Abstract
Objectives.
We aimed to develop a new calculation model for calcium requirements in dialysis patients following parathyroidectomy.
Methods.
A total of 98 patients with secondary hyperparathyroidism receiving parathyroidectomy from January 2014 to January 2022 were enrolled in this study. Among these patients, 78 were randomly selected for construction of the calcium requirement calculation model, and the remaining 20 patients were selected for model validation. The calcium requirement model estimated the total calcium supplementation for 1 week after surgery using variables with significant relationships in the derivation group by stepwise multiple linear regression analysis. Bias, precision, and accuracy were measured in the validation group to determine the performance of the model.
Results.
The model was as follows: calcium requirement for 1 week after surgery=33.798–8.929×immediate postoperative calcium+0.190×C-reactive protein–0.125×age+0.002×preoperative intact parathyroid hormone+0.003×preoperative alkaline phosphatase (R2=0.8). The model was successfully validated.
Conclusion.
We generated a novel model to guide calcium supplementation. This model can assist in stabilizing the serum calcium levels of patients during the early postoperative period. Furthermore, it contributes to the individualized and precise treatment of hypocalcemia in patients following parathyroidectomy.
Secondary hyperparathyroidism (SHPT) is a common and serious complication in patients with chronic kidney disease (CKD) [1]. This condition primarily presents as disturbances in calcium and phosphorus metabolism, musculoskeletal issues, sleep disorders, and pruritic skin [2]. Despite the development of new medications, parathyroidectomy (PTX) remains the definitive and effective treatment for SHPT. The Kidney Disease: Improving Global Outcomes (KDIGO) guidelines recommend PTX for patients with SHPT who are resistant to medical treatment [3,4]. Successful PTX can alleviate clinical symptoms, regulate calcium and phosphorus metabolism, and decrease serum intact parathyroid hormone (iPTH) levels [5]. Most notably, it can improve patients’ quality of life [6] and reduce the risk of cardiovascular mortality [7].
Hypocalcemia, a severe complication known to occur after PTX [5,8], is also referred to as hungry bone syndrome (HBS). It is considered a contributing factor to the readmission and mortality rates associated with PTX in patients with SHPT [9]. Therefore, maintaining a stable postoperative serum calcium level is vital. However, the current approach to postoperative calcium supplementation relies heavily on frequently monitoring serum calcium levels after surgery. Prior studies have attempted to develop models to estimate the total postoperative calcium requirements following PTX in patients with SHPT [10-12]. Regrettably, some of these models have not been verified, and others vary in their approach to calcium supplementation, making them challenging to apply clinically. Consequently, there is a need to establish a reliable model for predicting calcium requirements after PTX in SHPT patients. This study aimed to develop and validate a novel calculation model for estimating calcium requirements in dialysis patients following PTX.
This retrospective study carefully screened the hospital records of patients with SHPT who underwent a successful PTX between January 2014 and January 2022. All the dialysis patients with complete data of surgical treatment, who met the surgical indications, were included in the study. Indications for PTX include at least one of the following: (1) a high level of iPTH (>800 pg/mL), (2) uncontrolled hypercalcemia and hyperphosphatemia that is refractory to medical therapy, (3) detection of at least one enlarged parathyroid gland by imaging examinations. Exclusion criteria were as follows: patients with severe organ failure (such as heart failure, respiratory failure, liver failure, uremia and so on) that cannot tolerate surgery, patients with tumor, patients with serious coagulation dysfunction (activated partial thromboplastin time >37 seconds), pregnant women, and patients who were unable to tolerate general anesthesia. Patients with incomplete record of clinical and/or accessory examinations were excluded.
The research protocol was approved by the Institutional Review Board of Huashan Hospital, Fudan University (No. KY2018-369). All patients provided written informed consent for this study. Between 2014 and 2022, 119 patients with SHPT received PTX. Six of them were excluded due to incomplete records of clinical and/or accessory examinations, and 15 patients were excluded due to the type of operation. Ninety-eight patients were randomly divided into two groups: 78 of them were used as the model derivation group and 20 were used as the validation group. All patients were administered with routine intravenous calcium gluconate after PTX. Calcium supplementation was adjusted according to the serum calcium level. We calculated the total calcium requirements (both intravenous and oral calcium) for a week after surgery for each patient.
