Abstract
Backgrounds/Aims
Laparoscopic cholecystectomy within one week of acute cholecystitis is considered safe and advantageous. Surgery beyond first week is reserved for non-resolving attack or complications. To compare clinical outcomes of patients undergoing laparoscopic cholecystectomy in the first week and between two to six weeks of an attack of acute cholecystitis.
Methods
In an analysis of a prospectively maintained database, all patients who underwent laparoscopic cholecystectomy for acute cholecystitis were divided into two groups: group A, operated within one week; and group B, operated between two to six weeks of an attack. Main variables studied were mean operative time, conversion to open cholecystectomy, morbidity profile, and duration of hospital stay.
Results
A total of 116 patients (74 in group A and 42 in group B) were included. Mean interval between onset of symptoms & surgery was five days (range, 1–7 days) in group A and 12 days (range, 8–20 days) in group B. Operative time and incidence of subtotal cholecystectomy were higher in group B (statistically not significant). Mean postoperative stay was 2 days in group A and 3 days in group B. Laparoscopy was converted to open cholecystectomy in two patients in each group. There was no incidence of biliary injury. One patient in group B died during the postoperative period due to continued sepsis and multiorgan failure.
In the early days, acute cholecystitis (AC) was listed as a contraindication to laparoscopic cholecystectomy (LC) [1]. Presence of acute inflammation, difficulty in dissection, and higher chances of complications were initial apprehensions. With increasing experience, it is now well established that LC is safe. In addition, it is more advantageous than open cholecystectomy [2]. Surgery was recommended within 72 hours of attack. This time frame was called the ‘golden period.’ It was subsequently realized that the limit of ‘early cholecystectomy’ could be safely stretched up to one week to 10 days, which formed the basis of the most recent guidelines [3-8]. LC in the second week is considered formidable. Therefore, LC is recommended to be deferred beyond six weeks called ‘late cholecystectomy.’ This is done to allow inflammation to subside so that the procedure can be performed electively. In the intervening period (i.e., from second to sixth week, cholecystectomy is performed for non-resolving cholecystitis or complications such as empyema, gangrene, and perforation). During this period, surgery is perceived to be technically difficult with attendant high conversion & complication rates. Since our center is a tertiary care center, we often have to operate on such patients referred to us beyond the first week. With considerable experience in laparoscopic surgery, we are able to salvage most of these cases successfully. Encouraged by our results, we decided to subject all our AC patients who were fit for general anesthesia to LC irrespective of the time frame based on the logic that if it could be performed successfully under compelling circumstances, it could be attempted in a less demanding situation. A prospectively maintained database of all such patients with AC was analyzed with an aim to evaluate operative issues and immediate outcome of LC performed in the first week and beyond. This study was approved by Apollo Hospitals’ ethical committee (No. ECR/246/Inst/OR/2013/RR-2016).
In a prospectively maintained database, all patients admitted between May 2017 and February 2020 with the diagnosis of AC and operated within six weeks of the attack were included in this study. Patients were divided into two groups: group A, patients operated within one week of onset of an attack of AC (early cholecystectomy); and group B, those operated between two to six weeks (intermediate cholecystectomy). The allocation into respective groups was retrospective and non-randomized. It was based on the timing of presentation only. We followed Tokyo guidelines for the diagnosis of AC that was based on clinical presentation, ultrasound imaging, and hematological parameters [9]. Contrast enhanced computed tomography (CT) scan was done for doubtful diagnosis or suspected complications such as perforation or gangrene. Magnetic resonance cholangiopancreatography was performed for suspected common bile duct (CBD) stone(s). Charlson Comorbidity Index (CCI) and American Society of Anaesthesiologists Physical Status (ASA-PS) classification system were used for risk assessment [10,11]. Patients with CCI ≥ 4 and/or ASA ≥ 3 were excluded from this study and managed conservatively or by imaging guided percutaneous cholecystostomy. Surgery was performed for these patients electively after their conditions improved. In case of non-response or deterioration despite adequate conservative treatment, LC was offered as a last resort. All patients received broad spectrum antibiotics (cefoperazone and sulbactam) in the perioperative period. Patients with choledocholithiasis were treated with single sitting LC followed by endoscopic papillotomy and CBD stone clearance.
