Abstract
Background
Increasingly many patients have been diagnosed with stage I adenocarcinoma due to the use of low-dose chest computed tomography for lung cancer screening. Therefore, this study aimed to analyze tumor recurrence based on the predominant subtype in patients with stage I lung adenocarcinoma who underwent lobectomy.
Methods
We retrospectively analyzed 114 patients who underwent lobectomy for pathologic stage I lung adenocarcinoma from June 2001 to July 2019.
Results
In univariate analyses, significant factors were current smoking at the time of surgery (p=0.029), pathologic tumor size (p=0.006), central tumor location (p=0.003), maximum standardized uptake value on positron emission tomography-computed tomography (p=0.001), and the solid predominant subtype (p=0.012). In the multivariate analysis, only the solid predominant subtype (hazard ratio, 9.702; 95% confidence interval, 1.179–79.874; p=0.035) was an independent risk factor.
The use of low-dose chest computed tomography (CT) for lung cancer screening has led to an increased number of patients diagnosed with stage I lung adenocarcinoma. Lobectomy and mediastinal lymph node dissection are standard treatments for stage I adenocarcinoma and do not consider adjuvant therapy. The 5-year overall survival (OS) rate of early-stage non-small cell lung cancer (NSCLC; stage I) was as high as up to 80.4% [1]. However, in early-stage lung adenocarcinoma, some patients still suffer from recurrence; these patients are followed up without adjuvant therapy based on the treatment principle, the recurrence is judged until the tumor grows to some extent, and then, treatment is started. It is a dismal reality that the patient's time is consumed, and the chance of remission is reduced as the treatment time is delayed. Therefore, we thought that selective adjuvant therapy is required for patients with stage I lung adenocarcinoma who underwent lobectomy to investigate factors that increase the risk of recurrence.
Ethical statements: The Institutional Review Board of Daegu Catholic University Medical Center approved this study (IRB No: CR-22-181). The need for informed consent was waived due to the study's retrospective nature and patient data anonymization.
We retrospectively analyzed 114 patients who underwent lobectomy with systemic lymph node dissection or sampling for pathologic stage I lung adenocarcinoma from June 2001 to July 2019. All of them underwent contrast-enhanced chest CT, positron emission tomography-CT (PET-CT), bone scan, and brain magnetic resonance imaging at preoperative work-up to confirm the absence of metastases. The exclusion criteria of the patient group are as follows: induction therapy (preoperative chemotherapy and/or preoperative radiotherapy), history of lung cancer, death within 30 days postoperatively, and loss of postoperative follow-up.
The resected pulmonary and lymphatic tissues were stained with formalin in the Pathology Department, stained with hematoxylin and eosin, and read by a pathologist. All adenocarcinomas were divided into five subtypes: lepidic, acinar, papillary, solid, and micropapillary. The subtypes occupying the largest proportion were determined as predominant subtypes. There were five subtypes classified based on the International Association for the Study of Lung Cancer, American Thoracic Society, and European Respiratory Society classification system. Visceral pleural and lymphovascular invasion were noted. Pathological staging was based on the TNM classification (8th edition) of the American Joint Committee on Cancer. The pathological stage by previous TNM classification was revised according to the 8th edition by a thoracic surgeon based on the pathology report. Tumor location is divided into central and peripheral regions based on CT findings, centrally located tumor was defined as a case located in the inner one-third of the hemithorax adopted by drawing concentric lines arising from the midline [2].
The postoperative follow-up schedule is as follows: chest CT every 3 or 4 months in the first 2 years, every 6 months, and then after 3 years. Bone scan was performed once a year, brain magnetic resonance imaging was performed when neurological symptoms occurred, and PET-CT was performed when recurrence was suspected. Recurrence diagnosis was determined based on follow-up radiologic findings supplemented with pathologic examination and defined as including both loco-regional recurrence and distant recurrence. The definition of local recurrence was any new lesion adjacent to the bronchial stump, to the staple line. Regional recurrence was defined as any new lesion in the ipsilateral lung or in the lymph node stations of the ipsilateral thorax. Distant metastasis included pleural metastasis, malignant pleural effusion, malignant pericardial effusion, metastasis to the contralateral lung, or extrathoracic metastasis [3]. Exclusion criteria of recurrence included second primary lung cancer and lung metastasis from primary cancers other than the lungs. The definition of second primary lung cancer is as follows: the tumor has a different tumor type; tumors with the same tumor type have different major histologic patterns and tumors with the same major histologic pattern have different other histologic patterns and cytologic features. OS was defined as the duration from surgery to death of any cause. Freedom from recurrence (FFR) was defined as the duration from the date of surgery to the date of the first diagnosis of recurrence or the recent follow-up date without recurrence. Disease-specific survival (DSS) was defined as the duration from the date of surgery to the date of tumor-related death.