The clinical data recorded were age, sex, body mass index, durations of dialysis and SHPT. Additional biochemical data included preoperative alkaline phosphatase (ALP), serum creatinine, blood urea nitrogen, serum albumin, hemoglobin, potassium, Creactive protein (CRP), carbon dioxide binding capacity, preoperative and immediate postoperative serum calcium, phosphorous and iPTH. The resected glands’ weights and volumes, intravenous and oral calcium supplementation in one week, and days of intravenous calcium supplementation were faithfully recorded. All data were extracted from the hospital information system.
The specific surgical procedure is total PTX with autologous transplantation, performed by experienced surgeons (HW). The iPTH level would be at least 80% lower than preoperative level to be considered as a successful surgery. For parathyroid autologous transplantation, the gland was divided into 1 mm3 pieces, and 10 pieces were placed in a single muscle pocket of the brachioradialis muscle. Postoperatively, serum calcium, phosphorous and iPTH were measured within five minutes. Postoperative calcium supplementation was given in the form of intravenous calcium gluconate and oral calcium carbonate (CaCO3). Intravenous 5% calcium gluconate solution was initially started, then adjusted according to the serum calcium level measured every 6 hours. The intravenous calcium infusion was gradually replaced by the oral CaCO3 when serum calcium levels had stabilized. The oral calcium was calculated by assuming a fractional gastrointestinal absorption of 20% [13]. The total calcium requirements for a week after surgery was estimated.
Calcium supplementation of patients was under unified management. The principle of calcium supplementation is to maintain the serum calcium level within normal range. Calcium supplementation after surgery on the first day for reference: a central venous catheter was routinely recommended for intravenous calcium supplementation after operation. Intravenous calcium supplementation started immediately after surgery with 10% calcium gluconate and 5% dextrose injection restricted to a ratio of 1:1 and a speed of 30 mL/hr. The serum calcium level was closely monitored. The speed of intravenous calcium supplementation was adjusted or discontinued based on serum calcium levels. The protocol for the calcium supplement management is as followings. When the serum calcium level >3.0 mmol/L, calcium supplementation should be suspended. Calcium supplementation should be administered to a half of original when the serum calcium level between 2.5 and 3.0 mmol/L. When the serum calcium level between 2.0 and 2.5 mmol/L, we always maintain existing calcium supplementation. Intravenous calcium supplementation should be administered to the double of original when the serum calcium level <2.0 mmol/L or patients are symptomatic.
Based on our previous experience, intravenous calcium supplementation can be transitioned to oral calcium supplementation on the third or fourth day after surgery, which has a stable serum calcium level. CaCO3 is usually recommended 12–24 tablets a day (calcium 0.3 g/tablet). Concomitant use of active vitamin D and calcium preparations was also recommended. We prescribed rocaltrol 0.25–0.5 μg a day, and the individual dose was adjusted based on serum calcium levels and iPTH levels.
The model was derived from the derivation group data and the validation group was used to further test the accuracy of the model (Fig. 1). R2 was used to test the goodness-of-fit of the model, aiming for above 0.8 to guarantee a precise estimation [14]. Precision, bias, and accuracy were calculated to measure the performance of the model. Bias and precision are presented as the median and interquartile range (IQR) of the differences between actual calcium requirements and predicted calcium requirements. Accuracy is presented as the percentage of estimated value within 15 (P15), 30 (P30), and 50% (P50) of the measured value [15,16].
All analyses were performed using IBM SPSS ver. 21.0 (IBM Corp.) or GraphPad Prism software version 8.0 (GraphPad Software). The distribution of each parameter was detected using the Kolmogorov-Smirnov test. Quantitative data are presented as mean±standard deviation or median (IQR), while qualitative data are expressed as frequencies and percentages. Differences between groups were compared using unpaired t-test or the Mann-Whitney U-test for continuous variables and the chisquared test for categorical variables. To evaluate changes in biochemical parameters after PTX, paired t-test using preoperative and corresponding postoperative data was performed. Pearson and Spearman correlation coefficients were used to examine associations between calcium supplementation and other parameters. Among significant factors, a multiple linear regression analysis was performed to develop a model for the predicted total elemental calcium supplementation. Correlations between two variables were examined by scatter plot and Pearson’s correlation coefficient. P-values of less than 0.05 were considered statistically significant.
