1. Winawer SJ, Zauber AG, O’Brien MJ, et al. Randomized comparison of surveillance intervals after colonoscopic removal of newly diagnosed adenomatous polyps. The National Polyp Study Workgroup. N Engl J Med. 1993; 328:901–906.
2. Zauber AG, Winawer SJ, O’Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012; 366:687–696.
3. Ponugoti PL, Cummings OW, Rex DK. Risk of cancer in small and diminutive colorectal polyps. Dig Liver Dis. 2017; 49:34–37.
4. Iwatate M, Ikumoto T, Hattori S, et al. NBI and NBI combined with magnifying colonoscopy. Diagn Ther Endosc. 2012; 2012:173269.
5. Hewett DG, Kaltenbach T, Sano Y, et al. Validation of a simple classification system for endoscopic diagnosis of small colorectal polyps using narrow-band imaging. Gastroenterology. 2012; 143:599–607.
6. Ciocâlteu AM, CârŢână ET, Florescu DN, et al. Narrow band imaging with near-focus mode for colorectal polyps’ characterization. Rom J Morphol Embryol. 2016; 57(2 Suppl):619–626.
7. Iwatate M, Sano Y, Hattori S, et al. The addition of high magnifying endoscopy improves rates of high confidence optical diagnosis of colorectal polyps. Endosc Int Open. 2015; 3:E140–E145.
8. Ikematsu H, Matsuda T, Osera S, et al. Usefulness of narrow-band imaging with dual-focus magnification for differential diagnosis of small colorectal polyps. Surg Endosc. 2015; 29:844–850.
9. Rex DK. Narrow-band imaging without optical magnification for histologic analysis of colorectal polyps. Gastroenterology. 2009; 136:1174–1181.
10. Rastogi A, Keighley J, Singh V, et al. High accuracy of narrow band imaging without magnification for the real-time characterization of polyp histology and its comparison with high-definition white light colonoscopy: a prospective study. Am J Gastroenterol. 2009; 104:2422–2430.
11. Chiu HM, Chang CY, Chen CC, et al. A prospective comparative study of narrow-band imaging, chromoendoscopy, and conventional colonoscopy in the diagnosis of colorectal neoplasia. Gut. 2007; 56:373–379.
12. Kuruvilla N, Paramsothy R, Gill R, et al. A prospective dual-center proof-of-principle study evaluating the incremental benefit of narrow-band imaging with a fixed zoom function in real-time prediction of polyp histology: can we resect and discard? Gastrointest Endosc. 2015; 82:362–369.
13. The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003; 58(6 Suppl):S3–43.
14. Singh R, Bhat YM, Thurairajah PH, et al. Is narrow band imaging superior to high-definition white light endoscopy in the assessment of diminutive colorectal polyps? J Gastroenterol Hepatol. 2013; 28:472–478.
15. Nagtegaal ID, Odze RD, Klimstra D, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020; 76:182–188.
16. McGill SK, Evangelou E, Ioannidis JP, et al. Narrow band imaging to differentiate neoplastic and non-neoplastic colorectal polyps in real time: a meta-analysis of diagnostic operating characteristics. Gut. 2013; 62:1704–1713.
17. Kaltenbach T, Rastogi A, Rouse RV, et al. Real-time optical diagnosis for diminutive colorectal polyps using narrow-band imaging: the VALID randomised clinical trial. Gut. 2015; 64:1569–1577.
18. Hattori S, Iwatate M, Sano W, et al. Narrow-band imaging observation of colorectal lesions using NICE classification to avoid discarding significant lesions. World J Gastrointest Endosc. 2014; 6:600–605.
19. Szura M, Pasternak A, Bucki K, et al. Two-stage optical system for colorectal polyp assessments. Surg Endosc. 2016; 30:204–214.
20. Wallace MB, Crook JE, Coe S, et al. Accuracy of in vivo colorectal polyp discrimination by using dual-focus high-definition narrow-band imaging colonoscopy. Gastrointest Endosc. 2014; 80:1072–1087.
21. Singh R, Jayanna M, Navadgi S, et al. Narrow-band imaging with dual focus magnification in differentiating colorectal neoplasia. Dig Endosc. 2013; 25 Suppl 2:16–20.
22. Higashi R, Uraoka T, Kato J, et al. Diagnostic accuracy of narrow-band imaging and pit pattern analysis significantly improved for less-experienced endoscopists after an expanded training program. Gastrointest Endosc. 2010; 72:127–135.
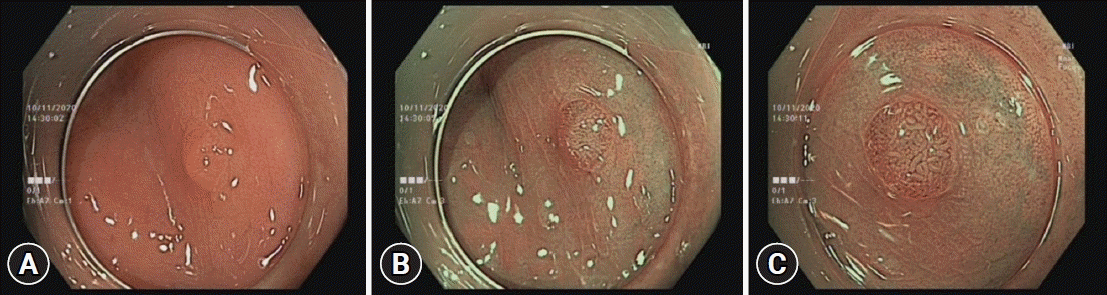




