Abstract
Background
Refractory angina pectoris (RAP) is a chronic, severe chest pain associated with coronary artery disease that cannot be resolved using optimal medical or surgical approaches. Spinal cord stimulation (SCS) is a suitable treatment option. Conventional waveforms of SCS have shown a potent effect on the tempering of RAP. However, SCS is associated with undesired paresthesia. The new burst SCS waveforms have been reported to have fewer adverse effects.
Go to : 
The incidence and prevalence of coronary artery diseases have steadily increased over the past ten years. Pharmacological medications, percutaneous coronary intervention (PCI), and coronary artery bypass grafting (CABG) have been used to treat these diseases. Chronic severe chest pain caused by coronary artery disease, which does not improve with these treatments, is known as refractory angina pectoris (RAP). RAP reduces the quality of life and interferes with physical activity. However, reliable therapy for RAP has not yet been found [1,2].
Spinal cord stimulation (SCS) is a possible treatment approach [1,2]. Although the conventional SCS waveform shows remarkable potential for managing RAP [3], it can cause undesired paresthesia and may not be effective for centralized pain. The recently developed burst waveform of SCS is considered a good alternative for addressing these challenges. It can suppress nociceptive and neuropathic pain by modulating both the medial and lateral pain pathways. In addition, it has been reported to provide better pain relief with a lower incidence of undesired paresthesia [4].
However, to our knowledge, few case reports have applied the burst waveform of SCS to RAP worldwide [5]. Thus, we present a case where the burst waveform of SCS was successfully applied to RAP.
A 52-year-old male with hypertension and diabetes mellitus visited the Department of Cardiology complaining of chest pain. The pain started one month prior and was worsening, with the patient reporting 8 points on the numeric rating scale (NRS, 0 = no pain, 10 = maximum pain). Coronary angiography (CAG) revealed 90% stenosis of the distal right coronary artery (dRCA). PCI was successfully performed on the dRCA, resulting in pain improvement.
However, despite the treatment, the patient revisited the hospital several times because of recurrent chest pain. As a result, CAGs were performed up to seven times over the following three years. Nevertheless, none of the CAG showed any significant interval changes. Moreover, none of the medications that had been administered thus far, including aspirin, clopidogrel, ticagrelor, trimetazidine, nicorandil, candesartan, bisoprolol, nebivolol, ezetimibe, and rosuvastatin, were effective. Thus, the cardiologist concluded that the pain was no longer of cardiac origin.
The patient was then referred to the Department of Anesthesiology and Pain Medicine. The pain was continuous, tightening, and dull, mainly in the left chest area, and radiated to the left shoulder (NSR, 3–4). However, he also had an irregular, intermittent episode of severe pain that lasted for 4–10 h, approximately twice a week (NRS, 10). No aggravating or alleviating factors were identified.
Despite administering painkillers, including acetaminophen, nonsteroidal anti-inflammatory drugs, tramadol, opioids, gabapentin, buprenorphine, and two stellate ganglion blocks, the pain did not subside. Thus, the pain was considered refractory to any treatment modality, including pharmacological medications, PCI, and nerve blocks. Therefore, we planned for SCS as a viable therapeutic alternative.
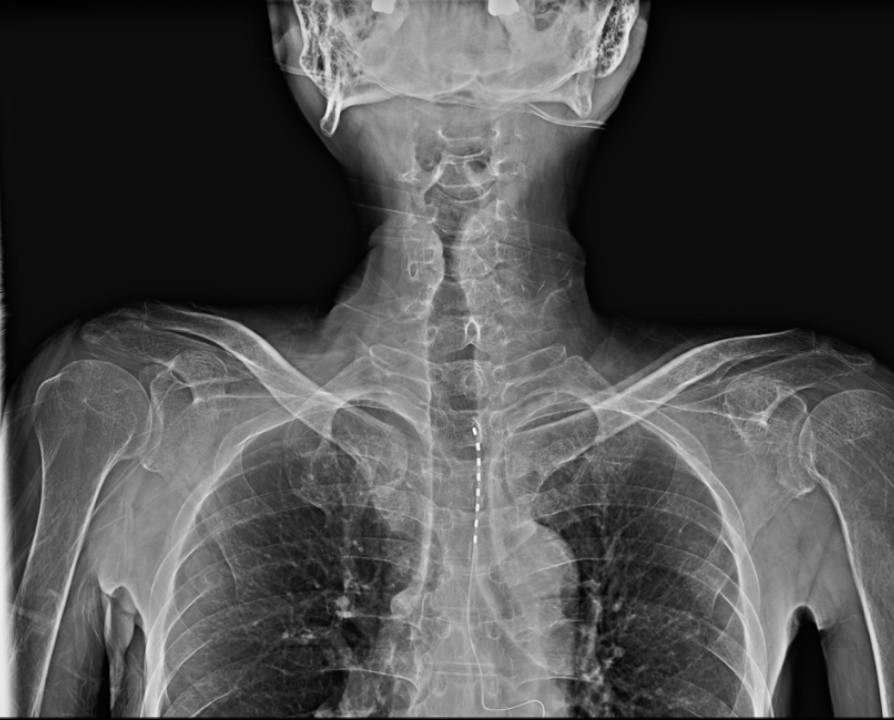
For trial stimulation, a 14-G epidural needle was inserted at the thoracic vertebra (T) 6–7 level under fluoroscopic guidance, through which an octa-lead was advanced until the electrode reached the T1–2 level (Fig. 1). The electrode was then connected to an external impulse generator, and the stimulus of the tonic waveform was applied to provoke symptoms at the pain site.
