This article has been
cited by other articles in ScienceCentral.
PRESENTATION OF CASE 11
Dr. Ba-Ron Yang: A 31-year-old woman visited the emergency room (ER) with persistent nausea, vomiting, and abdominal pain for two days. Symptoms were mild when the episode started, but they did not improve despite taking over-the-counter drugs for abdominal discomfort.
The patient had been diagnosed with type 2 diabetes at the age of 19 and was receiving oral combination therapy. The types and doses of the drugs were metformin 850 mg bid, glimepiride 4 mg bid, and empagliflozin 10 mg qd. The patient had undergone photocoagulation for progressive proliferative diabetic retinopathy in both eyes six months and three months prior to the hospital visit. When the patient had started receiving treatment for eye complications, she experienced a sharp decrease in food intake due to a depressive mood. She denied smoking and drinking, and she reported that none of the people who ate with her complained of symptoms suggestive of food poisoning.
Examination of the patient revealed a blood pressure of 128/71 mmHg, temperature of 36.3 C, pulse of 82 beats per minute, respiratory rate of 20 breaths per minute, and an oxygen saturation rate of 99%. She was alert and oriented. Her abdominal pain was diffuse and could not be localized. Abdominal palpation revealed soft and flat findings, unclear tenderness, and no rebound tenderness. The body mass index was 23.8 kg/m2 (160.0 cm and 61.1 kg). All other physical findings were normal.
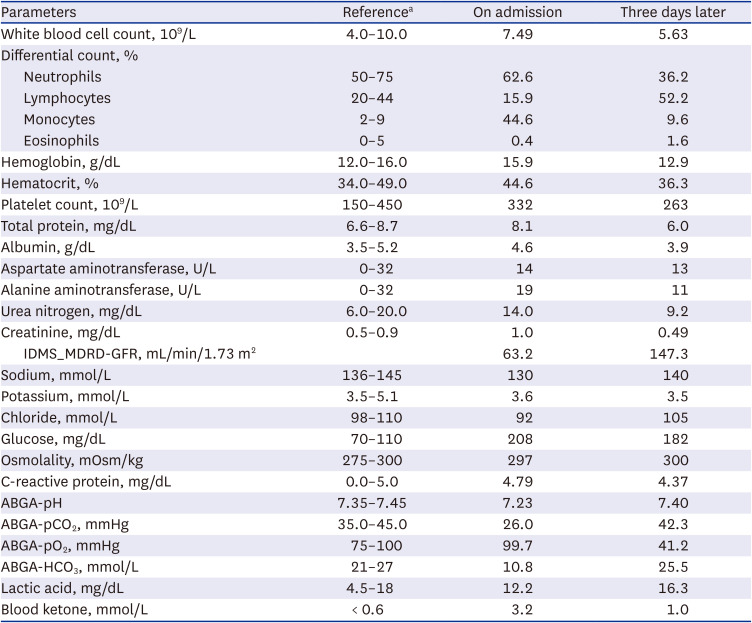
In the initial blood test, leukocytosis was not observed, C-reactive protein was normal, and both liver function and kidney function tests were within the normal range. Although the initial serum glucose level was 208 mg/dL, which was in the near normal range, arterial blood gas analysis (ABGA) showed metabolic acidosis with a pH of 7.23; pCO
2, 26.0 mmHg; pO
2, 99.7 mmHg; and HCO
3, 10.8 mmol/L. In the patient’s urine analysis (UA), pyuria was absent, but large amounts of glucose and ketone were identified. Serum electrolyte was confirmed as 130-3.6-92 mmol/L, and the calculated anion gap was 28 mmol/L, confirming high-anion gap acidosis. Also, lactic acid was within the normal range of 10.0 mg/dL, but blood ketone was elevated to 3.2 mmol/L (
Table 1).
Table 1
Patient’s routine blood chemistries
|
Parameters |
Referencea
|
On admission |
Three days later |
|
White blood cell count, 109/L |
4.0–10.0 |
7.49 |
5.63 |
|
Differential count, % |
|
|
|
|
Neutrophils |
50–75 |
62.6 |
36.2 |
|
Lymphocytes |
20–44 |
15.9 |
52.2 |
|
Monocytes |
2–9 |
44.6 |
9.6 |
|
Eosinophils |
0–5 |
0.4 |
1.6 |
|
Hemoglobin, g/dL |
12.0–16.0 |
15.9 |
12.9 |
|
Hematocrit, % |
34.0–49.0 |
44.6 |
36.3 |
|
Platelet count, 109/L |
150–450 |
332 |
263 |
|
Total protein, mg/dL |
6.6–8.7 |
8.1 |
6.0 |
|
Albumin, g/dL |
3.5–5.2 |
4.6 |
3.9 |
|
Aspartate aminotransferase, U/L |
0–32 |
14 |
13 |
|
Alanine aminotransferase, U/L |
0–32 |
19 |
11 |
|
Urea nitrogen, mg/dL |
6.0–20.0 |
14.0 |
9.2 |
|
Creatinine, mg/dL |
0.5–0.9 |
1.0 |
0.49 |
|
IDMS_MDRD-GFR, mL/min/1.73 m2
|
|
63.2 |
147.3 |
|
Sodium, mmol/L |
136–145 |
130 |
140 |
|
Potassium, mmol/L |
3.5–5.1 |
3.6 |
3.5 |
|
Chloride, mmol/L |
98–110 |
92 |
105 |
|
Glucose, mg/dL |
70–110 |
208 |
182 |
|
Osmolality, mOsm/kg |
275–300 |
297 |
300 |
|
C-reactive protein, mg/dL |
0.0–5.0 |
4.79 |
4.37 |
|
ABGA-pH |
7.35–7.45 |
7.23 |
7.40 |
|
ABGA-pCO2, mmHg |
35.0–45.0 |
26.0 |
42.3 |
|
ABGA-pO2, mmHg |
75–100 |
99.7 |
41.2 |
|
ABGA-HCO3, mmol/L |
21–27 |
10.8 |
25.5 |
|
Lactic acid, mg/dL |
4.5–18 |
12.2 |
16.3 |
|
Blood ketone, mmol/L |
< 0.6 |
3.2 |
1.0 |
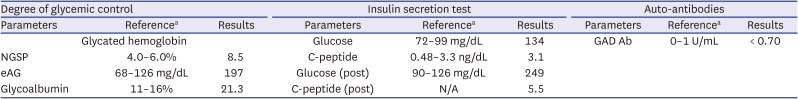
