiovasc Ther. 2013; 31:161–7.

20. Min YJ, Choi YH, Hyeon CW, Cho JH, Kim KJ, Kwon JE, et al. Fenofibrate reduces C-reactive protein levels in hypertriglyceridemic patients with high risks for cardiovascular diseases. Korean Circ J. 2012; 42:741–6.

21. Guo J, Meng F, Ma N, Li C, Ding Z, Wang H, et al. Meta-analysis of safety of the coadministration of statin with fenofibrate in patients with combined hyperlipidemia. Am J Cardiol. 2012; 110:1296–301.

22. Geng Q, Ren J, Chen H, Lee C, Liang W. Adverse events following statin-fenofibrate therapy versus statin alone: a meta-analysis of randomized controlled trials. Clin Exp Pharmacol Physiol. 2013; 40:219–26.

23. Lee SH, Cho KI, Kim JY, Ahn YK, Rha SW, Kim YJ, et al. Nonlipid effects of rosuvastatin-fenofibrate combination therapy in high-risk Asian patients with mixed hyperlipidemia. Atherosclerosis. 2012; 221:169–75.

24. Zhao S, Wang F, Dai Y, Lin L, Tong Q, Liao Y, et al. Efficacy and safety of fenofibrate as an add-on in patients with elevated triglyceride despite receiving statin treatment. Int J Cardiol. 2016; 221:832–6.

25. Park MS, Youn JC, Kim EJ, Han KH, Lee SH, Kim SH, et al. Efficacy and safety of fenofibrate-statin combination therapy in patients with inadequately controlled triglyceride levels despite previous statin monotherapy: a multicenter, randomized, double-blind, phase IV study. Clin Ther. 2021; 43:1735–47.

26. Woo Y, Shin JS, Shim CY, Kim JS, Kim BK, Park S, et al. Effect of fenofibrate in 1113 patients at low-density lipoprotein cholesterol goal but high triglyceride levels: real-world results and factors associated with triglyceride reduction. PLoS One. 2018; 13:e0205006.

27. Koh KK, Quon MJ, Shin KC, Lim S, Lee Y, Sakuma I, et al. Significant differential effects of omega-3 fatty acids and fenofibrate in patients with hypertriglyceridemia. Atherosclerosis. 2012; 220:537–44.

28. Koh KK, Oh PC, Sakuma I, Lee Y, Han SH, Shin EK. Vascular and metabolic effects of omega-3 fatty acids combined with fenofibrate in patients with hypertriglyceridemia. Int J Cardiol. 2016; 221:342–6.

29. Oikawa S, Yamashita S, Nakaya N, Sasaki J, Kono S. Effect of fenofibrate and ezetimibe combination treatment on lipid (EFECTL) study investigators. Efficacy and safety of long-term coadministration of fenofibrate and ezetimibe in patients with combined hyperlipidemia: results of the EFECTL Study. J Atheroscler Thromb. 2017; 24:77–94.

30. Wang HY, Jiao QP, Chen SY, Sheng J, Jiang H, Lu J, et al. Efficacy and safety of policosanol plus fenofibrate combination therapy in elderly patients with mixed dyslipidemia: a randomized, controlled clinical study. Am J Med Sci. 2018; 356:254–61.

31. Parhofer KG, Laufs U. The diagnosis and treatment of hypertriglyceridemia. Dtsch Arztebl Int. 2019; 116:825–32.

32. Bao W, Kong R, Wang N, Han W, Lu J. PPAR-alpha agonist fenofibrate combined with octreotide acetate in the treatment of acute hyperlipidemia pancreatitis. PPAR Res. 2021; 2021:6629455.