In the 14-day trial program, both tonic (frequency of 20 or 30 Hz, pulse width of 500 µs, and amplitude of 1.0–2.0 mA) and burst stimulation (five pulses at 500 Hz, 40 bursts/s, and amplitude of 0.3 mA) were tested. Although the patient reported similar reduction in background pain with both waveforms (NRS, 2–3), only the tonic waveform effectively alleviated the two episodes of breakthrough chest pain (NRS, 7–8). Therefore, he was more satisfied with the immediate response to the severe breakthrough chest pain.
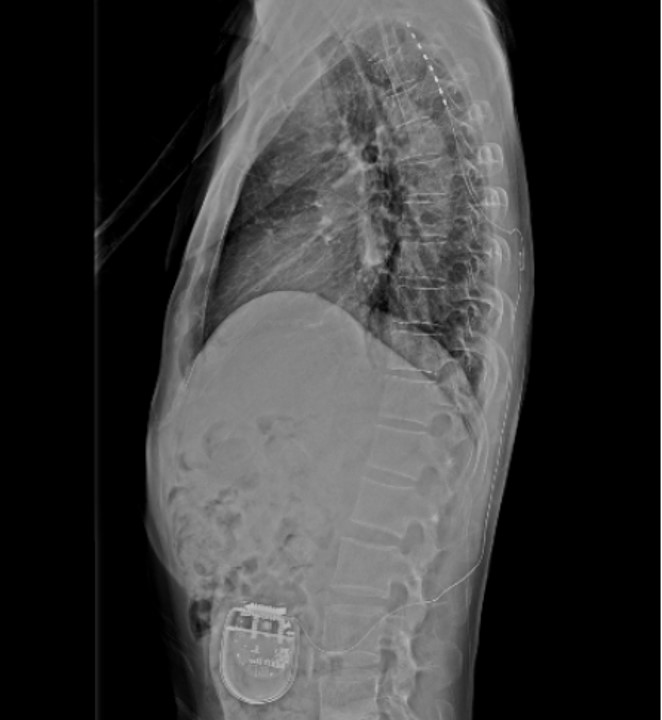
Therefore, permanent implantation was performed. A permanent pulse generator (Proclaim XR, Abbott) was implanted subcutaneously into the left lower quadrant of the abdomen (Fig. 2).
However, the patient complained of a persistent tingling sensation. Thus, we decided to use the burst waveform for background pain and switch to the tonic waveform in the event of sudden severe pain (the patient was instructed to change the waveform on his own). The results of this treatment approach were successful.
In contrast, when the amplitude of tonic stimulation was increased from 1.0–2.0 mA to 2.0–3.0 mA, breakthrough pain diminished substantially without notable side effects. However, when the amplitude of burst stimulation was increased from 0.3 mA to 0.4 mA, background pain decreased to near zero, but headache occurred. Therefore, with the amplitude of tonic stimulation increased to 2.0–3.0 mA and the amplitude of burst stimulation maintained at 0.3 mA, the patient’s pain could be managed effectively.
Three months after discharge, the two modes (tonic and burst) were appropriately applied as required, and both background and breakthrough pain were well controlled. The patient also reported that the incidence of sudden severe pain had decreased from approximately twice per week to approximately twice per month. In addition, even when the pain occurred, he reported that the intensity decreased to NRS 7–8 from the previous NRS 10. Even in this case, when tonic waveform was applied, the pain quickly disappeared; therefore, the patient did not require painkillers and did not revisit the emergency room after the treatment.
Go to : 
This case report is the first application of the burst waveform of SCS for RAP in South Korea. Although the successful application of the tonic waveform of SCS to RAP has been previously reported, only a few cases of successful application of the burst waveform of SCS have been reported worldwide.
SCS was developed based on the gate control theory of pain, which states that continuous stimulation of Aβ-fibers in the dorsal columns can inhibit C-fiber responses in the dorsal horn neurons [6]. SCS induces local alteration of the excitability of wide-dynamic-range (WDR) neurons, promotes physiological inhibitory mechanisms, alters the activity of several neurotransmitters, and decreases sympathetic activity [7,8].
The conventional waveform of SCS is the tonic waveform, in which a certain stimulus amplitude is repeatedly given at a fixed time interval (in this case, frequency, 20 or 30 Hz; pulse width, 500 µs; and amplitude, 2.0–3.0 mA). In contrast, in the newly developed burst waveform of SCS, a few consecutive stimuli of a relatively low amplitude are given, followed by a pause of no stimulus. Subsequently, the stimuli cluster is repeated as before (in this case, five pulses at 500 Hz, 40 bursts/s, and an amplitude of 0.3 mA). In the burst waveform of SCS, if the total energy of the stimuli is the same as that of stimuli in the tonic waveform, the amplitude of each stimulus becomes smaller (weaker energy) than that of the tonic waveform, and the number of stimuli increases. It may be hypothesized that a smaller amplitude of each stimulus accounts for the lower incidence of paresthesia and provocation in the burst waveforms.
Although burst stimulation affects the dorsal column and gamma-aminobutyric acid receptors less and induces less paresthesia, it can inhibit the firing of WDR neurons at the dorsal horn to a greater degree than the inhibition observed with tonic stimulation [9]. In addition, unlike tonic stimulation, which modulates only the lateral pain pathway, burst stimulation modulates both the medial and lateral pain pathways, affecting the neural activity in the somatosensory and anterior cingulate cortices. This implies that it affects the somatic aspect of pain and its emotional components, exerting a better pain-relief effect [10].
