1. Guanciali-Franchi P, Iezzi I, Soranno A, de Volo CP, Alfonsi M, Calabrese G, et al. Optimal cut-offs for Down syndrome contingent screening in a population of 10,156 pregnant women. Prenat Diagn. 2012; 32(12):1147–1150. PMID:
23007955.

2. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 88, December 2007. Invasive prenatal testing for aneuploidy. Obstet Gynecol. 2007; 110(6):1459–1467. PMID:
18055749.
3. Spencer K, Souter V, Tul N, Snijders R, Nicolaides KH. A screening program for trisomy 21 at 10-14 weeks using fetal nuchal translucency, maternal serum free beta-human chorionic gonadotropin and pregnancy-associated plasma protein-A. Ultrasound Obstet Gynecol. 1999; 13(4):231–237. PMID:
10341399.

4. Wøjdemann KR, Shalmi AC, Christiansen M, Larsen SO, Sundberg K, Brocks V, et al. Improved first-trimester Down syndrome screening performance by lowering the false-positive rate: a prospective study of 9941 low-risk women. Ultrasound Obstet Gynecol. 2005; 25(3):227–233. PMID:
15736185.

5. Baer RJ, Flessel MC, Jelliffe-Pawlowski LL, Goldman S, Hudgins L, Hull AD, et al. Detection rates for aneuploidy by first-trimester and sequential screening. Obstet Gynecol. 2015; 126(4):753–759. PMID:
26348180.

6. Malone FD, Canick JA, Ball RH, Nyberg DA, Comstock CH, Bukowski R, et al. First-trimester or second-trimester screening, or both, for Down’s syndrome. N Engl J Med. 2005; 353(19):2001–2011. PMID:
16282175.

7. Dashe JS. Aneuploidy screening in pregnancy. Obstet Gynecol. 2016; 128(1):181–194. PMID:
27275786.

8. Spencer K, Ong CY, Liao AW, Nicolaides KH. The influence of ethnic origin on first trimester biochemical markers of chromosomal abnormalities. Prenat Diagn. 2000; 20(6):491–494. PMID:
10861715.

9. Liao AW, Heath V, Kametas N, Spencer K, Nicolaides KH. First-trimester screening for trisomy 21 in singleton pregnancies achieved by assisted reproduction. Hum Reprod. 2001; 16(7):1501–1504. PMID:
11425838.

10. Spencer K, Heath V, El-Sheikhah A, Ong CY, Nicolaides KH. Ethnicity and the need for correction of biochemical and ultrasound markers of chromosomal anomalies in the first trimester: a study of Oriental, Asian and Afro-Caribbean populations. Prenat Diagn. 2005; 25(5):365–369. PMID:
15906426.

11. Kagan KO, Frisova V, Nicolaides KH, Spencer K. Dose dependency between cigarette consumption and reduced maternal serum PAPP-A levels at 11-13+6 weeks of gestation. Prenat Diagn. 2007; 27(9):849–853. PMID:
17590888.

12. Spencer K, Bindra R, Nicolaides KH. Maternal weight correction of maternal serum PAPP-A and free beta-hCG MoM when screening for trisomy 21 in the first trimester of pregnancy. Prenat Diagn. 2003; 23(10):851–855. PMID:
14558032.

13. Spencer K, Cicero S, Atzei A, Otigbah C, Nicolaides KH. The influence of maternal insulin-dependent diabetes on fetal nuchal translucency thickness and first-trimester maternal serum biochemical markers of aneuploidy. Prenat Diagn. 2005; 25(10):927–929. PMID:
16088864.

14. Spencer K, Staboulidou I, De Jesus Cruz J, Karagiannis G, Nicolaides KH. Maternal serum screening marker levels in women with a previous aneuploidy pregnancy. Prenat Diagn. 2009; 29(13):1242–1243. PMID:
19842139.

15. Dickey RP, Taylor SN, Lu PY, Sartor BM, Storment JM, Rye PH, et al. Spontaneous reduction of multiple pregnancy: incidence and effect on outcome. Am J Obstet Gynecol. 2002; 186(1):77–83. PMID:
11810089.

16. Evron E, Sheiner E, Friger M, Sergienko R, Harlev A. Vanishing twin syndrome: is it associated with adverse perinatal outcome? Fertil Steril. 2015; 103(5):1209–1214. PMID:
25772775.

17. Kim SY, Lee SM, Jun JK, Han YJ, Kim MH, Shim JY, et al. Prospective observations study protocol to investigate cost-effectiveness of various prenatal test strategies after the introduction of noninvasive prenatal testing. BMC Pregnancy Childbirth. 2018; 18(1):307. PMID:
30041617.

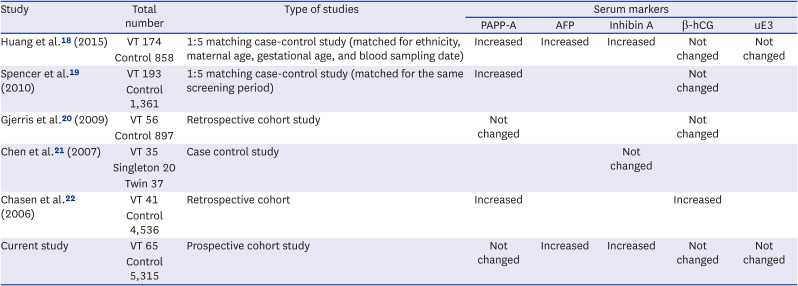
18. Huang T, Boucher K, Aul R, Rashid S, Meschino WS. First and second trimester maternal serum markers in pregnancies with a vanishing twin. Prenat Diagn. 2015; 35(1):90–96. PMID:
25200921.