This study enrolled a total of 98 dialysis patients with SHPT who had undergone PTX. The mean age of the patients was 52.2± 11.1 years, and 39.8% (n=39) of them were male. The mean weekly requirement for total calcium was 13.26±5.39 g. The baseline characteristics of the two groups were comparable, as shown in Table 1.
To explore the associations between calcium requirements for 1 week post-surgery and other variables, Pearson correlation analysis was performed for normally distributed variables and Spearman rank correlation analysis was conducted for non-normally distributed variables (Table 2). Preoperative calcium and immediate postoperative calcium were inversely related to calcium supplementation following PTX. The variables that showed a positive correlation included preoperative phosphorus, iPTH, ALP, and CRP. The variables showed complex correlations. These variables were not entirely independent from each other, indicating the necessity to examine collinearity in multiple regression analysis.
The final model was constructed to estimate the total calcium requirement for the first week post-surgery. This was achieved by utilizing variables that demonstrated significant relationships in the stepwise multiple linear regression analysis (Table 3). The model is as follows:
Y=33.798–8.929×X1+0.190×X2–0.125×X3+0.002×X4+0.003×X5,
where Y=the calcium (g) requirement for 1 week after surgery, X1=immediate postoperative calcium (mmol/L), X2=preoperative CRP (mg/L), X3=age (years), X4=preoperative iPTH (pg/mL), and X5=preoperative ALP (U/L) (R2=0.8). The model was statistically significant (F=27.327, P<0.001). To detect collinearity, the variance inflation factor (VIF) and the tolerance (T) values were calculated. If the VIF value is higher than 10 or the T value is smaller than 0.10, then collinearity may have a trivial impact on the estimates of the parameters. Multicollinearity testing based on the VIF and the tolerance values was performed, and the results indicated the absence of multicollinearity problems.
The model was validated by calculating the predicted calcium requirements of the 20 patients in the validation group. The mean predicted total calcium requirement for 1 week after surgery was 12.36±4.88 g, while the actual total calcium supplementation during this period was 12.60±6.50 g (Fig. 2A). A scatter plot showing the relationship between actual and predicted calcium requirements is shown in Fig. 2B. A significant correlation between the actual and predicted data is evident from the scatter plot. The correlation coefficient between the two was 0.8, indicating that the model provided good predictive utility (R=0.8, P<0.001). The performance of the predicted calcium requirement model is shown in Table 4. The percentages of estimated values within P15, P30, and P50 of the measured values were 25%, 75%, and 95%, respectively. This model was applied successfully to the patients in the validation group.
SHPT is a common problem in patients with end-stage renal disease. This condition arises as the glomerular filtration rate declines, leading to a disruption in phosphate excretion that can result in hyperphosphatemia. Additionally, deficiencies in circulating calcium and vitamin D3 can also trigger an increase in iPTH secretion and parathyroid hyperplasia [2]. Numerous studies have indicated that successful PTX is a safe and effective treatment strategy for lowering serum calcium and phosphorus levels, thereby improving the prognosis [5,17].
Hypocalcemia is a common complication following PTX. Significant hypocalcemia can result in tetany, arrhythmia, and even death. The incidence rate of postoperative hypocalcemia in patients with SHPT has been reported to range from 28% to 100% [18]. Therefore, postoperative hypocalcemia is a significant concern for patients who have undergone PTX. Studies have shown that preoperative calcitriol treatment can reduce the need for intravenous calcium administration by 56% and decrease the duration of hospital stay by 50% [19]. Another study found that preoperative calcium and calcitriol supplementation was linked to a lower incidence of HBS and length of stay [20]. However, none of these methods are currently recognized as standard practice for patients with PTX. The KDIGO guidelines recommend initiating immediate postoperative intravenous calcium infusion to maintain normal levels after PTX. Once patients can take oral medications, the guidelines suggest a daily intake of 1 or 2 g of CaCO3 thrice per day and 2 μg/day of calcitriol to maintain normal calcium levels. KDIGO recommends measuring serum calcium levels every 4 to 6 hours for the first 48 to 72 hours, and then twice daily until stable calcium levels are observed [3,4]. Currently, postoperative calcium supplementation is still largely based on empirical calculations and frequent monitoring of serum calcium levels.