SCS is an effective treatment approach for relieving ischemic pain in angina. First, pain can be relieved by reducing the stimulation of the dorsal columns and transmission of nociceptive impulses through the spinothalamic tract. Additionally, SCS directly improves ischemic conditions, supposedly playing an important role in inducing vasodilation by releasing calcitonin gene-related peptides and reducing myocardial oxygen demand with lowered sympathetic tone. In addition, the imbalance between oxygen supply and demand can be adjusted by improving the coronary blood flow at the microvascular level. Thus, ischemia can be improved with subsequent pain relief. Finally, SCS exerts an anti-ischemic function by inhibiting excessive intrinsic cardiac nervous system activity, which increases the incidence of arrhythmia or ischemia, reduces sympathetic activity, and redistributes myocardial blood flow from the non-ischemic to ischemic areas [11].
The efficacy of SCS in patients with angina has also been reported. According to a study conducted on CABG and SCS in patients with high-risk angina, no significant difference between SCS and CABG in pain reduction was noted, and more favorable results were reported with SCS for postoperative mortality and cerebrovascular events [1]. Additionally, a small difference in the effectiveness of SCS and percutaneous myocardial laser revascularization was noted in patients with refractory angina [2]. Therefore, SCS is recommended as a treatment option for refractory angina by the European Society of Cardiology (class IIb) and American College of Cardiology and American Heart Association (class IIb) guidelines [12].
Since SCS was first used as a treatment approach for angina in 1987, many successful applications have been reported [1,2,13]. However, most cases involve tonic stimulation, and burst stimulation has been reported in only one case [5]. In the abovementioned case, the pain-relief effect of tonic stimulation was approximately 30%; however, it increased to > 80% after burst stimulation.
As burst stimulation has been applied to refractory angina in only a few cases, it suggests that its efficacy and mechanism have limitations. However, for pain types other than breakthrough pain, burst stimulation that does not induce paresthesia may relieve pain without causing discomfort or a tingling sensation. Regarding pain relief, angina pain is thought to be caused by a combination of nociceptive and neuropathic pain. Tonic stimulation is primarily effective for neuropathic pain. However, burst stimulation has been reported to be additionally effective for nociceptive pain, rendering it superior to tonic stimulation. Further, burst stimulation modulates both the medial and lateral pathways of pain. Therefore, burst stimulation may be more effective in pain control in angina, as well as in the emotional and somatosensory aspects.
Studies have reported that burst stimulation has a better pain-relief effect than tonic stimulation in patients with failed back surgery syndrome and diabetic neuropathic pain [14]. However, previous studies have rarely addressed the difference between the efficacies of the burst and tonic modes in refractory angina. Therefore, further studies on applying burst stimulation for refractory angina are warranted.
In conclusion, SCS may be a promising treatment approach for patients with RAP without laboratory abnormalities. Additionally, as reported in this case, the appropriate use of tonic and burst stimulations according to symptoms is expected to maximize the effect of relieving chest pain induced by RAP and improve the patient’s quality of life.
Go to : 
Notes
DATA AVAILABILITY STATEMENT
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
AUTHOR CONTRIBUTIONS
Conceptualization: Jae Chul Koh. Writing - original draft: Daehun Yun. Writing - review & editing: JI Soo Kwak. Supervision: Kaehong Lee, Leegyeong Je, Taesan Kim, Yoon Sun Park.
Go to : 
REFERENCES
1. Mannheimer C, Eliasson T, Augustinsson L-E, Blomstrand C, Emanuelsson Hk, Larsson S, et al. Electrical stimulation versus coronary artery bypass surgery in severe angina pectoris: the ESBY study. Circulation. 1998; 97:1157–63.

