Introduction
Materials and Methods
1. Study population and treatment
2. Post-chemoradiotherapy pulmonary complications
3. Prognostic endpoints and statistics
Results
1. Patient characteristics
Table 1
Values are presented as median (range) or number (%). 3D-CRT, three-dimensional conformal radiation therapy; AJCC, American Joint Committee on Cancer; DLCO, diffusing capacity of carbon monoxide; ECOG, Eastern Cooperative Oncology Group; EQD2, equivalent dose in 2 Gy fractions; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; PCPC, post-chemoradiotherapy pulmonary complication; PTV, planning target volume; VMAT, volumetric modulated arc therapy.
2. Adverse events
Table 2
| Grade | Symptomatic radiation pneumonitis | Pneumonia | Anemia | Leukopenia | Neutropenia | Lymphocytopenia | Thrombocytopenia | Hypoalbuminemia |
|---|---|---|---|---|---|---|---|---|
| 1 | -a) | -b) | 215 (68.0) | 90 (28.5) | 37 (11.7) | 5 (1.6) | 78 (24.7) | 33 (10.4) |
| 2 | 63 (19.9) | 13 (4.1) | 77 (24.4) | 107 (33.9) | 59 (18.7) | 48 (15.1) | 10 (3.2) | 32 (10.1) |
| 3 | 10 (3.2) | 16 (5.0) | 0 | 56 (17.7) | 35 (11.1) | 179 (56.5) | 5 (1.6) | 2 (0.6) |
| 4 | 3 (0.9) | 4 (1.3) | 0 | 7 (2.2) | 4 (1.3) | 82 (25.9) | 1 (0.3) | 0 |
| 5 | 9 (2.8) | 10 (3.2) | 0 | -b) | -b) | -b) | -b) | 0 |
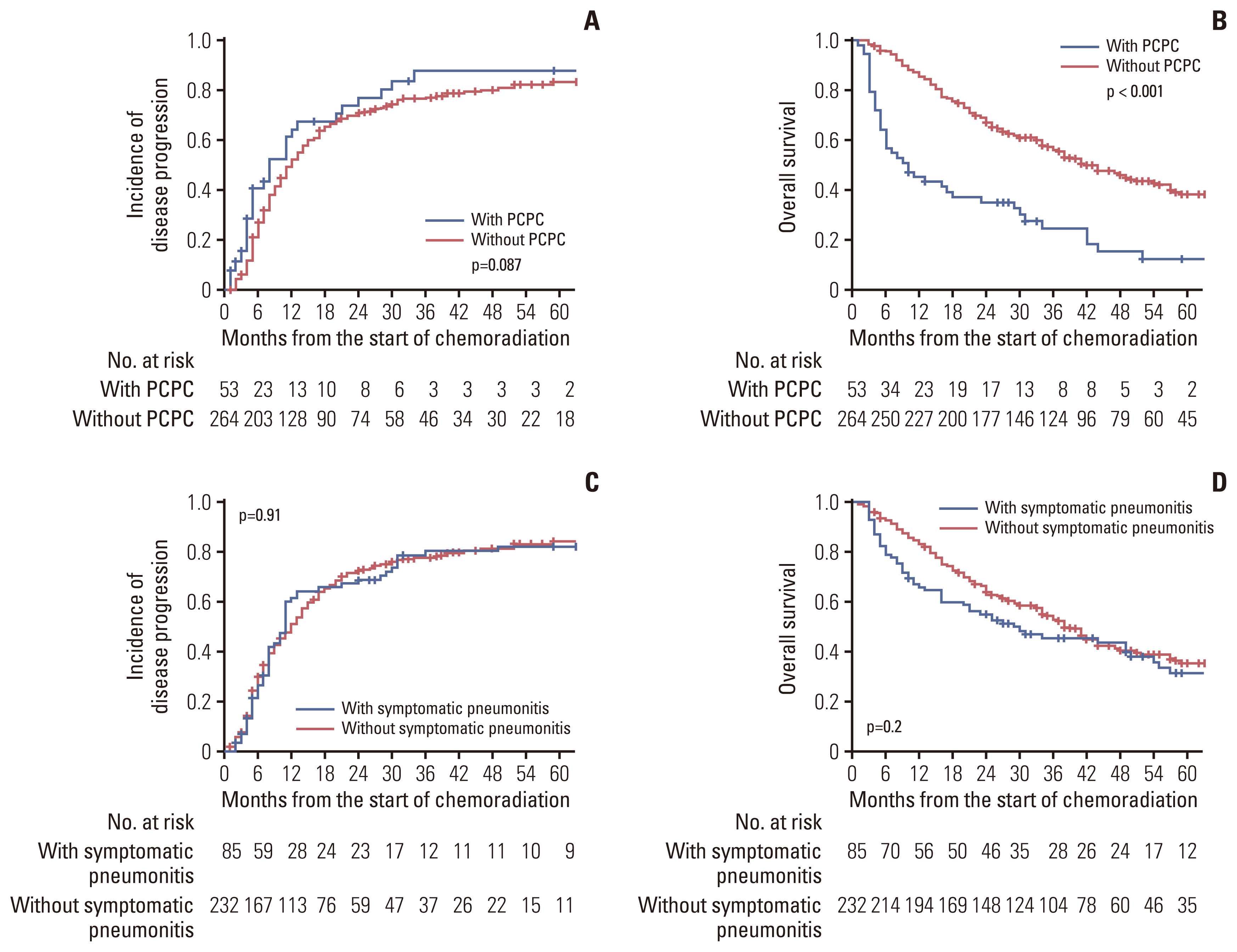
3. Association of post-chemoradiotherapy pulmonary complications with prognostic endpoints
4. Prediction of post-chemoradiotherapy pulmonary complications
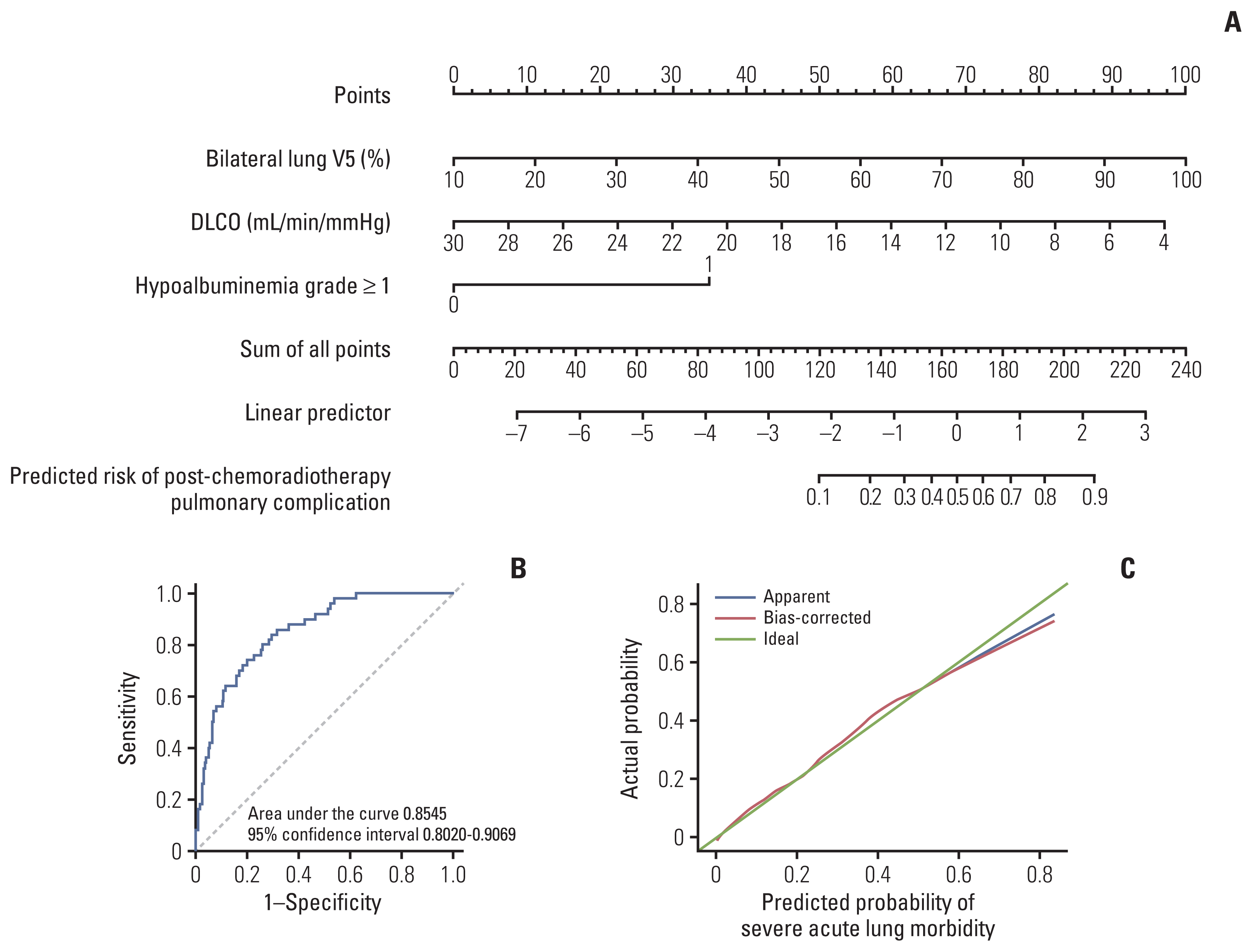 | Fig. 2Prediction nomogram for post-chemoradiotherapy pulmonary complications. Nomogram to predict the probability of post-chemoradiotherapy pulmonary complications (A), receiver operating characteristic curve (B), and calibration plot with bootstrapping of 100 repetitions for the nomogram (C). DLCO, diffusing capacity of carbon monoxide. |
