Abstract
Cerebellar hemorrhage in full-term infants is a rare condition recently recognized in high-risk newborns requiring intensive care with the availability of advanced neuroimaging techniques. Several aspects such as the incidence, pathophysiology, clinical features, and prognosis of cerebellar hemorrhage in full-term infants remain unknown. We present a case of cerebellar hemorrhage with subdural hemorrhage in a patient hospitalized for jaundice after birth without a history of traumatic delivery, such as breech presentation, prolonged labor or forceps delivery. A full-term female infant weighing 3,100 g at birth, with no complications during delivery, developed jaundice within 48 hours of birth and was admitted for intensive phototherapy in the first 3 days of life with a transcutaneous total bilirubin level of 18.1 mg/dL. Magnetic resonance imaging revealed cerebellar brain lesions with a subdural hemorrhage. At the age of 3 months, the infant exhibited leg rigidity and was referred for rehabilitation. The patient showed signs of improvement during treatment and was generally catching up well with her peers at the age of 9 months. Long-term follow-ups are required to evaluate the consequences on cognitive development, behavior, and motor performance subsequently in life.
Cerebellar hemorrhage (CBH) is one of the leading causes of cerebellar injury in very preterm infants under 32 weeks of age, although its exact incidence is unknown [1]. As a study using data from several neonatal intensive care centers showed an association between CBH and neurodevelopmental sequelae, the importance of cerebellar damage for long-term poor prognosis, such as academic disability, is increasingly being emphasized [2]. In premature infants experiencing rapid hemodynamic, endocrinologic, and nutritional changes, CBH appears to be associated with germinal matrix hemorrhage-intraventricular hemorrhage in the supratentorial structure [3]. The share mechanism in full-term infants is assumed to be a compressive force to the occiput which causes a contusion or laceration of the underlying cerebellum, making it vulnerable to blood flow and bleeding.
The incidence of CBH in late preterm or full-term infants is not well understood and tends to be underestimated compared to the true incidence because of the relatively low frequency of neuroimaging. CBH in full-term infants may differ from that in premature infants. According to a previous study, traumatic delivery, hypoxic-ischemic encephalopathy (HIE), cardiac anomaly, or non-cardiac anomaly surgery are associated with CBH in full-term infants [4-7]. In particular, this may be caused by direct laceration of the cerebellum or tentorium due to delivery injury or rupture of the bridging vein or occipital sinus in the cerebellar region. As in several case reports, the symptoms caused by CBH in full-term infants show nonspecific features, such as irritability, apnea, and seizures in several case reports. However, they often go undiagnosed due to the lack of obvious comorbidities [5].
There is still a lack of data on CBH in full-term infants because full-term infants undergo brain imaging less frequently when compared to preterm infants. To understand the clinical manifestations and neurological prognosis of cerebral hemorrhage, it is important to collect additional evidence from full-term infants. In this study, we report a case in which a full-term baby was born without any obvious birth injury or history of neonatal asphyxia and showed no clinical symptoms other than severe jaundice within 72 hours of birth. However, CBH was observed on brain magnetic resonance imaging (MRI).
A female newborn was delivered vaginally at a private clinic from a 33-year-old mother at a gestational age of 39 weeks and 1 day. She was the second child with no family history of jaundice. The birth weight was 3,100 g (25th to 50th percentile), height was 47 cm (10th to 25th percentile), and head circumference was 34.5 cm (50th to 75th percentile). The Apgar scores were 10 at 1 minute and 10 at 5 minutes. No specific abnormalities were observed on prenatal examination. According to the medical records of the obstetric clinic where the newborn was born, forceps or vacuum extractors were not used during delivery and there were no obvious birth injuries or perinatal neonatal asphyxia. The patient was healthy and had no family history of genetic diseases. The mother had no maternal diseases during pregnancy. The mother's blood type was Rh+ O, whereas the patient's blood type was Rh+ A.