Our center is a tertiary care referral center with exclusive interest in hepatobiliary surgery. The senior most surgeon has over 25 years’ experience in laparoscopic surgery. LC was performed by him directly or under his supervision. The operative time was recorded to grade technical difficulty. Less than one hour, between one to two hours, and more than two hours were considered as technically easy, moderately difficult, and difficult, respectively.
A standard four port approach was adopted. An additional 5 mm port was sometimes used to retract bowel or an overhanging liver and to improve exposure. The fundus of the gallbladder (GB) if covered by adhesions was exposed with a gentle sweep using 5 mm suction cannula. The distended GB was aspirated by using a thick bore needle. The GB fundus was grasped with tooth grasper and firmly retracted up. Adhesions on the GB wall were gently swept with a suction cannula. Any stone impacted in Hartman’s pouch was dislodged if possible and pushed up. Once the Hartman’s pouch was exposed, it was grasped with a stout tooth grasper. Posterior adhesions were separated gently till it could be freely lifted up. The hepatocystic triangle was dissected to isolate cystic duct (CD) and artery after demonstrating critical view of safety (CVS) [12]. If that was not possible, the following techniques were adopted. The cystic artery was first isolated lateral to the cystic lymph node close to the GB, clipped, and divided. This could facilitated dissection of the CVS. If that too was not possible either, we attempted antegrade (fundus-first) technique. The dissection was kept close to the GB as it was mobilized from its bed. The cystic duct was the last structure to be divided after defining and circumferentially dissecting the infundibulum of the GB. We used Hem-o-lok clips (Weck Closure Systems) to secure CD if it appeared too short or wide. In certain extremely difficult situations, we incised the GB neck to deliver the impacted stone, caught hold of the incised margin, and again tried to dissect structures (CD and artery). If unsuccessful, the Hartman’s pouch was flushed with saline and its interior was inspected for any leftover stone fragment(s). The margin was then trimmed, leaving behind just enough healthy tissue for safe approximation with interrupted Vicryl suture, close to the presumed CD opening (subtotal cholecystectomy, reconstituting type) [13]. The excised GB was always delivered through umbilical port using endo-bag. In situations where the dissection appeared hazardous due to dense adhesions and/or obscure anatomy, we chose to convert to open cholecystectomy. We routinely placed suction drain in Hartman’s pouch. When the CD was satisfactorily secured with clips, the drain was removed after 24 to 48 hours or the patient was discharged with the drain removed one week later when he/she returned for suture removal. Fisher exact test was used to compare results between two groups.