Table 3
| Characteristic (comparison vs. reference) | Univariate analysis | Multivariate analysisa) | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | Adjusted p-value | OR | 95% CI | p-value | |
| Age at diagnosis (continuous, per a year) | 1.050 | 1.015–1.088 | 0.019 | 1.007 | 0.959–1.056 | 0.790 |
| Sex (female vs. male) | 0.782 | 0.306–1.751 | 0.767 | - | - | - |
| Amount of smoking (continuous, per pack-year) | 1.013 | 1.001–1.026 | 0.083 | 1.010 | 0.992–1.028 | 0.248 |
| Body mass index (continuous, per kg/m2) | 0.985 | 0.893–1.083 | 0.834 | - | - | - |
| Histology (others vs. adenocarcinoma) | 1.156 | 0.639–2.123 | 0.771 | - | - | - |
| FEV1 (continuous, L) | 0.660 | 0.401–1.061 | 0.162 | - | - | - |
| FEV1, percentage predicted value (continuous, per 10%) | 0.999 | 0.862–1.160 | 0.994 | - | - | - |
| FVC (continuous, per L) | 0.628 | 0.415–0.936 | 0.062 | 0.741 | 0.398–1.346 | 0.333 |
| FVC, percentage predicted value (continuous, per 10%) | 0.916 | 0.757–1.110 | 0.545 | - | - | - |
| FEV1/FVC, percentage predicted value (continuous, per 10%) | 1.075 | 0.821–1.427 | 0.769 | - | - | - |
| DLCO (continuous, per mL/min/mmHg) | 0.801 | 0.729–0.872 | < 0.001 | 0.855 | 0.743–0.974 | 0.022 |
| DLCO, percentage predicted value (continuous, per 10%) | 0.662 | 0.544–0.793 | < 0.001 | - | - | - |
| Regimen of concurrent chemotherapy (others vs. carboplatin and paclitaxel) | 0.711 | 0.341–1.388 | 0.523 | - | - | - |
| Radiation therapy technique (3D-CRT vs. VMAT) | 0.499 | 0.264–0.915 | 0.064 | 0.671 | 0.294–1.498 | 0.333 |