 PDF
PDF Citation
Citation Print
Print



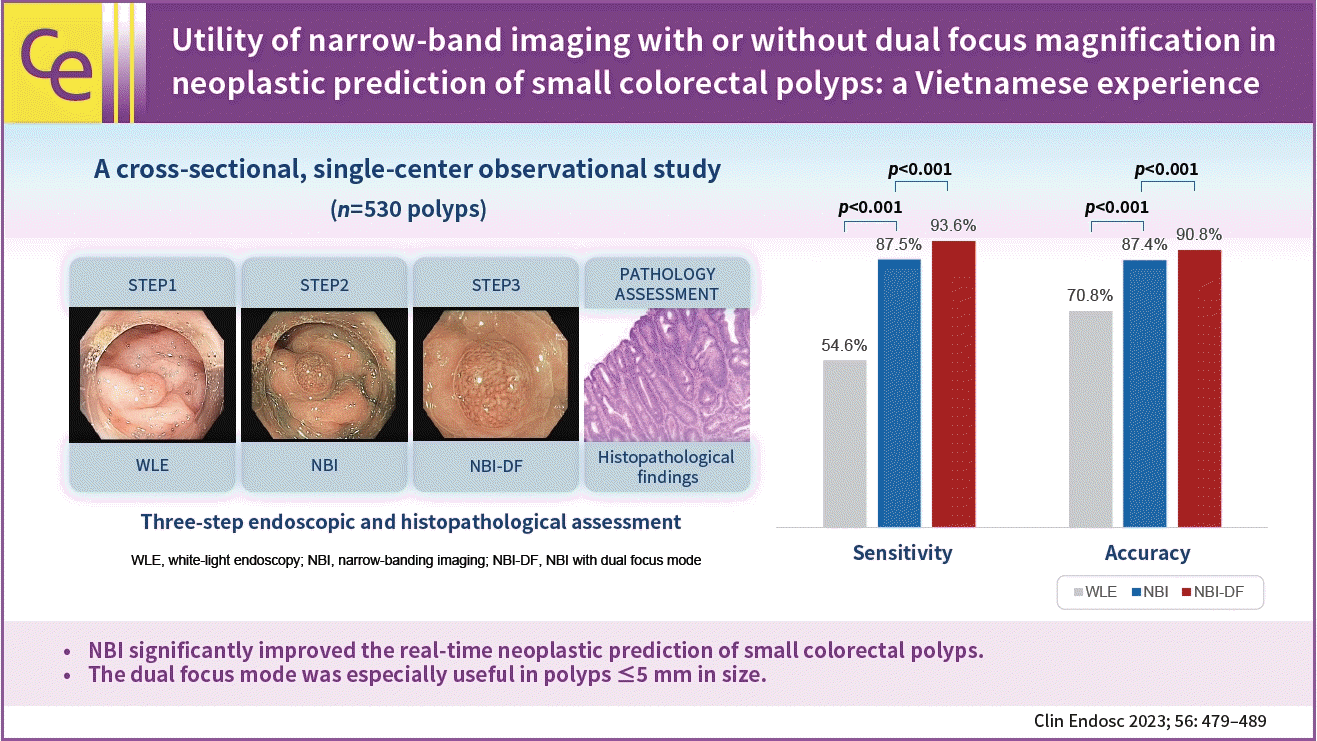
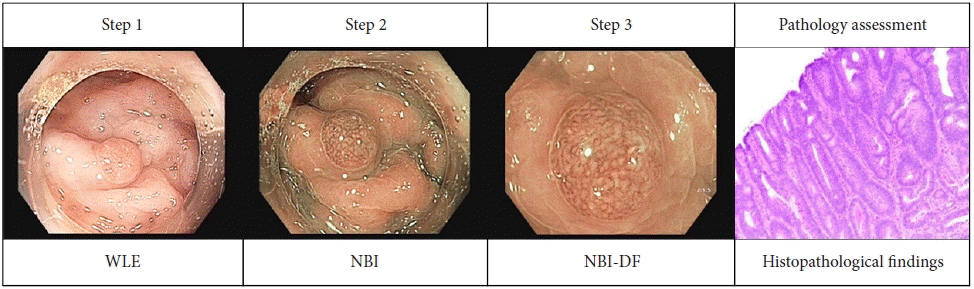

 XML Download
XML Download