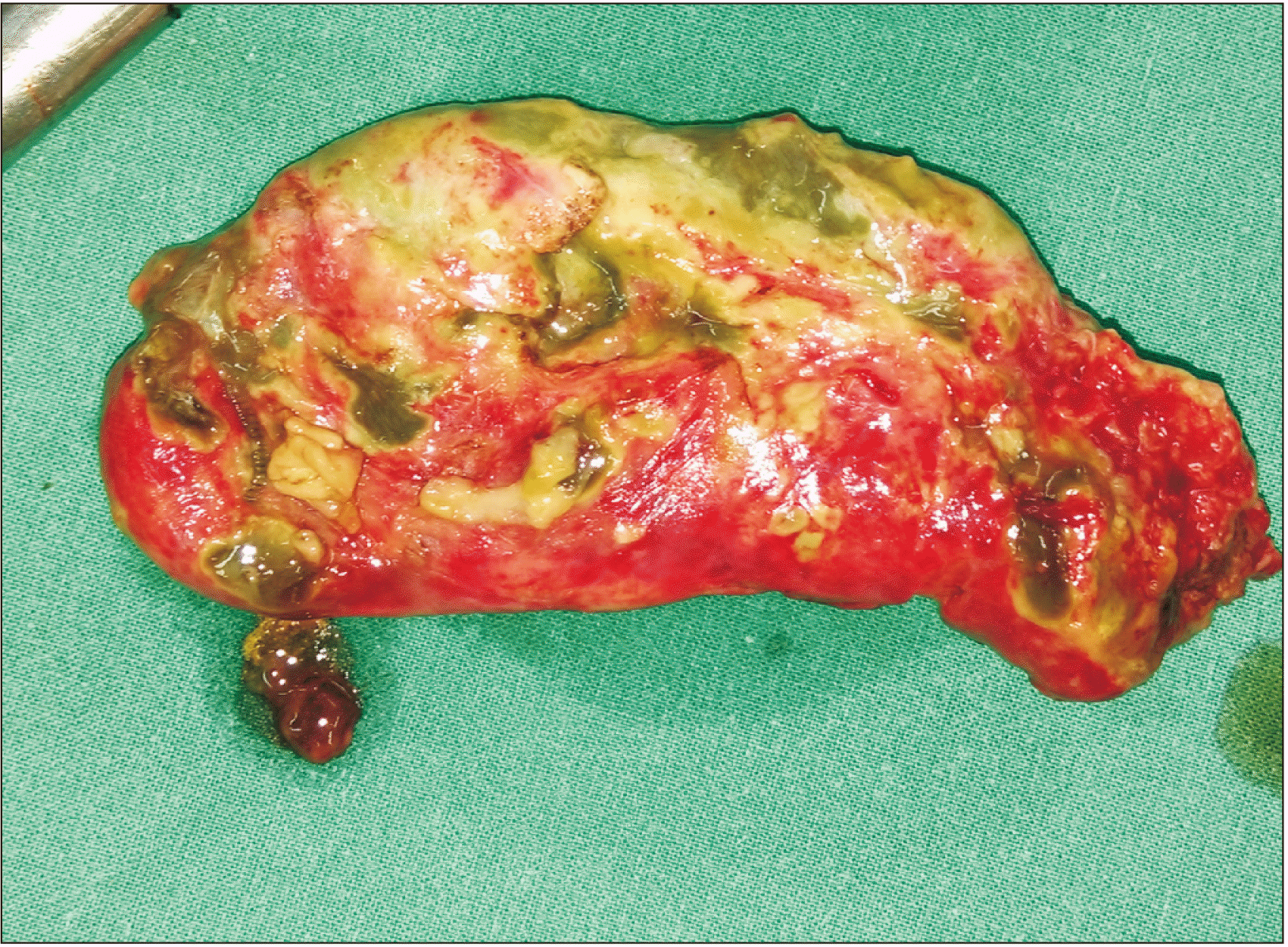
During the duration of this study, there were a total of 623 patients with GB disease. Of them, 116 patients presenting with AC were included in this study (Fig. 1). They were divided into group A (n = 74) and group B (n = 42). The two groups had similar demographic characteristics. Clinical presentation, hemogram, liver function test, imaging studies, and diagnosis of both groups are summarized in Table 1. Diabetes mellitus (n = 34) and hypertension (n = 33) were the commonest comorbidities, followed by hypothyroidism (n = 10), coronary artery disease (n = 7), asthma (n = 3), chronic renal disease (n = 2), and others (rheumatic heart disease, thalassemia major, sickle cell disease, and dilated cardiomyopathy, n = 1 for each). All patients who were considered fit for general anesthesia underwent LC. Seven patients in our series (4 in group A and 3 in group B) had prior infraumbilical abdominal surgery (caesarean section in 5 and appendicectomy in 2 patients). There was no patient in our series who had or required prior cholecystostomy or any biliary drainage. Thirteen patients were treated with ‘reconstituting type’ subtotal cholecystectomy because of difficulty in delineating CVS. We did not have to perform subtotal fenestrating cholecystectomy in any patient. It was converted to open in four (3.4%) patients (two in group A and two in group B). In group A, conversion was because of undissectable Calot’s triangle anatomy due to superimposed acute pancreatitis in one and malignant GB neck mass in another. Conversion in one patient in group B was because of dense pericholecystic adhesions and intrahepatic abscess. Twelve (16.2%) patients in group A had gangrenous GB wall/perforation and 5 (12.0%) patients in group B had similar findings (Fig. 2). Another patient (male) with multiple co-morbidities had intrahepatic perforation of GB, leading to liver abscess (Fig. 3). The liver abscess was drained by ultrasound guided catheter. However, he continued to be in sepsis. Hence, he was operated upon. Laparoscopy in this patient was abandoned and converted to open cholecystectomy due to dense pericholecystic inflammatory adhesions and unclear Calot’s triangle anatomy. This patient died during the postoperative period due to continued sepsis and multiorgan failure. Except for this death (0.9%), there were no major postoperative complications such as CBD injury, bile leak, or bleeding. The mean postoperative hospital stay was two days in group A and three days in group B. Histopathology of all cases showed features suggestive of AC.