A few studies have attempted to model the prediction of total calcium requirements following PTX in patients with SHPT. One study from Korea proposed an equation to estimate calcium supplementation during hospitalization, demonstrating a correlation between calcium requirement and preoperative levels of ALP, iPTH, and changes in phosphorus levels at 48 hours postoperation [10]. However, this equation has not been validated. Another study developed prediction equations for various factors, including the average rate of decrease in postoperative serum corrected calcium concentration prior to calcium supplementation, the duration of intravenous calcium supplementation, the dosage of intravenous calcium supplementation, and the dosage of oral calcium supplementation. However, these equations have also not been validated [11]. Another study identified preoperative ALP and iPTH levels as predictors of calcium supplementation [12]. However, this model was designed solely to estimate intravenous calcium requirements during the initial 66 hours postoperation, a period when patients’ serum calcium levels may still be unstable. It is important to note that there are differences in baseline characteristics of patients and the administration of calcium requirements across these studies. Therefore, the present study was conducted to provide more precise treatment strategies for patients with SHPT.
The proposed model in this study is as follows: calcium requirement=33.798–8.929×immediate postoperative calcium+ 0.190×CRP–0.125×age+0.002×iPTH+0.003×ALP. The power of these characteristics was less than 0.05 (Table 3). In this study, the total calcium requirement was found to be positively correlated with CRP, preoperative iPTH and ALP, but negatively correlated with postoperative serum calcium and age. The predicted total calcium requirement for 1 week after surgery was 12.36±4.88 g, while the actual total calcium supplementation for 1 week after surgery was 12.60±6.50 g (P=0.895). The correlation coefficient between the actual and predicted calcium requirements was 0.8 (P<0.001), proving that this model provides accurate and valuable predictions.
Unlike previous studies that highlighted preoperative calcium as a risk factor [11,21], this study identified immediate postoperative calcium as a risk factor for postoperative hypocalcemia. At our center, we can typically determine immediate postoperative serum calcium levels within an average of 2 hours. This allows us to anticipate and plan for total calcium treatment in the 7 days following PTX.
Age has been identified as a risk factor for hypocalcemia in patients with primary hyperparathyroidism following PTX [22]. Studies have also shown a significant correlation between age and HBS in patients with uremia [23,24]. Our research found that younger patients required more calcium supplementation, suggesting that osteoblastic activity may be higher in this demographic. Consequently, the bone formation driven by osteoblasts post-PTX is more pronounced in younger patients, making hypocalcemia more probable and increasing the demand for calcium after surgery.
CRP is a widely recognized marker of nonspecific inflammation. Research has consistently shown that CRP levels tend to rise in patients with CKD [25]. In this study, we found a correlation between CRP levels and the calcium requirements of patients 1 week postoperation. This suggests that patients with infections may have increased calcium needs. In patients with SHPT, the proliferation of parathyroid gland cells and the excessive secretion of iPTH result in mineral and bone disorders. Following PTX, there is a rapid decrease in iPTH levels. This disrupts the balance of bone metabolism, leading to a rapid deposition of calcium into the bone [26]. These factors collectively contribute to hypocalcemia. Our findings align with the majority of previous studies, which reported that patients with higher iPTH levels are at an increased risk of hypocalcemia [27,28]. Numerous studies to date have reported an independent association between preoperative ALP levels and calcium requirements [29,30]. This is believed to be linked to increased osteoblast activity following PTX.
The predictor variables in this model can be readily obtained from clinical and biochemical data. The model was effectively applied to the validation group, indicating its potential to provide accurate and personalized calcium supplementation for patients post-PTX. Patients with estimated calcium supplementation above the mean (13.43 g) should have their serum calcium levels monitored more frequently after surgery, as their calcium needs are higher. Conversely, if a patient’s estimated calcium supplementation is significantly below average, they may require less calcium supplementation in the initial treatment following surgery. This could successfully prevent hypercalcemia and the inhibitory effect on the survival of the parathyroid autograft. Therefore, understanding total calcium requirements post-PTX is crucial for accurately supplementing calcium preparation and improving patient prognosis.
The present study also has some limitations. First, the results in this retrospective study may have been influenced by selection bias. Secondly, there was a lack of additional patients for external validation since all the data were from a single center. External validation data could be used to validate this model. Further multicenter studies with larger sample sizes are needed to validate and, if necessary, modify the predictive model developed herein.
In conclusion, we developed a new model to quantify postoperative calcium requirements, which could contribute to the evaluation of calcium balance and individualized therapy for hypocalcemia in patients after PTX. The model can be used to guide calcium supplementation treatment in future clinical practice.
▪ We established a novel model to quantify postoperative calcium requirements.