33. Liu J, Lu R, Wang Y, Hu Y, Jia Y, Yang N, et al. PPARα agonist fenofibrate reduced the secreting load of β-cells in hypertriglyceridemia patients with normal glucose tolerance. PPAR Res. 2016; 2016:6232036.
34. Ma M, Liu H, Yu J, He S, Li P, Ma C, et al. Triglyceride is independently correlated with insulin resistance and islet beta cell function: a study in population with different glucose and lipid metabolism states. Lipids Health Dis. 2020; 19:121.

35. Lee CY, Huang KH, Lin CC, Tsai TH, Shih HC. A neutral risk on the development of new-onset diabetes mellitus (NODM) in Taiwanese patients with dyslipidaemia treated with fibrates. ScientificWorldJournal. 2012; 2012:392734.

36. Feng X, Gao X, Jia Y, Xu Y. PPAR-α agonist fenofibrate reduces insulin resistance in impaired glucose tolerance patients with hypertriglyceridemia: a cross-sectional study. Diabetes Ther. 2017; 8:433–44.

37. Yan F, Tian L, Xiao Z, Li S, Fu M, Tian H. Comparison of the efficacy of fenofibrate and acipimox on plasma lipoprotein subclasses distribution in the Chinese population with type 2 diabetes mellitus and hypertriglyceridemia. Clin Lipidol. 2014; 9:171–7.

38. Shinnakasu A, Yamamoto K, Kurano M, Arimura H, Arimura A, Kikuti A, et al. The combination therapy of fenofibrate and ezetimibe improved lipid profile and vascular function compared with statins in patients with type 2 diabetes. J Atheroscler Thromb. 2017; 24:735–48.

39. Keech A, Simes RJ, Barter P, Best J, Scott R, Taskinen MR, et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet. 2005; 366:1849–61.

40. ACCORD Study Group, Ginsberg HN, Elam MB, Lovato LC, Crouse JR 3rd, Leiter LA, et al. Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010; 362:1563–74.

41. Jo SH, Nam H, Lee J, Park S, Lee J, Kyoung DS. Fenofibrate use is associated with lower mortality and fewer cardiovascular events in patients with diabetes: results of 10,114 patients from the Korean National Health Insurance Service Cohort. Diabetes Care. 2021; 44:1868–76.

42. Ferreira JP, Vasques-Novoa F, Ferrao D, Saraiva F, Falcao-Pires I, Neves JS, et al. Fenofibrate and heart failure outcomes in patients with type 2 diabetes: analysis from ACCORD. Diabetes Care. 2022; 45:1584–91.

43. Feng X, Gao X, Jia Y, Zhang H, Pan Q, Yao Z, et al. PPAR-α agonist fenofibrate decreased serum irisin levels in type 2 diabetes patients with hypertriglyceridemia. PPAR Res. 2015; 2015:924131.
44. Ho MY, Wang CY. Role of irisin in myocardial infarction, heart failure, and cardiac hypertrophy. Cells. 2021; 10:2103.

45. Feng X, Gao X, Jia Y, Zhang H, Xu Y, Wang G. PPAR-α agonist fenofibrate decreased RANTES levels in type 2 diabetes patients with hypertriglyceridemia. Med Sci Monit. 2016; 22:743–51.

46. Das Pradhan A, Glynn RJ, Fruchart JC, MacFadyen JG, Zaharris ES, Everett BM, et al. Triglyceride lowering with pemafibrate to reduce cardiovascular risk. N Engl J Med. 2022; 387:1923–34.

47. d’Emden MC, Jenkins AJ, Li L, Zannino D, Mann KP, Best JD, et al. Favourable effects of fenofibrate on lipids and cardiovascular disease in women with type 2 diabetes: results from the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study. Diabetologia. 2014; 57:2296–303.

48. Keech AC, Mitchell P, Summanen PA, O’Day J, Davis TM, Moffitt MS, et al. Effect of fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): a randomised controlled trial. Lancet. 2007; 370:1687–97.