Chest X-ray and abdominal computed tomography (CT) findings were normal, and electrocardiogram (ECG) showed no specific findings other than sinus tachycardia. Insulin secretion was preserved with fasting C-peptide 3.07 ng/mL, and anti-glutamic acid decarboxylase (GAD) antibody was undetectable (
Table 2).
Table 2
Laboratory tests to evaluate the patient’s diabetes
|
Degree of glycemic control |
Insulin secretion test |
Auto-antibodies |
|
Parameters |
Referencea
|
Results |
Parameters |
Referencea
|
Results |
Parameters |
Referencea
|
Results |
|
Glycated hemoglobin |
Glucose |
72–99 mg/dL |
134 |
GAD Ab |
0–1 U/mL |
< 0.70 |
|
NGSP |
4.0–6.0% |
8.5 |
C-peptide |
0.48–3.3 ng/dL |
3.1 |
|
|
eAG |
68–126 mg/dL |
197 |
Glucose (post) |
90–126 mg/dL |
249 |
|
Glycoalbumin |
11–16% |
21.3 |
C-peptide (post) |
N/A |
5.5 |
DIFFERENTIAL DIAGNOSIS
Dr. Jinyoung Kim: According to the patient’s symptoms and medical history, it is necessary to consider the following diagnoses.
Food poisoning/Acute gastroenteritis/Appendicitis
Since the patient complained of gastrointestinal symptoms, food poisoning, gastroenteritis, or appendicitis could be suspected. In the case of severe diarrhea, the condition itself can cause metabolic acidosis, but the patient did not complain of symptoms other than nausea and abdominal discomfort. These diseases were excluded based on the lack of suspicious food intake, normal inflammation indicators in blood chemistry, and normal CT findings.
Shock
Metabolic acidosis can be caused by cardiogenic or septic shock because tissue hypoperfusion in these pathological conditions can induce lactic acidosis. However, the patient’s vital signs were stable throughout the ER stay. ECG and cardiac markers in blood studies were also reported to be in the normal range. Also, lactic acid was found to be normal in the ABGA.
Latent autoimmune diabetes in adults (LADA)
In particular, the patient had a history of diabetes, and her initial blood ketone level was positive; diabetic ketoacidosis (DKA) was diagnosed. LADA is initially diagnosed as type 2 diabetes, but the autoimmune destruction of the pancreas progresses slowly and worsens over months to years to form type 1 diabetes. Therefore, LADA is occasionally diagnosed with DKA caused by an insulin deficiency that has progressed under treatment with an oral hypoglycemic agent. The patient had preserved insulin secretion ability and was negative for anti-GAD antibodies; thus, DKA in type 1 diabetes and LADA were ruled out.
Finally, SGLT2 inhibitor-associated euglycemic DKA was diagnosed considering her relatively low initial glucose level and medical history of oral hypoglycemic agents including an SGLT2 inhibitor.
PROGRESS
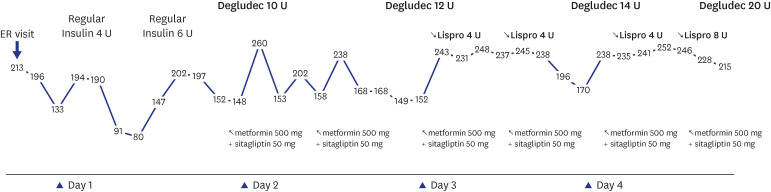
Dr. Ba-Ron Yang: The patient improved with 3 days of supportive care including hydration and insulin treatment (
Fig. 1).
Fig. 1
Changing trend of strip blood sugar tests and treatment during hospitalization.
ER = emergency room, U = units.
FINAL DIAGNOSIS
Dr. Ki-Ho Song: SGLT2 inhibitor-associated euglycemic DKA.
DISCUSSION
Dr. Seung-Hwan Lee: With recent introduction of SGLT2 inhibitors, cases of euglycemic DKA are rapidly increasing.
1 Is there a reason this drug causes these adverse effects?
Dr. Jinyoung Kim: The SGLT2 inhibitor induces glycosuria by blocking SGLT2 in the kidney proximal tubule, which is responsible for reabsorption of 80–90% of glucose filtered from the glomerulus. While this condition has the benefit of weight loss, it can also precipitate carbohydrate-deficit that can cause a state of increased counter-regulatory hormone. Additionally, SGLT2 inhibitors directly stimulate pancreatic alpha-cells to increase glucagon production. An increase in the glucagon-to-insulin ratio is a mechanism for increased ketone production.
2 Meanwhile, during the use of SGLT2 inhibitors, human metabolism oxidizes lipids instead of glucose, which can lead to a mechanism that helps with weight loss but increases the risk of ketoacidosis.