▪ The predictor variables in the model can be easily obtained from clinical and biochemical data.
▪ The model can contribute to assessing calcium balance and offer accurate, individualized calcium supplementation for patients following parathyroidectomy.
▪ Severe hypocalcemia can result in negative clinical outcomes, while over-supplementation of calcium can inhibit the survival of parathyroid autografts; therefore, determining the total calcium requirement following parathyroidectomy is crucial for accurately administering calcium supplements and improving patient prognosis.
ACKNOWLEDGMENTS
This research was supported by Shanghai Science and Technology Committee (19411967800, 21Y11904200), National Natural Science Foundation of China (81870501, 81400745).
REFERENCES
1. Tentori F, Blayney MJ, Albert JM, Gillespie BW, Kerr PG, Bommer J, et al. Mortality risk for dialysis patients with different levels of serum calcium, phosphorus, and PTH: the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis. 2008; Sep. 52(3):519–30.
2. Lau WL, Obi Y, Kalantar-Zadeh K. Parathyroidectomy in the management of secondary hyperparathyroidism. Clin J Am Soc Nephrol. 2018; Jun. 13(6):952–61.
3. National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003; Oct. 42(4 Suppl 3):S1–201.
4. Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl (2011). 2017; Jul. 7(1):1–59.
5. Zhang Y, Lu Y, Feng S, Zhan Z, Shen H. Evaluation of laboratory parameters and symptoms after parathyroidectomy in dialysis patients with secondary hyperparathyroidism. Ren Fail. 2019; Nov. 41(1):921–9.
6. van der Plas WY, Dulfer RR, Engelsman AF, Vogt L, de Borst MH, van Ginhoven TM, et al. Effect of parathyroidectomy and cinacalcet on quality of life in patients with end-stage renal disease-related hyperparathyroidism: a systematic review. Nephrol Dial Transplant. 2017; Nov. 32(11):1902–8.
7. Apetrii M, Goldsmith D, Nistor I, Siriopol D, Voroneanu L, Scripcariu D, et al. Impact of surgical parathyroidectomy on chronic kidney disease-mineral and bone disorder (CKD-MBD): a systematic review and meta-analysis. PLoS One. 2017; 12(11):e0187025.
8. Wetmore JB, Liu J, Do TP, Lowe KA, Ishani A, Bradbury BD, et al. Changes in secondary hyperparathyroidism-related biochemical parameters and medication use following parathyroidectomy. Nephrol Dial Transplant. 2016; Jan. 31(1):103–11.
9. Ferrandino R, Roof S, Ma Y, Chan L, Poojary P, Saha A, et al. Unplanned 30-day readmissions after parathyroidectomy in patients with chronic kidney disease: a nationwide analysis. Otolaryngol Head Neck Surg. 2017; Dec. 157(6):955–65.
10. Kang BH, Hwang SY, Kim JY, Hong YA, Jung MY, Lee EA, et al. Predicting postoperative total calcium requirements after parathyroidectomy in secondary hyperparathyroidism. Korean J Intern Med. 2015; Nov. 30(6):856–64.
11. Yang G, Zha X, Mao H, Yu X, Wang N, Xing C. Hypocalcemia-based prediction of hungry bone syndrome after parathyroidectomy in hemodialysis patients with refractory secondary hyperparathyroidism. J Int Med Res. 2018; Dec. 46(12):4985–94.
12. Ge P, Liu S, Sheng X, Li S, Xu M, Jiang J, et al. Serum parathyroid hormone and alkaline phosphatase as predictors of calcium requirements after total parathyroidectomy for hypocalcemia in secondary hyperparathyroidism. Head Neck. 2018; Feb. 40(2):324–9.
13. Coburn JW, Koppel MH, Brickman AS, Massry SG. Study of intestinal absorption of calcium in patients with renal failure. Kidney Int. 1973; Apr. 3(4):264–72.
14. Stevens LA, Schmid CH, Zhang YL, Coresh J, Manzi J, Landis R, et al. Development and validation of GFR-estimating equations using diabetes, transplant and weight. Nephrol Dial Transplant. 2010; Feb. 25(2):449–57.
15. Zhang W, Du Q, Xiao J, Bi Z, Yu C, Ye Z, et al. Modification and validation of the phosphate removal model: a multicenter study. Kidney Blood Press Res. 2021; 46(1):53–62.