Univariate and multivariate analyses were performed using Cox proportional hazard model in the SPSS statistics (version 18, IBM Corp.). Variables with p-values of <0.05 in univariate analysis were entered into the multivariate analysis. A p-value of <0.05 was considered statistically significant. The OS and FFR rates were calculated using the Kaplan-Meier method.
The mean age of patients was 63.8±9.6 years and median follow-up time was 59.5 months (range, 7.0–206.3 months). The patient characteristics are summarized in Table 1. Smoking status at the time of surgery was 79 (69.3%) for never-smokers, 10 (8.8%) for ex-smokers, and 25 (21.9%) for current smokers. The median tumor size was 2.3 cm (range, 0.8–4.0 cm). The median maximum standardized uptake value (SUVmax) of PET-CT was 2.7 (range, 0.1–11.4). Tumor location was central and peripheral in found in nine (7.9%) and 105 (92.1%) patients. Pathologic predominant subtypes were 16 lepidic (14.0%), 55 acinar (48.2%), 27 papillary (23.7%), and 16 solid (14.0%); however, the micropapillary subtype was excluded because there was only one case.
Of the 114 patients, 29 (25.4%) recurrence occurred and 85 (74.6%) had no recurrence. The median time from surgery to recurrence was 24.2 months (range, 5.8–90.2 months) (Table 1). Recurrence rates according to predominant subtype and stage are summarized in Table 2. Compared with other subtypes, the recurrence rate of the solid predominant pattern is very high at 56.3% (Table 2). Loco-regional and distant recurrence were 16 (14.0%) and 13 (11.4%), respectively (Table 3). The loco-reginal and distant recurrence according to the predominant subtype is shown in Table 3.
A total of 114 patients pathologically diagnosed with stage I adenocarcinoma were analyzed for risk factors associated with recurrence. In univariate analyses, significant factors were current smokers at the surgery time (p=0.029), pathologic tumor size (p=0.006), central tumor location (p=0.003), SUVmax of PET-CT (p=0.001), and solid predominant subtype (p=0.012). Visceral pleural invasion (p=0.055) showed a trend toward recurrence. Lymphovascular invasion (p=0.194) and papillary predominant subtype (p=0.086) were not significant factors (Table 4). In the multivariate analysis, only the solid predominant subtype (hazard ratio [HR], 9.702; 95% confidence interval [CI], 1.179–79.874; p=0.035) was an independent risk factor (Table 4). In Kaplan-Meier analyses, the 5-year OS, DSS, and FFR rates were 76.0%, 81.3%, and 73.8%, respectively (Fig. 1). The rate of 5-year FFR was 100%, 77.2%, 64.2%, and 47.1% for the lepidic, acinar, papillary, and solid groups, respectively (Fig. 2A). When divided into non-solid and solid groups, the 5-year FFR was 78.1% and 47.1%, respectively (Fig. 2B). The 5-year DSS rates were 100%, 82.9%, 81.6%, and 53.8% for lepidic, acinar, papillary, and solid, respectively (p=0.007) (Fig. 3A), and when classified as non-solid and solid groups, the 5-year DSS rates were 85.3%, and 53.8%, respectively (p=0.001) (Fig. 3B). The 5-year OS rates were 100%, 73.3%, 78.1%, and 57.8% for lepidic, acinar, papillary, and solid groups, respectively (p=0.038) (Fig. 4A), and when divided into non-solid and solid groups, the 5-year OS rates were 79.0%, and 57.8%, respectively (p=0.010) (Fig. 4B).
Surgical resection is the standard treatment for stage I lung adenocarcinoma. However, we observed unfortunate recurrences even in patients who underwent lobectomy. Therefore, this study sought to find risk factors related to recurrence in 114 patients who underwent lobectomy in stage I adenocarcinoma.