The neonate remained healthy in a postnatal care center but presented with jaundice on the 2nd day of life. She was admitted to the neonatal intensive care unit for phototherapy, with a transcutaneous total bilirubin level of 18.1 mg/dL during the first 3 days of life. The neonate had stable vital signs: heart rate, 112 beats/min; respiratory rate, 45 breaths/min; systolic blood pressure, 77 mm Hg; diastolic blood pressure 43 mm Hg; mean blood pressure 56 mm Hg; and body temperature, 36.7 °C. Normal neurological findings, normal muscle tone without irritability or lethargy, and no signs of cephalohaematoma or ecchymosis were observed. The peripheral blood test showed a white blood cells count of 10,500/mm3 (neutrophils 67%, lymphocytes 16%, monocytes 13%), hemoglobin value of 17 g/dL, hematocrit value of 50%, platelet count of 268,000/mm3, reticulocytes level of 3.4%, and a normal C-reactive protein level. In serum biochemistry tests, the blood urea nitrogen, creatinine, and electrolyte concentrations were within normal ranges. The total bilirubin and direct bilirubin were increased to 18.85 and 0.80 mg/dL, respectively; additionally, the aspartate transaminase and alanine aminotransferase levels were increased to 192 and 71 U/L, respectively. Both direct and indirect Coombs tests were negative, and venous blood gas analysis and chest radiography were normal. Antibody testing revealed the absence of anti-E and anti-C antibodies in the neonate and mother.
anti-C antibodies in the neonate and mother. The patient underwent intensive phototherapy while preparing for a potential exchange transfusion. After 6 hours of intensive phototherapy, the total bilirubin level decreased to 15.5 mg/dL. The total bilirubin improved to 13.9 mg/dL on the 2nd day of hospitalization under fluid therapy and intensive phototherapy. A mastoid fontanel view was not obtained and no obvious bleeding was observed on brain ultrasonography. Subsequently, while gradually reducing the area for phototherapy, the serum bilirubin level increased slightly to 14.1 mg/dL on the 3rd day of hospitalization, decreased to 11.3 mg/dL on the 5th day of hospitalization, and then increased again to 14.77 mg/dL on the 6th day of hospitalization. During phototherapy, the patient's overall feeding status and activity level were good, and there were no other factors that could explain the pathological jaundice within 72 hours after birth. Therefore, abdominal ultrasonography was performed to differentiate adrenal hemorrhage. In addition, because a slight increase in pressure in the fontanel was suspected and it was necessary to assess the effects of bilirubin deposition in the brain, a brain MRI was performed to differentiate hemorrhagic diseases, such as intracranial and subgaleal hemorrhages, which are difficult to detect using brain ultrasound. Brain MRI included sagittal three-dimensional (3D) T1-weighted and axial T2-weighted imaging (3D T1-weighted repetition time [TR] 8.1 milisecond [ms]; echo time [TE] 3.7 ms; slice thickness 1 mm; field of view [FOV] 180×180 mm and T2-weighted TR 4,800 ms; TE 90 ms, slice thickness 3 mm; FOV 180×180 mm), as well as susceptibility weighted imaging (3D gradient-echo sequence with flow compensation, multishot echo-planar imaging; TR 52 ms; TE 30 ms, slice thickness 2 mm and echo-planar imaging factor). Abdominal ultrasonography showed normal findings; however, brain MRI revealed a subacute subdural hemorrhage in the cerebellar convexity and a subacute intracranial hemorrhage within the cerebellum. The hemorrhagic area was observed focally in the right cerebellum, with a lesion larger than 5 mm in size, and microbleeding was observed on both sides. MRI revealed no bilirubin deposition in specific brain regions (Figure 1). Blood tests were performed to check for coagulopathy on the 12th day after birth. The prothrombin time was 10.8 seconds (international normalized ratio, 1.00) and the activated partial thromboplastin time was 42 seconds, which were all within the normal range. The patient was hospitalized for 10 days, and on the day of discharge, the total bilirubin improved to 8.6 mg/dL.