There was an initial reluctance amongst surgeons, exemplified by the fact that in UK and USA, only 20% to 30% of surgeons were willing to perform LC for AC [14,15]. However, a paradigm shift has seen. Now more than 90% of surgeons are willing to perform LC for AC [16]. It is now well established that ‘early cholecystectomy’ in these patients is as safe as ‘late cholecystectomy’ with added advantages such as reduced cost, shorter hospital stays, and less loss of working days [3,7,17,18]. Traditionally, GB in the intervening period (i.e., between two to six weeks) is considered ‘untouchable’ and LC (here in called ‘intermediate cholecystectomy’) is reserved only for ‘obstinate’ cases. Although a number of trials are available comparing early cholecystectomy and late cholecystectomy, there is a paucity of literature on the issue of ‘intermediate cholecystectomy.’ A multi-center prospective randomized trial from Germany (ACDC Study, NCT00447304) has addressed the issue of immediate cholecystectomy (i.e., within 24 hours of hospitalization) vis a vis cholecystectomy after at least two days of antibiotic therapy. Surgery was offered to the latter group within 7 to 45 days of hospitalization [5]. That study was similar to ours with respect to the time frame of LC except that we operated on all patients. We subsequently analyzed the data depending on the day of surgery. Our study was a single center study that obviated the ‘operator bias’ inherent to a multi-center trial.
In LC, barring operating surgeon’s experience, major indicators of technical difficulties include operating time, frequency of resorting to alternative techniques such as subtotal cholecystectomy, and rate of conversion from laparoscopy to open cholecystectomy [19]. The median operation time in our study was between one and two hours. There was no significant difference in operating time between the two groups, although we observed a trend toward a longer operating time in group B. This was similar to the experience reported in the ACDC study [5]. The incidence of subtotal cholecystectomy was also similar in both groups.
The overall conversion rate of LC has been reported to be 1%–15% [15-17,20,21]. The overall conversion rate of LC for AC is 6%–34.4% and 45% in a subgroup of patients operated for non-resolution of symptoms or recurrence of the acute attack in the waiting period [3,5,16,17,21-23]. The overall conversion rate in our series was 3.4% without significant difference between groups A and B.
Injury to bile duct is a major concern in LC with an overall incidence of 0.1%–0.6% [16,21]. A similar incidence of 0.07%–0.7% and no difference between early and late interventions have set to rest early apprehensions, making laparoscopy equally safe in AC [3,5,6]. We did not have any bile duct injury in our experience. There was no significant morbidity either. The ACDC study reported a higher incidence of non-biliary complications in the group operated in the second week than that in the group operated within 24 hours of admission [5]. However, most of these complications were pre-existing, attributable to the disease itself rather than a delay in surgery.
The reasons for this low conversion rate and no major complication like bile duct injury could be due to our longstanding tertiary care experience and the operative technique we used for subtotal cholecystectomy for undissectable Calot’s triangle. The latter required not only sound surgical judgment, but also laparoscopic suturing skills to approximate the residual GB flap close to the CD opening.
In our series, one patient in group B died in the postoperative period due to continued sepsis and multiorgan failure. He was subjected to LC after exhausting all conservative treatments including image guided percutaneous catheter drainage. The single death (0.9%) in our series was comparable to the reported mortality incidence of up to 0.8% in various studies [3,5,16,18].
Shortened hospital stay, less hospital cost, and early return to work are proven benefits of early over delayed cholecystectomy for AC [3,18]. The mean postoperative stay in our patients was two days (Table 2). There was no significant difference between the two groups, thereby extending advantages of ‘early cholecystectomy’ to ‘intermediate cholecystectomy’ group as well.
Major limitations of our study were a small case number and the retrospective nature of analysis. However, this can be used as the basis for planning a prospective multicentric study that can authenticate our findings in the future.
In a tertiary care setting with adequate surgical expertise, LC can be safely performed in patients of AC irrespective of the time of presentation. Morbidity, mortality, conversion rates, and postoperative hospital stay of ‘intermediate cholecystectomy’ are similar to ‘early cholecystectomy’ and comparable to those of cholecystectomy performed in elective settings.