In our study, current smoker at the surgery time, pathologic tumor size, central tumor location, SUVmax of PET-CT, and solid predominant subtype were statistically significant risk factors in univariate analyses. These results have also been shown in other studies. Wu et al. [4] reported that current smokers at surgery (HR=1.63, p=0.037) and larger tumor size (≤2 cm reference; 2–3 cm HR=1.55, 3–5 cm HR=2.03, p=0.031) were significant risk factors for recurrence in 356 patients with stage I NSCLC who underwent pulmonary resection. Shiono et al. [5] reported that SUV index is a significant predictor for recurrence (HR=1.26, p<0.01) in 183 patients with stage IA lung cancer who underwent surgical resection and SUV index of <1.0 suggests limited resection because recurrence is less likely. Several studies reported that the central tumor location was a worse independent predictor of recurrence-free survival in patients with stage I NSCLC who underwent complete resection [6,7]. In other studies, visceral pleural invasion and lymphovascular invasion were also significantly associated with recurrence [6,8]; however, only visceral pleural invasion had a trend in our study (p=0.055).
In our study, the solid predominant subtype was the only independent risk factor in multivariate analysis. The recurrence rate was highest at 56.3% in patients with stage I adenocarcinoma with the solid predominant subtype, and the HR for recurrence was 9.7 (p=0.035). In some studies, micropapillary and solid predominant components were significantly associated with poor disease-free survival and OS and recurrence. Tsao et al. [9] evaluated 575 patients who underwent surgical resection with adenocarcinoma and reported poorer DSS and a significant benefit from adjuvant chemotherapy in the solid predominant pattern. Yoshizawa et al. [10] analyzed 514 patients with pathologic stage I lung adenocarcinoma who underwent lobectomy. In the multivariate analysis, solid, micropapillary, invasive mucinous adenocarcinoma and colloid predominant subtypes were poor prognostic factors, which all noted that adjuvant therapy may be needed. Motono et al. [11] reported the lepidic/acinar/papillary group has a significantly better prognosis for recurrence-free survival than the micropapillary/solid/invasive mucinous adenocarcinoma group in a study of 197 completely resected pathologic stage I invasive lung adenocarcinoma.
We believe that the high recurrence rate in the solid predominant subtype is caused by a more aggressive behavior, resulting in a higher tendency for early distant metastasis. In some studies, the association between lymph node metastasis or micro-metastasis and micropapillary or solid subtype component was investigated. Hung [12] reported that the solid or micropapillary subtype was a significant predictor of occult lymph node metastasis or micro-metastasis in stage I lung adenocarcinoma and was associated with occult N2 lymph node metastasis [13], which has a more aggressive behavior, indicating a higher probability of initial extrathoracic only recurrence and early distant metastasis in lung adenocarcinoma [14]. These results suggest that the solid predominant pattern has a more aggressive course, and therefore, adjuvant therapy should be considered even in early-stage lung adenocarcinoma. Several studies evaluated the effects of adjuvant chemotherapy on stage I NSCLC. Tsutani et al. [15] reported that adjuvant chemotherapy provides significantly better recurrence-free survival and OS in patients with stage I NSCLC with 2 cm or more invasive component and lymphatic invasion than observation-alone patients. Park et al. [16] reported that platinum-based adjuvant chemotherapy provided better OS (HR=0.428, p=0.049) and disease-free survival (HR=0.57, p=0.043) in 119 patients with stage IB NSCLC who underwent lobectomy and mediastinal lymph node dissection, respectively.
We analyzed Kaplan-Meier curves for FFR, DSS, and OS. These results indicate that the solid predominant subtype has a significant bad effect on patient’s OS, DSS, and FFR (Fig. 1). Therefore, we think it is reasonable to consider adjuvant therapy in patients who underwent surgical resection with a solid predominant subtype even in stage I adenocarcinoma. Currently, the efficacy of adjuvant chemotherapy in early-stage NSCLC has been assessed as having modest benefit; however, no specific criteria have been established for patients who are expected to benefit from adjuvant chemotherapy and who are likely to be disadvantageous due to its toxicity [17].