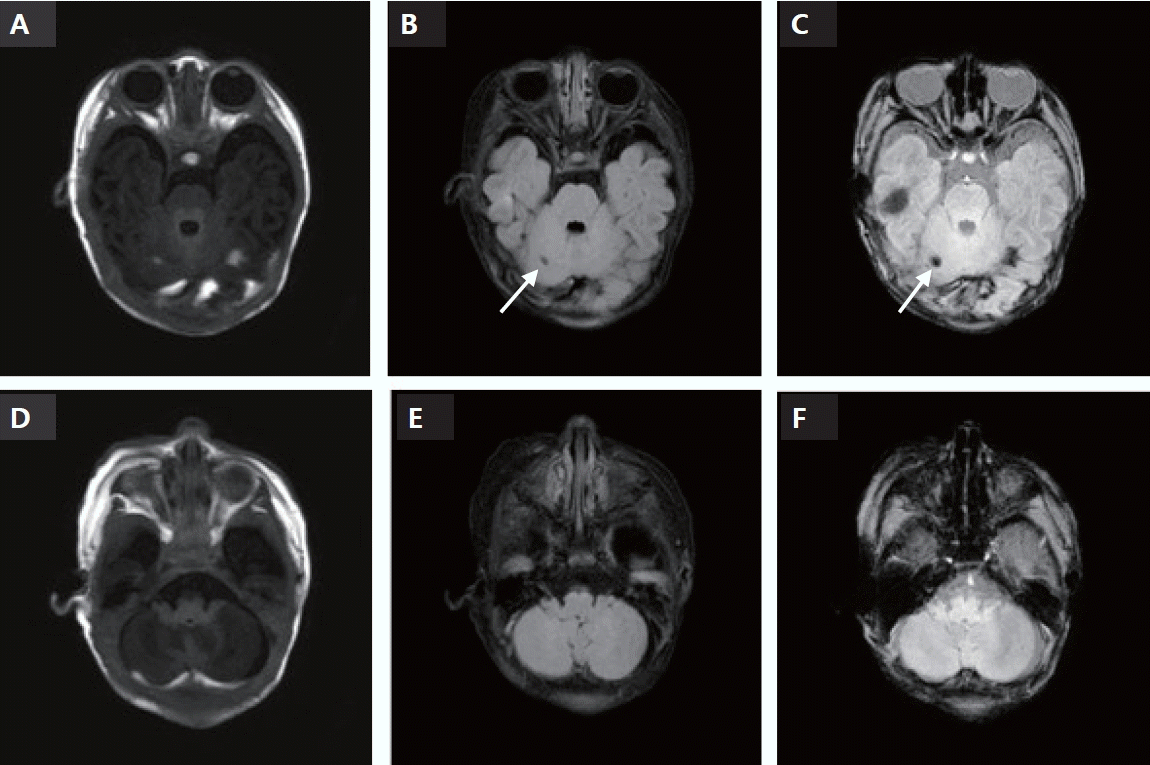
Because this was a rare case of CBH without any birth injury or perinatal asphyxia, genetic tests were performed to rule out the C677T mutation in the 5,10-methylenetetra-hydrofolate reductase gene, which is involved in venous infarction and ischemic stroke in neonates. However, no genetic mutations have been identified. Brain MRI performed at 2-month intervals revealed that the subdural hemorrhage had resolved, and the intracranial hemorrhage showed only old hemorrhagic residual lesions along with hemosiderin deposition (Figure 2). The outpatient followups was performed at the Department of Rehabilitation Medicine to monitor the neurodevelopmental prognosis. At approximately 3 months of age, the patient showed mild motor disability according to the Denver Developmental Screening Test II, and rehabilitation treatment was initiated and maintained until 9 months of age. Roll-over was not possible at approximately 4 months of age but was possible at approximately 6 months of age. The Korean Developmental Screening Test for Infants and Children was performed at approximately 6 months of age and was revealed to be within the normal range or above the peer level.
This case provides evidence that full-term infants with severe jaundice within the first 3 days of life are at risk of developing cerebellar brain lesions with subdural hemorrhage. This was not related to mechanical ventilation, HIE, cardiac malformations, or non-cardiac malformations, which are known risk factors for CBH in full-term infants. The prevalence of subdural hemorrhages in asymptomatic full-term infants has been reported to range from 8% to 46%, predominantly in the posterior fossa [8]. However, the presence of hemosiderin and free iron in the extracerebral space surrounding the cerebellum has been associated with cerebellar growth impairment [9], which may have detrimental effects on brain perfusion and could be particularly harmful in the absence of adequate autoregulation. The etiology and prognosis of CBH remains largely unclear and warrant further investigation to accumulate more data and to recognize its importance.
The causes of pathological jaundice in newborns vary; however, they can be crudely divided into mechanisms that increase the production of bilirubin and those that decrease its clearance [10]. However, it is often multifactorial, and increased bilirubin production is often predominant in newborns with clinically significant jaundice. Non-immune causes that may be related to neonatal jaundice within 48 hours after birth include inherited red blood cell (RBC) defects, RBC enzyme (glucose-6-phosphate dehydrogenase [G6PD], pyruvate kinase) deficiency, neonatal sepsis, and enclosed hemorrhage (cephalohematoma, subgaleal hemorrhage, intracranial hemorrhage, and adrenal hemorrhage) [11,12]. Screening for G6PD deficiency is indicated most often in male patients with a suggestive family history; however, our patient had no such family history. Furthermore, Heinz bodies and polychromasia, which represent denatured hemoglobin and increased RBC production, respectively, were not detected in the peripheral smear. There were no risk factors for neonatal hyperbilirubinemia, suggesting that the CBH in this case may have been related to neonatal jaundice during the early period, at least to a certain extent. Therefore, we concluded that the cause of jaundice was CBH. However, this method has limitations because it is a diagnosis of exclusion.