REFERENCES
1. The role of laparoscopic cholecystectomy (L.C.). 1993; Guidelines for clinical application. Society of American Gastrointestinal Endoscopic Surgeons (SAGES). Surg Endosc. 7:369–370. DOI: 10.1007/BF00725963. PMID: 8351617.
2. Catena F, Ansaloni L, Bianchi E, Di Saverio S, Coccolini F, Vallicelli C, et al. 2013; The ACTIVE (Acute Cholecystitis Trial Invasive Versus Endoscopic) Study: multicenter randomized, double-blind, controlled trial of laparoscopic versus open surgery for acute cholecystitis. Hepatogastroenterology. 60:1552–1556. PMID: 24634923.
3. Gurusamy KS, Davidson C, Gluud C, Davidson BR. Early versus delayed laparoscopic cholecystectomy for people with acute cholecystitis. Cochrane Database Syst Rev. 2013; (6):CD005440. DOI: 10.1002/14651858.CD005440.pub3. PMID: 23813477.
4. Banz V, Gsponer T, Candinas D, Güller U. 2011; Population-based analysis of 4113 patients with acute cholecystitis: defining the optimal time-point for laparoscopic cholecystectomy. Ann Surg. 254:964–970. DOI: 10.1097/SLA.0b013e318228d31c. PMID: 21817893.
5. Gutt CN, Encke J, Köninger J, Harnoss JC, Weigand K, Kipfmüller K, et al. 2013; Acute cholecystitis: early versus delayed cholecystectomy, a multicenter randomized trial (ACDC study, NCT00447304). Ann Surg. 258:385–393. DOI: 10.1097/SLA.0b013e3182a1599b. PMID: 24022431.
6. Jensen KK, Roth NO, Krarup PM, Bardram L. 2019; Surgical management of acute cholecystitis in a nationwide Danish cohort. Langenbecks Arch Surg. 404:589–597. DOI: 10.1007/s00423-019-01802-0. PMID: 31297607.
7. Okamoto K, Suzuki K, Takada T, Strasberg SM, Asbun HJ, Endo I, et al. Tokyo Guidelines 2018: flowchart for the management of acute cholecystitis. J Hepatobiliary Pancreat Sci. 2018; 25:55–72. Erratum in: J Hepatobiliary Pancreat Sci 2019;26:534. DOI: 10.1002/jhbp.516. PMID: 29045062.
8. Ansaloni L, Pisano M, Coccolini F, Peitzmann AB, Fingerhut A, Catena F, et al. 2016; 2016 WSES guidelines on acute calculous cholecystitis. World J Emerg Surg. 11:25. Erratum in: World J Emerg Surg 2016;11:52. DOI: 10.1186/s13017-016-0088-z. PMID: 27822294. PMCID: PMC5097400.
9. Hirota M, Takada T, Kawarada Y, Nimura Y, Miura F, Hirata K, et al. 2007; Diagnostic criteria and severity assessment of acute cholecystitis: Tokyo Guidelines. J Hepatobiliary Pancreat Surg. 14:78–82. DOI: 10.1007/s00534-006-1159-4. PMID: 17252300. PMCID: PMC2784516.
10. Charlson ME, Pompei P, Ales KL, MacKenzie CR. 1987; A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 40:373–383. DOI: 10.1016/0021-9681(87)90171-8. PMID: 3558716.
11. American Society of Anesthesiologists (ASA). ASA physical status classification system [Internet]. Available from: https://www.asahq.org/resources/clinical-information/asa-physical-status-classification-system. American Society of Anesthesiologists;2014. cited 2023 Jan 1.
12. Strasberg SM, Hertl M, Soper NJ. 1995; An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg. 180:101–125. PMID: 8000648.
13. Strasberg SM, Pucci MJ, Brunt LM, Deziel DJ. 2016; Subtotal cholecystectomy-"fenestrating" vs "reconstituting" subtypes and the prevention of bile duct injury: definition of the optimal procedure in difficult operative conditions. J Am Coll Surg. 222:89–96. DOI: 10.1016/j.jamcollsurg.2015.09.019. PMID: 26521077.