3 Finally, SGLT2 inhibitors also inhibit ketone re-absorption. Their use may lead to exacerbation of ketoacidosis in the presence of ketone-producing medical conditions such as dietary restrictions, heavy alcohol use, prolonged exercise, and other acute illness.
4 In a previous study, the risk of DKA increased more than three times when using SGLT2 inhibitors compared to the patient group using DPP4 inhibitors. The researchers reported that the effect was produced not by one specific drug but by the class of SGLT2 inhibitors.
5
Dr. Seung-Hwan Lee: Are there patients at high-risk for SGLT2 inhibitor-associated euglycemic DKA?
Dr. Yeoree Yang: In patients with type 1 diabetes, the risk of DKA increases more than five-fold when SGLT2 inhibitors are used, and euglycemia has been confirmed in more than half of the cases.
6 Insulin secretion can be reduced in type 2 diabetes patients with long disease duration or high insulin requirement. Therefore, caution is needed when prescribing SGLT2 inhibitors to patients with severely decreased beta cell function.
Dr. Chaiho Jeong: Because of the mechanism by which drugs induce glycosuria, genitourinary infections are on the rise as another common side effect. According to a previous study using Korean claims data, the use of SGLT2 inhibitors significantly increased genital and urinary tract infections, and these side effects were reported to occur more commonly in older patients and in females.
7 Therefore, it is necessary to be cautious in prescribing drugs of the SGLT2 inhibitor class to elderly women and in cases with a history of previous genitourinary infection.
Dr. Mee Kyoung Kim: In clinics, UA can be performed periodically to help monitor patient compliance and adverse effects. If the patient is taking the medication as prescribed, glycosuria is clearly identified, and early genitourinary infection and ketosis can be detected by examining pyuria and ketone levels. In addition, patients themselves can home-monitor ketonemia. There is a strip method to measure glucose and blood ketone together and a breath analyzer that detects ketones in the patient’s exhalation. Currently, SGLT2 inhibitors are not approved for type 1 diabetic patients in Korea, but home-monitoring of ketonemia is recommended in some other countries for the off-label use of this drug in type 1 diabetic patients.
8
Dr. Seung-Hwan Lee: Is there a way to prevent euglycemic DKA in patients using SGLT2 inhibitors?
Dr. Yeoree Yang: Physicians should advise patients to drink plenty of water while taking the medication. In addition, it is necessary to educate patients to stop medications when fasting for a long time, exercising for prolonged periods, or suffering from a catabolic status such as acute infectious disease with fever.
9 In the case of a planned surgery, it is necessary to discontinue the drug for at least three days before the operation.
10
CONCLUSION
Dr. Ki-Ho Song: Recently, SGLT2 inhibitors have been widely used, because they have many advantages such as weight loss and prevention of heart and kidney disease. However, euglycemic DKA is a dangerous adverse effect of this class of drug that can lead to missed diagnosis, as it is not accompanied by hyperglycemia. Therefore, it is important to fully understand this pathological condition for accurate diagnosis and immediate management.
ACKNOWLEDGMENTS
The Case Conference section is prepared from the monthly case conference of the Department of Internal Medicine, The Catholic University of Korea College of Medicine, Seoul, Korea.
References
1. Taylor SI, Blau JE, Rother KI. SGLT2 inhibitors may predispose to ketoacidosis. J Clin Endocrinol Metab. 2015; 100(8):2849–2852. PMID:
26086329.