19. Spencer K, Staboulidou I, Nicolaides KH. First trimester aneuploidy screening in the presence of a vanishing twin: implications for maternal serum markers. Prenat Diagn. 2010; 30(3):235–240. PMID:
20066674.

20. Gjerris AC, Loft A, Pinborg A, Christiansen M, Tabor A. The effect of a ‘vanishing twin’ on biochemical and ultrasound first trimester screening markers for Down’s syndrome in pregnancies conceived by assisted reproductive technology. Hum Reprod. 2009; 24(1):55–62. PMID:
18835870.

21. Chen HJ, Huang LW, Lin YH, Seow KM, Hsieh BC, Hwang JL, et al. Midtrimester maternal serum inhibin A levels after multifetal pregnancy reduction. Prenat Diagn. 2007; 27(5):431–434. PMID:
17295349.

22. Chasen ST, Perni SC, Predanic M, Kalish RB, Chervenak FA. Does a “vanishing twin” affect first-trimester biochemistry in Down syndrome risk assessment? Am J Obstet Gynecol. 2006; 195(1):236–239. PMID:
16626615.

23. Pitman EJ. Significance tests which may be applied to samples from any populations. II. The correlation coefficient test. Suppl J R Stat Soc. 1937; 4(2):225–232.

24. Corsello G, Piro E. The world of twins: an update. J Matern Fetal Neonatal Med. 2010; 23(Suppl 3):59–62. PMID:
20807162.
25. Krantz DA, Hallahan TW, Macri VJ, Macri JN. Maternal weight and ethnic adjustment within a first-trimester Down syndrome and trisomy 18 screening program. Prenat Diagn. 2005; 25(8):635–640. PMID:
16049986.
26. Spaggiari E, Ruas M, Dreux S, Valat AS, Czerkiewicz I, Guimiot F, et al. Management strategy in pregnancies with elevated second-trimester maternal serum alpha-fetoprotein based on a second assay. Am J Obstet Gynecol. 2013; 208(4):303.e1–303.e7.

27. Anfuso S, Soncini E, Bonelli P, Piantelli G, Gramellini D. Second-trimester maternal serum alpha-fetoprotein elevation and its association with adverse maternal/fetal outcome: ten years experience. Acta Bio-med Ateneo Parm. 2007; 78(3):214–219.
28. Hsieh TT, Hung TH, Hsu JJ, Shau WY, Su CW, Hsieh FJ. Prediction of adverse perinatal outcome by maternal serum screening for Down syndrome in an Asian population. Obstet Gynecol. 1997; 89(6):937–940. PMID:
9170469.

29. Waller DK, Lustig LS, Cunningham GC, Golbus MS, Hook EB. Second-trimester maternal serum alpha-fetoprotein levels and the risk of subsequent fetal death. N Engl J Med. 1991; 325(1):6–10. PMID:
1710779.

30. Harris R, Jennison RF, Barson AJ, Laurence KM, Ruoslahti E, Seppälä M. Comparison of amniotic-fluid and maternal serum alpha-fetoprotein levels in the early antenatal diagnosis of spina bifida and anencephaly. Lancet. 1974; 1(7855):428–429. PMID:
4131431.

31. Bass HN, Oliver JB, Srinivasan M, Petrucha R, Ng W, Lee JE. Persistently elevated AFP and AChE in amniotic fluid from a normal fetus following demise of its twin. Prenat Diagn. 1986; 6(1):33–35. PMID:
2419890.

32. Streit JA, Penick GD, Williamson RA, Weiner CP, Benda JA. Prolonged elevation of alphafetoprotein and detectable acetylcholinesterase after death of an anomalous twin fetus. Prenat Diagn. 1989; 9(1):1–6. PMID:
2473466.

33. Grau P, Robinson L, Tabsh K, Crandall BF. Elevated maternal serum alpha-fetoprotein and amniotic fluid alpha-fetoprotein after multifetal pregnancy reduction. Obstet Gynecol. 1990; 76(6):1042–1045. PMID:
1700349.
34. Florio P, Luisi S, Ciarmela P, Severi FM, Bocchi C, Petraglia F. Inhibins and activins in pregnancy. Mol Cell Endocrinol. 2004; 225(1-2):93–100. PMID:
15451573.

35. Crandall BF, Lau HL. Alpha-fetoprotein: a review. Crit Rev Clin Lab Sci. 1981; 15(2):127–185. PMID:
6169490.

36. Abbas A, Johnson M, Bersinger N, Nicolaides K. Maternal alpha-fetoprotein levels in multiple pregnancies. Br J Obstet Gynaecol. 1994; 101(2):156–158. PMID:
7508260.

37. Muttukrishna S, Child TJ, Groome NP, Ledger WL. Source of circulating levels of inhibin A, pro alpha C-containing inhibins and activin A in early pregnancy. Hum Reprod. 1997; 12(5):1089–1093. PMID:
9194671.

38. Chang CS, Choi Y, Kim SY, Yee C, Kim M, Sung JH, et al. Prenatal ultrasonographic findings of esophageal atresia: potential diagnostic role of the stomach shape. Obstet Gynecol Sci. 2021; 64(1):42–51. PMID:
33285619.

39. Ko HS, Kwak DW, Oh SY, Choi SK, Hong JS, Hwang HS, et al. Clinical significance of soft markers in second trimester ultrasonography for pregnant Korean women: a multicenter study and literature review. Obstet Gynecol Sci. 2022; 65(2):145–155. PMID:
35184524.







 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download