14. Senapati PS, Bhattarcharya D, Harinath G, Ammori BJ. 2003; A survey of the timing and approach to the surgical management of cholelithiasis in patients with acute biliary pancreatitis and acute cholecystitis in the UK. Ann R Coll Surg Engl. 85:306–312. DOI: 10.1308/003588403769162404. PMID: 14594533. PMCID: PMC1964333.
15. Livingston EH, Rege RV. 2004; A nationwide study of conversion from laparoscopic to open cholecystectomy. Am J Surg. 188:205–211. DOI: 10.1016/j.amjsurg.2004.06.013. PMID: 15450821.
16. Navez B, Ungureanu F, Michiels M, Claeys D, Muysoms F, Hubert C, et al. 2012; Surgical management of acute cholecystitis: results of a 2-year prospective multicenter survey in Belgium. Surg Endosc. 26:2436–2445. DOI: 10.1007/s00464-012-2206-7. PMID: 22407152.
17. Gurusamy K, Samraj K, Gluud C, Wilson E, Davidson BR. 2010; Meta-analysis of randomized controlled trials on the safety and effectiveness of early versus delayed laparoscopic cholecystectomy for acute cholecystitis. Br J Surg. 97:141–150. Erratum in: Br J Surg 2010;97:624. DOI: 10.1002/bjs.6870. PMID: 20035546.
18. Hartwig W, Büchler MW. 2014; Acute cholecystitis: early versus delayed surgery. Adv Surg. 48:155–164. DOI: 10.1016/j.yasu.2014.05.008. PMID: 25293613.
19. Nassar AHM, Ng HJ, Wysocki AP, Khan KS, Gil IC. 2021; Achieving the critical view of safety in the difficult laparoscopic cholecystectomy: a prospective study of predictors of failure. Surg Endosc. 35:6039–6047. DOI: 10.1007/s00464-020-08093-3. PMID: 33067645. PMCID: PMC8523408.
20. Sutcliffe RP, Hollyman M, Hodson J, Bonney G, Vohra RS, Griffiths EA. 2016; Preoperative risk factors for conversion from laparoscopic to open cholecystectomy: a validated risk score derived from a prospective U.K. database of 8820 patients. HPB (Oxford). 18:922–928. DOI: 10.1016/j.hpb.2016.07.015. PMID: 27591176. PMCID: PMC5094477.
21. Hu ASY, Menon R, Gunnarsson R, de Costa A. 2017; Risk factors for conversion of laparoscopic cholecystectomy to open surgery - a systematic literature review of 30 studies. Am J Surg. 214:920–930. DOI: 10.1016/j.amjsurg.2017.07.029. PMID: 28739121.
22. Overby DW, Apelgren KN, Richardson W, Fanelli R. 2010; SAGES guidelines for the clinical application of laparoscopic biliary tract surgery. Surg Endosc. 24:2368–2386. DOI: 10.1007/s00464-010-1268-7. PMID: 20706739.
23. Tan JK, Goh JC, Lim JW, Shridhar IG, Madhavan K, Kow AW. 2017; Same admission laparoscopic cholecystectomy for acute cholecystitis: is the "golden 72 hours" rule still relevant? HPB (Oxford). 19:47–51. DOI: 10.1016/j.hpb.2016.10.006. PMID: 27825751.
Fig. 3
(A, B) Contrast enhanced computed tomography scan showing necrotic gall bladder (GB) wall. (C, D) Intrahepatic abscess caused by GB perforation.