2. Nasa P, Chaudhary S, Shrivastava PK, Singh A. Euglycemic diabetic ketoacidosis: a missed diagnosis. World J Diabetes. 2021; 12(5):514–523. PMID:
33995841.

3. Daniele G, Xiong J, Solis-Herrera C, Merovci A, Eldor R, Tripathy D, et al. Dapagliflozin enhances fat oxidation and ketone production in patients with type 2 diabetes. Diabetes Care. 2016; 39(11):2036–2041. PMID:
27561923.

4. Monami M, Nreu B, Zannoni S, Lualdi C, Mannucci E. Effects of SGLT-2 inhibitors on diabetic ketoacidosis: a meta-analysis of randomised controlled trials. Diabetes Res Clin Pract. 2017; 130:53–60. PMID:
28570924.

5. Douros A, Lix LM, Fralick M, Dell’Aniello S, Shah BR, Ronksley PE, et al. Sodium-glucose cotransporter-2 inhibitors and the risk for diabetic ketoacidosis: a multicenter cohort study. Ann Intern Med. 2020; 173(6):417–425. PMID:
32716707.

6. Wolfsdorf JI, Ratner RE. SGLT inhibitors for type 1 diabetes: proceed with extreme caution. Diabetes Care. 2019; 42(6):991–993. PMID:
31110116.

7. Yang H, Choi E, Park E, Na E, Chung SY, Kim B, et al. Risk of genital and urinary tract infections associated with SGLT-2 inhibitors as an add-on therapy to metformin in patients with type 2 diabetes mellitus: a retrospective cohort study in Korea. Pharmacol Res Perspect. 2022; 10(1):e00910. PMID:
35005849.

8. Danne T, Garg S, Peters AL, Buse JB, Mathieu C, Pettus JH, et al. International consensus on risk management of diabetic ketoacidosis in patients with type 1 diabetes treated with sodium-glucose cotransporter (SGLT) inhibitors. Diabetes Care. 2019; 42(6):1147–1154. PMID:
30728224.

9. Goldenberg RM, Berard LD, Cheng AY, Gilbert JD, Verma S, Woo VC, et al. SGLT2 inhibitor-associated diabetic ketoacidosis: clinical review and recommendations for prevention and diagnosis. Clin Ther. 2016; 38(12):2654–2664.e1. PMID:
28003053.

10. Mehta PB, Robinson A, Burkhardt D, Rushakoff RJ. Inpatient perioperative euglycemic diabetic ketoacidosis due to sodium-glucose cotransporter-2 inhibitors - lessons from a case series and strategies to decrease incidence. Endocr Pract. 2022; 28(9):884–888. PMID:
35753675.