Predefined prenatal and perinatal risk factors, including neonatal surgery for non-cardiac congenital anomalies [7] and prolonged mechanical ventilation [13], are associated with CBH in full-term infants. Hemorrhagic punctate lesions may arise from fluctuations in cerebral perfusion, including signs of cerebral hypoperfusion and hypocarbia. Impaired cerebral autoregulation has also been observed in full-term infants receiving mechanical ventilation, which may play a role in disturbances in the cerebral blood flow, including fluctuations in the CO2 levels and inflammation [14]. In Korea, Kim et al. [13] studied the clinical features and neurological prognosis of 12 preterm newborns whose CBH was found on brain MRI; high-grade intraventricular hemorrhage, periventricular leukomalacia, and post-hemorrhagic hydrocephalus were found in nine patients. The study included two full-term infants: one full-term infant who underwent brain MRI because a CBH was detected on antenatal ultrasound and another full-term infant who underwent brain ultrasound because of neonatal asphyxia. This may be because of the routine application of the mastoid fontanel brain ultrasound method to view cerebellar lesions in the mastoid fontanel during routine neonatal brain ultrasound examinations in the neonatal intensive care unit of the research institute. They emphasized the importance of the mastoid fontanel view in the diagnosis of CBH in newborns. Merrill et al. [15] reported that they used the routine mastoid fontanel view to enhance posterior fossa imaging and revealed clinically asymptomatic (silent) CBHs which were not evident with the traditional anterior fontanel view. Therefore, we recommend that the mastoid fontanel view should be included when performing brain ultrasound ultrasonography in the neonatal intensive care unit to differentiate occult CBH in newborns.
Kidokoro et al. [16] categorized CBH into four grades on at new grading systems. Grade 1 CBH included single, small punctate lesions smaller than 3 mm on one side; grade 2 included bilateral punctate lesions; grade 3 involved a single lesion larger than 3 mm on one side; and grade 4 was diagnosed when extensive lesions were observed on both sides. In cases where infants presented with two different grades within the same injury category, at higher grade was assigned.
A multicenter study of 218 preterm infants [2] exploring the prognosis of CBH and developmental outcomes retrospectively found that at a corrected age of 2 years, massive lesions were associated with a higher risk of poor outcomes than that of punctate or limited lesion hemorrhages. Additionally, larger CBH lesions were associated with an increased frequency of poor prognosis, which was more pronounced in the group without severe parenchymal brain lesions.
Research on CBH in preterm infants has recently received attention [2,3,17,18]; however, in full-term infants, there are still several unknown aspects, such as the incidence, pathophysiology, clinical features, and prognosis [5,19]. This appears to be because reports of CBH are rare and underestimated compared to the actual incidence, and research is limited because of the lack of data on full-term neonates. If data can be collected through more active imaging tests in full-term infants, previously unknown information, such as the incidence of CBH, clinical features, and neurodevelopmental prognosis, can be discovered. Limperopoulos et al. [5] reported that prenatal and intrapartum factors frequently documented in 23 full-term infants with cerebellar injury included primiparity (55%), advanced maternal age (30%), group B streptococcus-positive mothers (35%), abnormal fetal heart rate (35%), instrumented delivery (30%), and cesarean section (25%). At the follow-up of 18 patients (median age, 32 months), 39% of patients were observed to present with neurological abnormalities.
In this case, a follow-up MRI at an outpatient clinic showed that the subdural hemorrhage had resolved, and the CBH had improved. The patient was found to have increased muscle tension in both extremities. Early intervention was initiated at 3 months of age, with rehabilitation therapy maintained. The patient showed signs of improvement during treatment and was generally catching up well with her peers in terms of development at 9 months of age. Long-term follow-ups are required to evaluate the consequences on cognitive development, behavior, and motor performance later in life.
In conclusion, despite the low prevalence of CBH in full-term infants, the additional value of mastoid fontanel brain ultrasound or brain MRI, especially for the detection of CBH, is considerable in full-term newborns with jaundice, which is difficult to clearly explain.