49. Chew EY, Davis MD, Danis RP, Lovato JF, Perdue LH, Greven C, et al. The effects of medical management on the progression of diabetic retinopathy in persons with type 2 diabetes: the action to control cardiovascular risk in diabetes (ACCORD) eye study. Ophthalmology. 2014; 121:2443–51.

50. Ju HB, Zhang FX, Wang S, Song J, Cui T, Li LF, et al. Effects of fenofibrate on inflammatory cytokines in diabetic retinopathy patients. Medicine (Baltimore). 2017; 96:e7671.

51. Kawasaki R, Konta T, Nishida K. Lipid-lowering medication is associated with decreased risk of diabetic retinopathy and the need for treatment in patients with type 2 diabetes: a real-world observational analysis of a health claims database. Diabetes Obes Metab. 2018; 20:2351–60.

52. Kawasaki R, Kitano S, Sato Y, Yamashita H, Nishimura R, Tajima N, et al. Factors associated with non-proliferative diabetic retinopathy in patients with type 1 and type 2 diabetes: the Japan diabetes complication and its prevention prospective study (JDCP study 4). Diabetol Int. 2018; 10:3–11.

53. Kang EY, Chen TH, Garg SJ, Sun CC, Kang JH, Wu WC, et al. Association of statin therapy with prevention of vision-threatening diabetic retinopathy. JAMA Ophthalmol. 2019; 137:363–71.

54. Shi R, Zhao L, Qi Y. The effect of fenofibrate on early retinal nerve fiber layer loss in type 2 diabetic patients: a case-control study. BMC Ophthalmol. 2018; 18:100.

55. Lin YC, Chen YC, Horng JT, Chen JM. Association of fenofibrate and diabetic retinopathy in type 2 diabetic patients: a population-based retrospective cohort study in Taiwan. Medicina (Kaunas). 2020; 56:385.

56. Meer E, Bavinger JC, Yu Y, VanderBeek BL. Association of fenofibrate use and the risk of progression to vision-threatening diabetic retinopathy. JAMA Ophthalmol. 2022; 140:529–32.

57. Kim NH, Choi J, Kim YH, Lee H, Kim SG. Addition of fenofibrate to statins is associated with risk reduction of diabetic retinopathy progression in patients with type 2 diabetes and metabolic syndrome: a propensity-matched cohort study. Diabetes Metab. 2023; 49:101428.

58. Lim AK. Diabetic nephropathy: complications and treatment. Int J Nephrol Renovasc Dis. 2014; 7:361–81.
59. Keech A, Drury P, Davis TM, Donoghoe M, Laakso M, Whiting M, et al. Abstract 983: protection against nephropathy with fenofibrate in type 2 diabetes mellitus: the FIELD study. Circulation. 2009; 120:S419–20.
60. Sun X, Liu J, Wang G. Fenofibrate decreased microalbuminuria in the type 2 diabetes patients with hypertriglyceridemia. Lipids Health Dis. 2020; 19:103.

61. Yen CL, Fan PC, Lin MS, Lee CC, Tu KH, Chen CY, et al. Fenofibrate delays the need for dialysis and reduces cardiovascular risk among patients with advanced CKD. J Clin Endocrinol Metab. 2021; 106:1594–605.

62. Kim NH, Han KH, Choi J, Lee J, Kim SG. Use of fenofibrate on cardiovascular outcomes in statin users with metabolic syndrome: propensity matched cohort study. BMJ. 2019; 366:l5125.

63. Scott R, O’Brien R, Fulcher G, Pardy C, D’Emden M, Tse D, et al. Effects of fenofibrate treatment on cardiovascular disease risk in 9,795 individuals with type 2 diabetes and various components of the metabolic syndrome: the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study. Diabetes Care. 2009; 32:493–8.
64. Hermans MP. Impact of fenofibrate on type 2 diabetes patients with features of the metabolic syndrome: subgroup analysis from FIELD. Curr Cardiol Rev. 2010; 6:112–8.