2. McNab D, Khan SN, Sharples LD, Ryan JY, Freeman C, Caine N, et al. An open label, single-centre, randomized trial of spinal cord stimulation vs. percutaneous myocardial laser revascularization in patients with refractory angina pectoris: the SPiRiT trial. Eur Heart J. 2006; 27:1048–53.

3. Murphy DF, Giles KE. Dorsal column stimulation for pain relief from intractable angina pectoris. Pain. 1987; 28:365–8.

4. De Ridder D, Vanneste S, Plazier M, Van der Loo E, Menovsky T. Burst spinal cord stimulation: toward paresthesia-free pain suppression. Neurosurgery. 2010; 66:986–90.
5. Abusabha Y, Slotty PJ. Successful burst spinal cord stimulation in refractory angina. Neuromodulation. 2019; 22:229–30.

6. Moayedi M, Davis KD. Theories of pain: from specificity to gate control. J Neurophysiol. 2013; 109:5–12.

7. Guan Y. Spinal cord stimulation: neurophysiological and neurochemical mechanisms of action. Curr Pain Headache Rep. 2012; 16:217–25.

8. Foreman RD, Linderoth B. Neural mechanisms of spinal cord stimulation. Int Rev Neurobiol. 2012; 107:87–119.

9. Chakravarthy K, Fishman MA, Zuidema X, Hunter CW, Levy R. Mechanism of action in burst spinal cord stimulation: review and recent advances. Pain Med. 2019; 20(Suppl 1):S13–S22.

10. Hou S, Kemp K, Grabois M. A systematic evaluation of burst spinal cord stimulation for chronic back and limb pain. Neuromodulation. 2016; 19:398–405.

11. Latif OA, Nedeukovic SS, Stevenson LW. Spinal cord stimulation for chronic intractable angina pectoris: a unified theory on its mechanism. Clin Cardiol. 2001; 24:533–41.

12. Fihn SD, Gardin JM, Abrams J, Berra K, Blankenship JC, Dallas AP, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 2012; 126:e354–e471.
Go to : 




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download