Notes
Ethical statement
This study was approved by the Institutional Review Board of the Hanyang University Hospital (IRB No. 2021-04-027-004). The Board waived the requirement for informed consent.
REFERENCES
1. Steggerda SJ, Leijser LM, Wiggers-de Bruine FT, van der Grond J, Walther FJ, van Wezel-Meijler G. Cerebellar injury in preterm infants: incidence and findings on US and MR images. Radiology. 2009; 252:190–9.
2. Boswinkel V, Steggerda SJ, Fumagalli M, Parodi A, Ramenghi LA, Groenendaal F, et al. The CHOPIn study: a multicenter study on cerebellar hemorrhage and outcome in preterm infants. Cerebellum. 2019; 18:989–98.
3. Pierson CR, Al Sufiani F. Preterm birth and cerebellar neuropathology. Semin Fetal Neonatal Med. 2016; 21:305–11.
4. Bulas DI, Taylor GA, Fitz CR, Revenis ME, Glass P, Ingram JD. Posterior fossa intracranial hemorrhage in infants treated with extracorporeal membrane oxygenation: sonographic findings. AJR Am J Roentgenol. 1991; 156:571–5.
5. Limperopoulos C, Robertson RL, Sullivan NR, Bassan H, du Plessis AJ. Cerebellar injury in term infants: clinical characteristics, magnetic resonance imaging findings, and outcome. Pediatr Neurol. 2009; 41:1–8.
6. Steggerda SJ, de Bruine FT, Smits-Wintjens VE, Verbon P, Walther FJ, van Wezel-Meijler G. Posterior fossa abnormalities in high-risk term infants: comparison of ultrasound and MRI. Eur Radiol. 2015; 25:2575–83.
7. Stolwijk LJ, Keunen K, de Vries LS, Groenendaal F, van der Zee DC, van Herwaarden MY, et al. Neonatal surgery for noncardiac congenital anomalies: neonates at risk of brain injury. J Pediatr. 2017; 182:335–41.
8. Whitby EH, Griffiths PD, Rutter S, Smith MF, Sprigg A, Ohadike P, et al. Frequency and natural history of subdural haemorrhages in babies and relation to obstetric factors. Lancet. 2004; 363:846–51.
9. Biran V, Verney C, Ferriero DM. Perinatal cerebellar injury in human and animal models. Neurol Res Int. 2012; 2012:858929.
10. Anderson NB, Calkins KL. Neonatal indirect hyperbilirubinemia. Neoreviews. 2020; 21:e749–60.
11. Singh J, Dalal P, Bhalla K, Rattan KN. Neonatal adrenal hemorrhage presenting as prolonged hyperbilirubinemia. J Neonatal Surg. 2016; 5:22.
12. Dennery PA, Seidman DS, Stevenson DK. Neonatal hyperbilirubinemia. N Engl J Med. 2001; 344:581–90.
13. Kim C, Choi GY, Son SH, Kim YJ, Shin SH, Lee JM, et al. The clinical characteristics and the neurodevelopmental outcomes of the neonates with cerebellar hemorrhage. Neonatal Med. 2013; 20:113–20.
14. Berg RM, Plovsing RR. Effects of short-term mechanical hyperventilation on cerebral blood flow and dynamic cerebral autoregulation in critically ill patients with sepsis. Scand J Clin Lab Invest. 2016; 76:226–33.
15. Merrill JD, Piecuch RE, Fell SC, Barkovich AJ, Goldstein RB. A new pattern of cerebellar hemorrhages in preterm infants. Pediatrics. 1998; 102:E62.
16. Kidokoro H, Anderson PJ, Doyle LW, Woodward LJ, Neil JJ, Inder TE. Brain injury and altered brain growth in preterm infants: predictors and prognosis. Pediatrics. 2014; 134:e444–53.
17. Ecury-Goossen GM, Dudink J, Lequin M, Feijen-Roon M, Horsch S, Govaert P. The clinical presentation of preterm cerebellar haemorrhage. Eur J Pediatr. 2010; 169:1249–53.
18. Steggerda SJ, van Wezel-Meijler G. Cranial ultrasonography of the immature cerebellum: role and limitations. Semin Fetal Neonatal Med. 2016; 21:295–304.
19. Williamson WD, Percy AK, Fishman MA, Cheek WR, Desmond MM, LaFevers N, et al. Cerebellar hemorrhage in the term neonate: developmental and neurologic outcome. Pediatr Neurol. 1985; 1:356–60.
Figure 1.