The limitation of our study is a retrospective design. The number of patients included in the study was relatively small. The micropapillary predominant subtype was excluded from the study because there was only one case.
In conclusion, the solid predominant subtype is an independent risk factor associated with recurrence after lobectomy in patients with stage I adenocarcinoma. Therefore, if the solid subtype is predominant in pathologic findings, consideration of adjuvant therapy after standard surgical therapy may help reduce the risk of tumor recurrence and increase survival.
Notes
Author contributions
Conceptualization: YHJ. Data curation: CHL, YHJ. Formal analysis: CHL. Investigation: CHL. Methodology: CHL. Project administration: YHJ. Resources: YHJ. Software: CHL. Supervision: YHJ. Validation: YHJ. Visualization: CHL, YHJ. Writing - original draft: CHL. Writing - review and editing: YHJ.
References
1. Albano D, Bilfinger T, Nemesure B. 1-, 3-, and 5-year survival among early-stage lung cancer patients treated with lobectomy vs SBRT. Lung Cancer (Auckl). 2018; 9:65–71.
2. Shin SH, Jeong DY, Lee KS, Cho JH, Choi YS, Lee K, et al. Which definition of a central tumour is more predictive of occult mediastinal metastasis in nonsmall cell lung cancer patients with radiological N0 disease? Eur Respir J. 2019; 53:1801508.
3. Lou F, Huang J, Sima CS, Dycoco J, Rusch V, Bach PB. Patterns of recurrence and second primary lung cancer in early-stage lung cancer survivors followed with routine computed tomography surveillance. J Thorac Cardiovasc Surg. 2013; 145:75–82.
4. Wu CF, Fu JY, Yeh CJ, Liu YH, Hsieh MJ, Wu YC, et al. Recurrence risk factors analysis for stage I non-small cell lung cancer. Medicine (Baltimore). 2015; 94:e1337.
5. Shiono S, Abiko M, Sato T. Limited resection for clinical Stage IA non-small-cell lung cancers based on a standardized-uptake value index. Eur J Cardiothorac Surg. 2013; 43:e7–12.
6. Zhang Y, Sun Y, Xiang J, Zhang Y, Hu H, Chen H. A clinicopathologic prediction model for postoperative recurrence in stage Ia non-small cell lung cancer. J Thorac Cardiovasc Surg. 2014; 148:1193–9.
7. Kang SW, Jeong WG, Lee JE, Oh IJ, Song SY, Lee BC, et al. Prognostic significance of location index in resected T1-sized early-stage non-small cell lung cancer. Acta Radiol. 2023; 64:1028–37.
8. Huang H, Wang T, Hu B, Pan C. Visceral pleural invasion remains a size-independent prognostic factor in stage I non-small cell lung cancer. Ann Thorac Surg. 2015; 99:1130–9.
9. Tsao MS, Marguet S, Le Teuff G, Lantuejoul S, Shepherd FA, Seymour L, et al. Subtype classification of lung adenocarcinoma predicts benefit from adjuvant chemotherapy in patients undergoing complete resection. J Clin Oncol. 2015; 33:3439–46.
10. Yoshizawa A, Motoi N, Riely GJ, Sima CS, Gerald WL, Kris MG, et al. Impact of proposed IASLC/ATS/ERS classification of lung adenocarcinoma: prognostic subgroups and implications for further revision of staging based on analysis of 514 stage I cases. Mod Pathol. 2011; 24:653–64.
11. Motono N, Matsui T, Machida Y, Usuda K, Uramoto H. Prognostic significance of histologic subtype in pStage I lung adenocarcinoma. Med Oncol. 2017; 34:100.
12. Hung JJ. Histologic subtype component predicts lymph node micrometastasis and prognosis in patients with stage I lung adenocarcinoma. J Thorac Dis. 2017; 9:3623–5.
13. Hung JJ, Yeh YC, Jeng WJ, Wu YC, Chou TY, Hsu WH. Factors predicting occult lymph node metastasis in completely resected lung adenocarcinoma of 3 cm or smaller. Eur J Cardiothorac Surg. 2016; 50:329–36.
14. Hung JJ, Yeh YC, Jeng WJ, Wu KJ, Huang BS, Wu YC, et al. Predictive value of the international association for the study of lung cancer/American Thoracic Society/European Respiratory Society classification of lung adenocarcinoma in tumor recurrence and patient survival. J Clin Oncol. 2014; 32:2357–64.