65. Yang YS, Kim HL, Kim SH, Moon MK. Lipid management in Korean people with type 2 diabetes mellitus: Korean diabetes association and Korean society of lipid and atherosclerosis consensus statement. Diabetes Metab J. 2023; 47:1–9.

66. Ferraro RA, Leucker T, Martin SS, Banach M, Jones SR, Toth PP. Contemporary management of dyslipidemia. Drugs. 2022; 82:559–76.

67. Koh N, Ference BA, Nicholls SJ, Navar AM, Chew DP, Kostner K, et al. Asian pacific society of cardiology consensus recommendations on dyslipidaemia. Eur Cardiol. 2021; 16:e54.

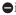 indicates inhibition.
indicates inhibition. 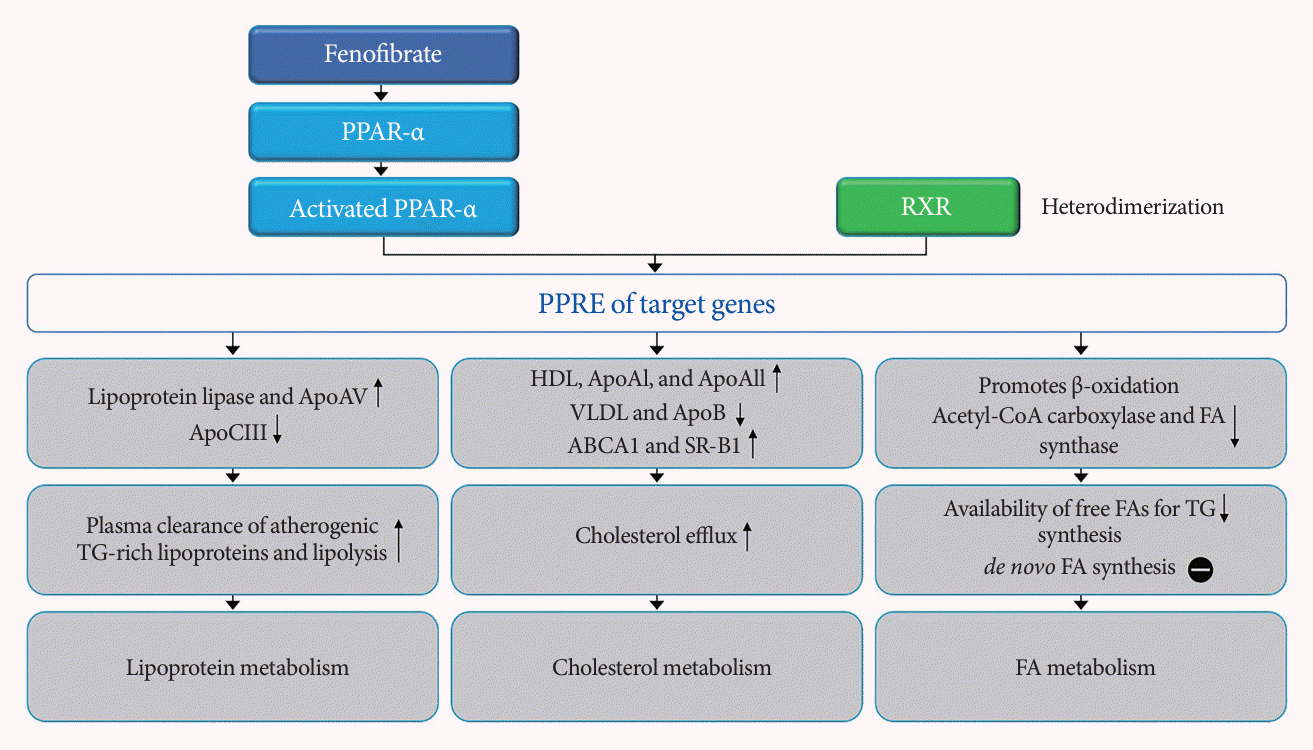




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download