Subacute cerebellar hemorrhage and subacute subdural hemorrhage (SDH) (12 days after birth). (A-C) Arrows indicate the areas of cerebellar hemorrhage. (D-F) Arrows indicate the areas of subdural hemorrahge. (A) cT1: subacute cerebellar hemorrhagic lesion (CBH) on the left and right sides of the cerebellum. (B) cT2: fluid-attenuated inversion recovery (FLAIR). Subacute CBH is found in the same area. (C) Susceptibility weighted imaging with phase enhancement (SWIp): subacute CBH is found in the same area. Microbleeding is found in the SWIp view. (D) cT1: subacute stage SDH along both T-O and cerebellar convexity. (E) cT2: FLAIR. Subacute SDH lesion is found in the same area. (F) SWIp: subacute SDH lesion is found in the same area.
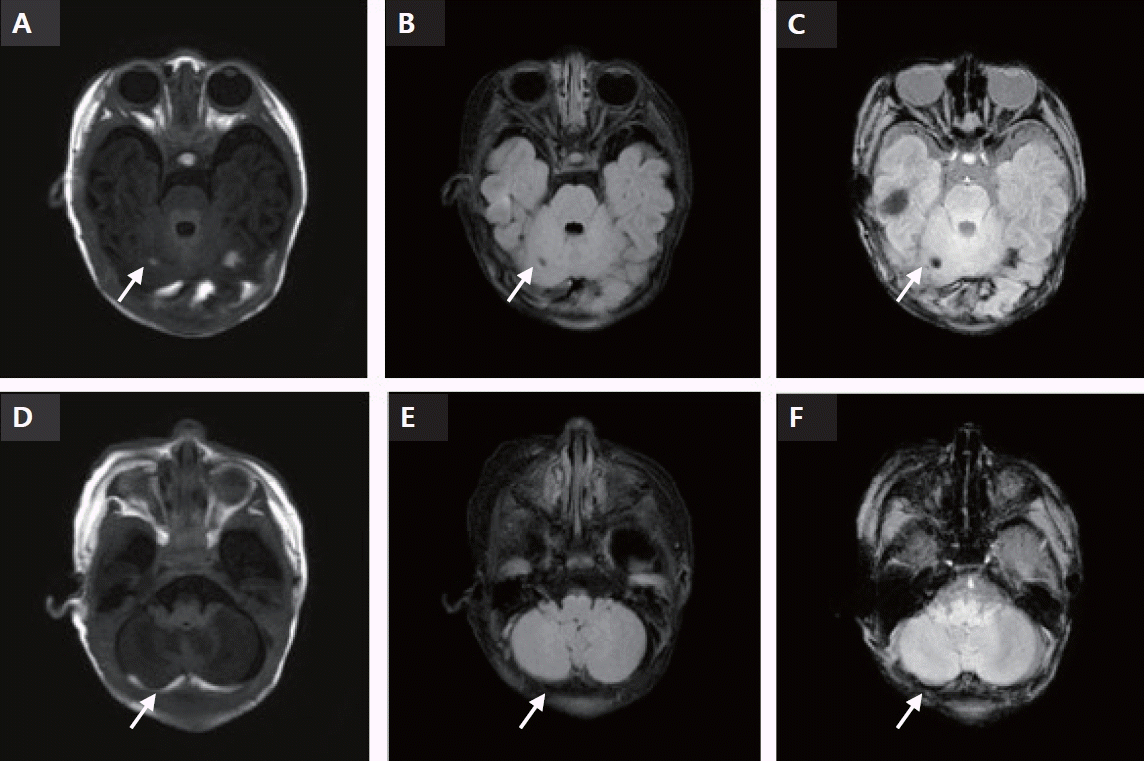
Figure 2.
Follow-up brain magnetic resonance imaging performed 78 days after birth. (A) cT1: hemorrhagic lesion is not visible in the T1 image. (B) cT2: fluid-attenuated inversion recovery (FLAIR). Focal hemosiderin deposit is found in the same area. (C) Susceptibility weighted imaging with phase enhancement (SWIp): focal hemosiderin deposit at right cerebellum and smaller microbleeds at the cerebellum and the left occipital lobe. (B, C) Arrows indicate the areas of hemosiderin deposit. (D) cT1: resolved subdural hemorrhage (SDH) along both T-O convexity and cerebellar convexity. (E) cT2: FLAIR. Resolved SDH in the same area. (F) SWIp: resolved SDH in the same area.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download