16. Ma YC, Zuo L, Chen JH, Luo Q, Yu XQ, Li Y, et al. Modified glomerular filtration rate estimating equation for Chinese patients with chronic kidney disease. J Am Soc Nephrol. 2006; Oct. 17(10):2937–44.
17. Lim CT, Kalaiselvam T, Kitan N, Goh BL. Clinical course after parathyroidectomy in adults with end-stage renal disease on maintenance dialysis. Clin Kidney J. 2018; Apr. 11(2):265–9.
18. Jain N, Reilly RF. Hungry bone syndrome. Curr Opin Nephrol Hypertens. 2017; Jul. 26(4):250–5.
19. Alsafran S, Sherman SK, Dahdaleh FS, Ruhle B, Mercier F, Kaplan EL, et al. Preoperative calcitriol reduces postoperative intravenous calcium requirements and length of stay in parathyroidectomy for renal-origin hyperparathyroidism. Surgery. 2019; Jan. 165(1):151–7.
20. Maxwell AK, Shonka DC, Robinson DJ, Levine PA. Association of preoperative calcium and calcitriol therapy with postoperative hypocalcemia after total thyroidectomy. JAMA Otolaryngol Head Neck Surg. 2017; Jul. 143(7):679–84.
21. Ho LY, Wong PN, Sin HK, Wong YY, Lo KC, Chan SF, et al. Risk factors and clinical course of hungry bone syndrome after total parathyroidectomy in dialysis patients with secondary hyperparathyroidism. BMC Nephrol. 2017; Jan. 18(1):12.
22. Brasier AR, Nussbaum SR. Hungry bone syndrome: clinical and biochemical predictors of its occurrence after parathyroid surgery. Am J Med. 1988; Apr. 84(4):654–60.
23. Kritmetapak K, Kongpetch S, Chotmongkol W, Raruenrom Y, Sangkhamanon S, Pongchaiyakul C. Incidence of and risk factors for postparathyroidectomy hungry bone syndrome in patients with secondary hyperparathyroidism. Ren Fail. 2020; Nov. 42(1):1118–26.
24. Latus J, Roesel M, Fritz P, Braun N, Ulmer C, Steurer W, et al. Incidence of and risk factors for hungry bone syndrome in 84 patients with secondary hyperparathyroidism. Int J Nephrol Renovasc Dis. 2013; 6:131–7.
25. Menon V, Wang X, Greene T, Beck GJ, Kusek JW, Marcovina SM, et al. Relationship between C-reactive protein, albumin, and cardiovascular disease in patients with chronic kidney disease. Am J Kidney Dis. 2003; Jul. 42(1):44–52.
26. Liu S, Zhu W, Li S, Cui T, Li Z, Zhang B, et al. The effect of bovine parathyroid hormone withdrawal on MC3T3-E1 cell proliferation and phosphorus metabolism. PLoS One. 2015; 10(3):e0120402.
27. Yang M, Zhang L, Huang L, Sun X, Ji H, Lu Y. Factors predictive of critical value of hypocalcemia after total parathyroidectomy without autotransplantation in patients with secondary hyperparathyroidism. Ren Fail. 2016; Sep. 38(8):1224–7.
28. Tsai WC, Peng YS, Chiu YL, Wu HY, Pai MF, Hsu SP, et al. Risk factors for severe hypocalcemia after parathyroidectomy in prevalent dialysis patients with secondary hyperparathyroidism. Int Urol Nephrol. 2015; Jul. 47(7):1203–7.
29. Ding Y, Wang H, Zou Q, Jin Y, Zhang Z, Huang J. Factors associated with calcium requirements after parathyroidectomy in chronic kidney disease patients. Int Urol Nephrol. 2018; Mar. 50(3):535–40.
30. Tan PG, Ab Hadi IS, Zahari Z, Yahya MM, Wan Zain WZ, Wong MP, et al. Predictors of early postoperative hypocalcemia after total parathyroidectomy in renal hyperparathyroidism. Ann Surg Treat Res. 2020; Jan. 98(1):1–6.
Fig. 2.
Actual and predicted calcium requirements in 1-week postoperation. (A) The comparison between actual and predicted calcium requirements. (B) Scatter plot of actual and predicted calcium requirements.