15. Tsutani Y, Miyata Y, Kushitani K, Takeshima Y, Yoshimura M, Okada M. Propensity score-matched analysis of adjuvant chemotherapy for stage I non-small cell lung cancer. J Thorac Cardiovasc Surg. 2014; 148:1179–85.
16. Park SY, Lee JG, Kim J, Byun GE, Bae MK, Lee CY, et al. Efficacy of platinum-based adjuvant chemotherapy in T2aN0 stage IB non-small cell lung cancer. J Cardiothorac Surg. 2013; 8:151.
17. Nagasaka M, Gadgeel SM. Role of chemotherapy and targeted therapy in early-stage non-small cell lung cancer. Expert Rev Anticancer Ther. 2018; 18:63–70.
Fig. 1.
Kaplan-Meier analysis for overall survival (5-year overall survival rate; 76.0%) (A), disease-specific survival (5-year disease-specific survival rate; 81.3%) (B), and freedom from recurrence (5-year freedom from recurrence rate; 73.8%) (C) in 114 patients with pathologic stage I adenocarcinoma.
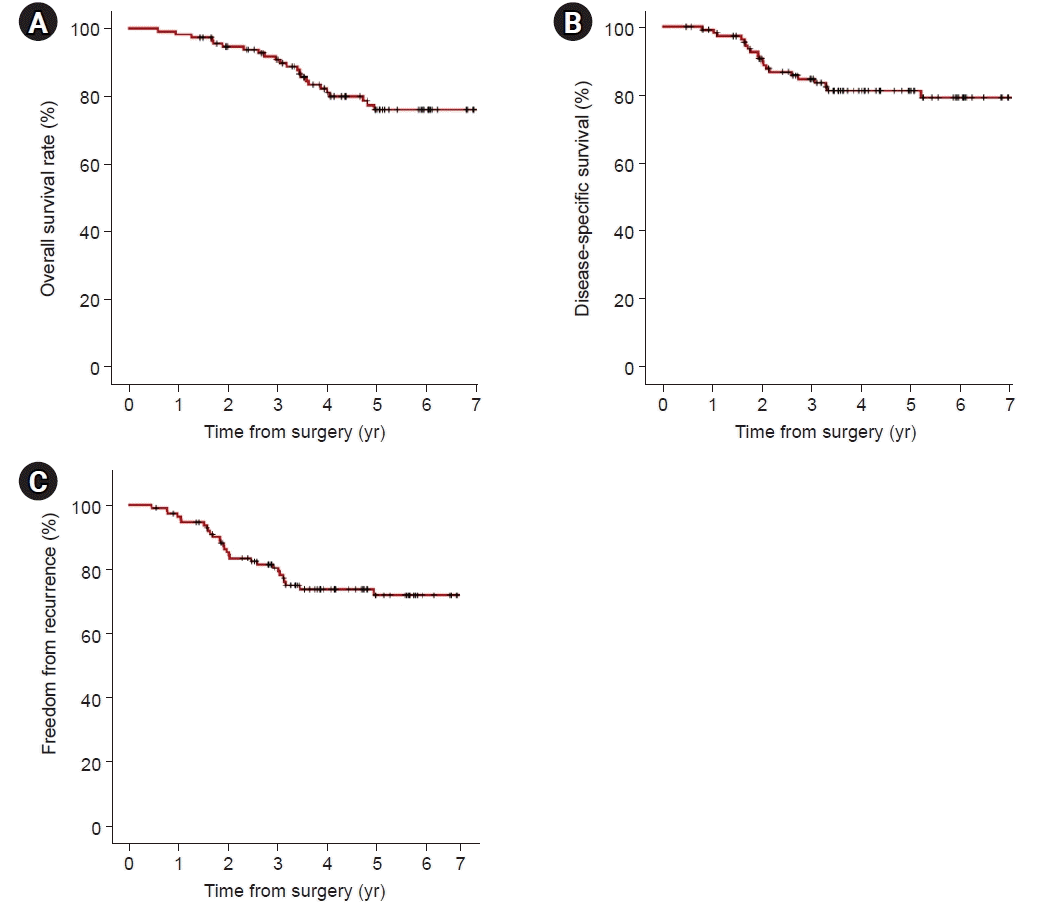
Fig. 2.