| EQD2 of prescribed dose (continuous, per Gy) | 0.942 | 0.885–0.999 | 0.098 | 0.949 | 0.874–1.021 | 0.184 |
| Bilateral lung mean dose (continuous, per Gy) | 1.178 | 1.081–1.293 | 0.001 | - | - | - |
| Bilateral lung V5 (continuous, per 10%) | 1.692 | 1.386–2.095 | < 0.001 | 1.872 | 1.336–2.699 | < 0.001 |
| Bilateral lung V20 (continuous, per 10%) | 2.358 | 1.548–3.705 | 0.001 | - | - | - |
| Bilateral lung volume (continuous, per 100 cm3) | 0.976 | 0.939–1.012 | 0.323 | - | - | - |
| Heart mean dose (continuous, per Gy) | 1.065 | 1.031–1.102 | 0.001 | 0.969 | 0.918–1.020 | 0.231 |
| Size of PTV (continuous, per 100 cm3) | 1.175 | 1.039–1.327 | 0.027 | 0.938 | 0.781–1.118 | 0.479 |
| Duration of radiation therapy (continuous, per day) | 0.981 | 0.934–1.030 | 0.630 | - | - | - |
| Anemia grade ≥ 2 (yes vs. no) | 3.084 | 1.645–5.754 | 0.001 | 1.357 | 0.575–3.135 | 0.478 |
| Leukopenia grade ≥ 2 (yes vs. no) | 1.002 | 0.552–1.830 | 0.994 | - | - | - |
| Neutropenia grade ≥ 2 (yes vs. no) | 1.097 | 0.570–2.047 | 0.834 | - | - | - |
| Lymphocytopenia grade ≥ 3 (yes vs. no) | 3.005 | 1.159–10.27 | 0.085 | 1.067 | 0.323–4.311 | 0.920 |
| Thrombocytopenia grade ≥ 2 (yes vs. no) | 0.714 | 0.110–2.663 | 0.773 | - | - | - |
| Hypoalbuminemia grade ≥ 1 (yes vs. no) | 7.186 | 3.806–13.77 | < 0.001 | 5.670 | 2.487–13.40 | < 0.001 |
3D-CRT, three-dimensional conformal radiation therapy; CI, confidence interval; DLCO, diffusing capacity of carbon monoxide; EQD2, equivalent dose in 2 Gy fractions; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; OR, odds ratio; PTV, planning target volume; VMAT, volumetric modulated arc therapy.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download