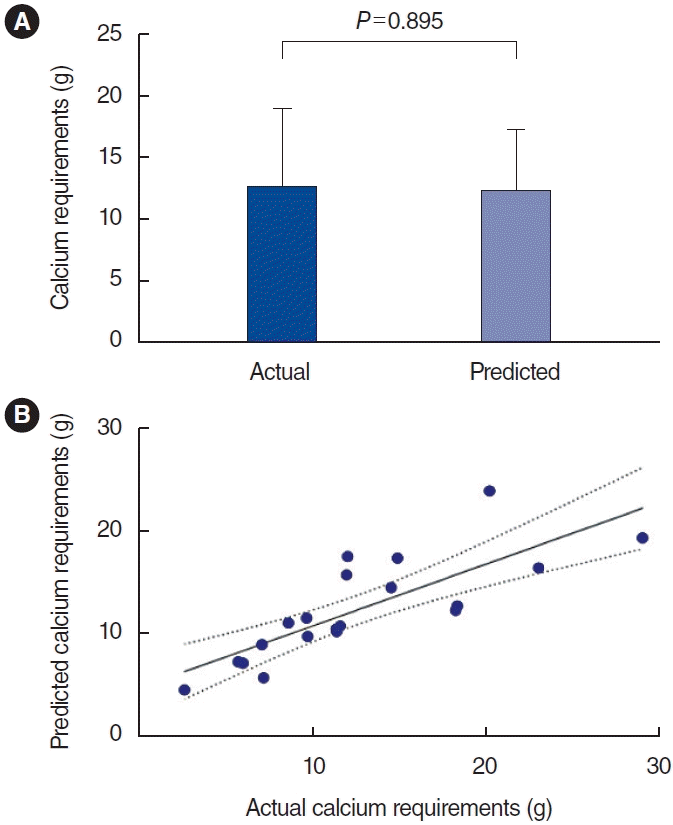
Table 1.
Clinical and laboratory characteristics of 98 patients
Table 2.
Correlations between total calcium requirements and characteristics by Pearson or Spearman correlation analysis
| Variable | Pearson or Spearman correlation analysis | P-value | ||
|---|---|---|---|---|
| Pearson R | ||||
| Age | –0.203 | 0.075 | ||
| BMI | 0.124 | 0.281 | ||
| Preoperative data | ||||
| Calcium | –0.242 | 0.033* | ||
| Phosphorus | 0.162 | 0.156 | ||
| Creatinine | –0.024 | 0.835 | ||
| BUN | 0.045 | 0.695 | ||
| Albumin | –0.073 | 0.526 | ||
| Hemoglobin | –0.163 | 0.154 | ||
| Potassium | 0.262 | 0.021* | ||
| Carbon dioxide combining power | –0.174 | 0.128 | ||
| Postoperative data | ||||
| Immediately | ||||
| Calcium | –0.596 | <0.001** | ||
| Phosphorus | 0.001 | 0.998 | ||
| Preoperative and postoperative differences | ||||
| Calcium | 0.534 | <0.001** | ||
| Phosphorus | 0.306 | 0.007** | ||
| Spearman R | ||||
| Sex | 0.018 | 0.877 | ||
| Dialysis duration | –0.012 | 0.920 | ||
| Duration of SHPT | –0.039 | 0.736 | ||
| Preoperative data | ||||
| iPTH | 0.638 | <0.001** | ||
| ALP | 0.546 | <0.001** | ||
| CRP | 0.280 | 0.013** | ||
| Postoperative data | ||||
| Immediately | ||||
| iPTH | 0.036 | 0.751 | ||
| Preoperative and postoperative differences | ||||
| iPTH | 0.588 | <0.001** | ||
| Parathyroid gland weight | 0.117 | 0.309 | ||
| Parathyroid gland volume | 0.179 | 0.118 | ||
| Parathyroid gland density | –0.160 | 0.161 | ||
Table 3.
Multiple linear regression model of predictors of calcium requirements
| Characteristic | β | Standard error | t-value | P-value |
|---|---|---|---|---|
| Constant | 33.798 | 7.046 | 4.797 | |
| Immediate postoperative calcium | –8.929 | 2.474 | –3.610 | <0.001** |
| CRP | 0.190 | 0.033 | 5.719 | <0.001** |
| Age | –0.125 | 0.045 | –2.766 | 0.009** |
| Preoperative iPTH | 0.002 | 0.001 | 2.297 | 0.027* |
| Preoperative ALP | 0.003 | 0.001 | 2.079 | 0.044* |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download