Kaplan-Meier analysis (log-rank test) for the freedom from recurrence rate according to all pathologic predominant subtypes (5-year freedom from recurrence rate; lepidic 100%, acinar 77.2%, papillary 64.2%, and solid 47.1%) (A) and freedom from recurrence rate for non-solid and solid tumors (5-year freedom from recurrence rate; non-solid 78.1% and solid 47.1%) (B).
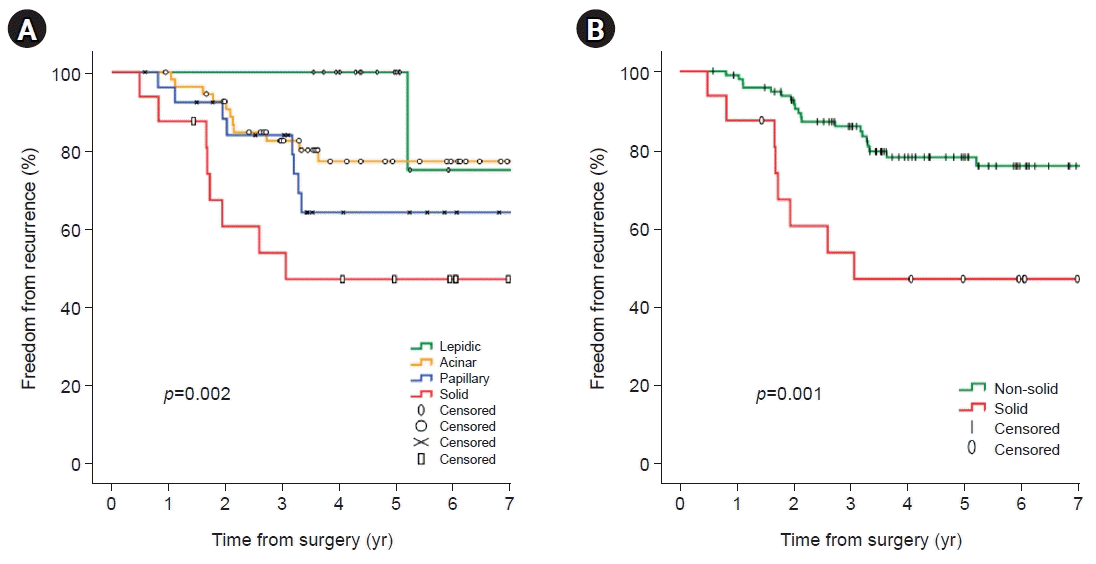
Fig. 3.
Kaplan-Meier analysis (log-rank test) for disease-specific survival according to all pathologic predominant subtypes (5-year disease-specific survival rate; lepidic 100%, acinar 82.9%, papillary 81.6%, and solid 53.8%) (A) and disease-specific survival for non-solid and solid tumors (5-year disease-specific survival rate; non-solid 85.3% and solid 53.8%) (B).
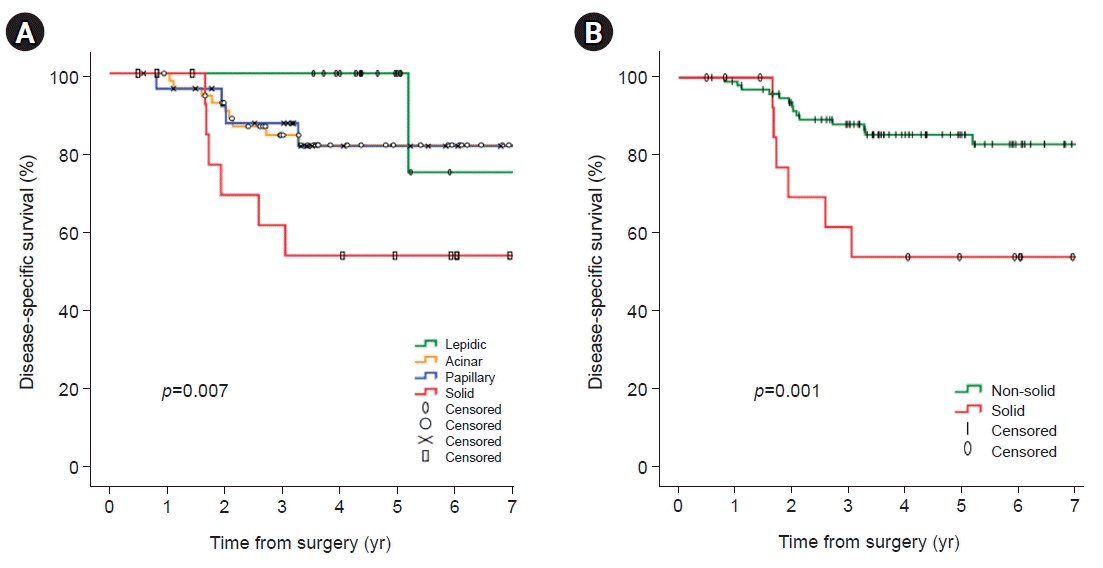
Fig. 4.