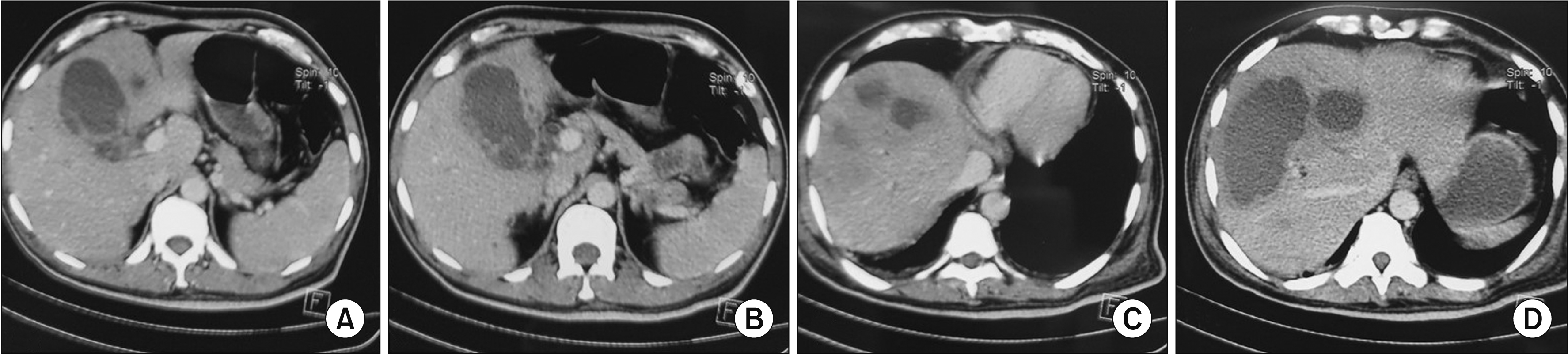
Table 1
Clinical presentation, lab reports and final diagnosis
| Group A (n = 74) | Group B (n = 42) | p-value | |
|---|---|---|---|
| Demography | |||
| Male | 35 (47.3) | 18 (42.9) | |
| Female | 39 (52.7) | 24 (57.1) | |
| Mean age (yr) | 47.3 (22–68) | 48.5 (15–80) | |
| Clinical presentation | |||
| Pain | 74 (100) | 42 (100) | NA |
| Fever | 24 (32.4) | 8 (19.0) | 0.121a) |
| Jaundice | 14 (18.9) | 6 (14.2) | 0.525a) |
| Murphy’s sign (+) | 57 (77.0) | 27 (64.2) | 0.140a) |
| Previous attack of cholecystitis | 9 (12.5) | 8 (19.0) | 0.314a) |
| Hemogram | |||
| Leukocytosis (normal 4,000–11,000/µL) | 52 (70.3) | 26 (61.9) | 0.027* |
| Liver function test | |||
| Serum bilirubin > 2 mg/dL | 14 (18.9) | 6 (14.3) | 0.525 a) |
| Raised serum transaminase (normal 15–37 U/L) | 15 (20.3) | 7 (16.7) | 0.634 a) |
| Raised serum transpeptidase (normal 30–65 U/L) | 15 (20.3) | 7 (16.7) | 0.634 a) |
| Raised serum alkaline phosphatase (normal 50–136 U/L) | 27 (36.4) | 7 (16.7) | 0.024* |
| Raised serum gama glutamyl transpeptidase (normal 5–55 U/L) | 25 (33.7) | 8 (19.0) | 0.091a) |
| Final diagnosis | |||
| Empyema GB | 12 (16.2) | 6 (14.2) | 0.783a) |
| GB gangrene/perforation | 12 (16.2) | 5 (12.0) | 0.528a) |
| Acalculous cholecystitis | 1 (1.3) | 0 (0) | NA |
| Acute cholecystitis with acute pancreatitis | 1 (1.3) | 0 (0) | |
| Acute cholecystitis with GB malignancy | 1 (1.3) | 0 (0) | < 0.001* |
| Acute cholecystitis with choledocholithiasis | 1 (1.3) | 2 (4.7) | 0.297a) |
Table 2
Operative details
| Group A (n = 74) | Group B (n = 42) | p-value | |
|---|---|---|---|
| Thick walled, edematous, inflamed GB wall | 74 (100) | 42 (100) | NA |
| Gangrene/perforation of GB | 12 (16.2) | 5 (11.9) | 0.528a) |
| Impacted stone in Hartman’s pouch | 27 (36.4) | 13 (31.0) | 0.547a) |
| Both cystic duct & artery identifiable | 64 (86.4) | 35 (83.3) | 0.644a) |
| Subtotal cholecystectomy | 8 (10.8) | 5 (11.9) | > 0.999a) |
| Duration of surgery (min) | 0.945a) | ||
| < 60 | 31 (41.9) | 19 (45.2) | |
| 60–90 | 32 (43.2) | 17 (40.5) | |
| 90–120 | 8 (10.8) | 5 (11.9) | |
| > 120 | 3 (4.1) | 1 (2.4) | |
| Conversion to open | 2 (2.7) | 2 (4.7) | 0.625a) |
| Mean postoperative stay (day) | 2 (1–6) | 3 (1–8) |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download