Kaplan-Meier analysis (log-rank test) for overall survival according to all pathologic predominant subtypes (5-year overall survival rate; lepidic 100%, acinar 73.3%, papillary 78.1%, and solid 57.8%) (A) and overall survival based for non-solid and solid tumors (5-year overall survival rate; non-solid 79.0% and solid 57.8%) (B).
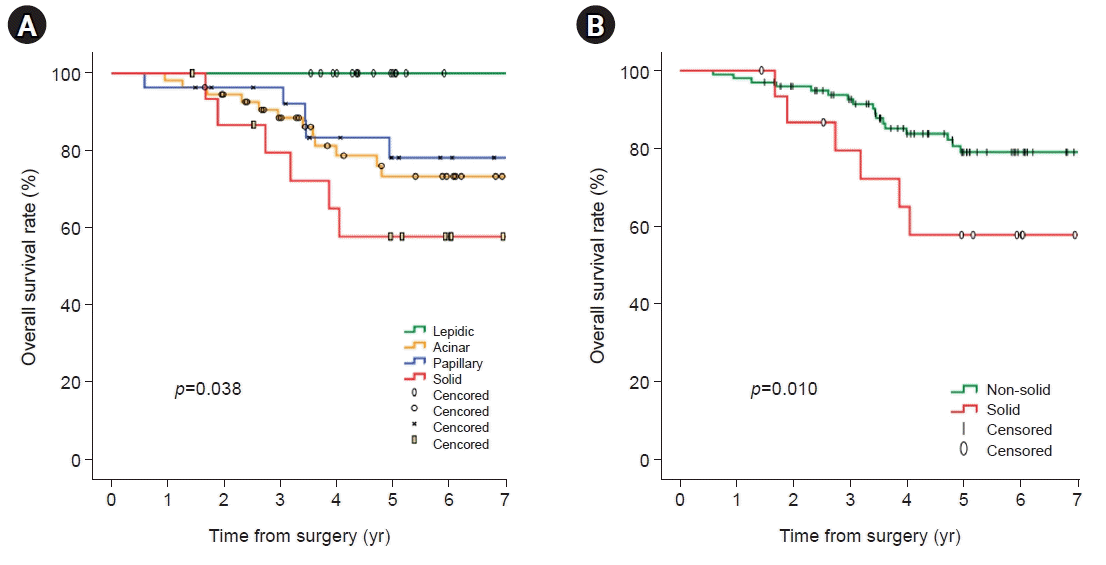
Table 1.
Clinicopathological variables in 114 patients with pathologic stage I adenocarcinoma
Table 2.
Recurrence rate according to the predominant subtype and stage
Table 3.
Loco-regional and distant recurrence according to the predominant type
| Loco-regional recurrence | Distant recurrence | |
|---|---|---|
| Lepidic (n=16) | 1 (6.3) | 0 |
| Acinar (n=55) | 6 (10.9) | 5 (9.1) |
| Papillary (n=27) | 5 (18.5) | 3 (11.1) |
| Solid (n=16) | 4 (25.0) | 5 (31.3) |
| Total | 16 (14.0) | 13 (11.4) |
Table 4.
Univariate and multivariate analyses of risk factors for recurrence in 114 patients with pathologic stage I adenocarcinoma




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download