Abstract
Objectives
Age-related hearing loss (ARHL), or presbycusis, is caused by disorders of sensory hair cells and auditory neurons. Many studies have suggested that the accumulation of mitochondrial DNA damage, the production of reactive oxygen species, noise, inflammation, and decreased antioxidant function are associated with subsequent cochlear senescence in response to aging stress. Long non-coding RNA (lncRNA) has been reported to play important roles in various diseases. However, the function of lncRNA in ARHL remains unclear. In this study, we analyzed the common expression profiles of messenger RNA (mRNA) and lncRNA through ARHL-related RNA-sequencing datasets.
Methods
We selected and downloaded three different sets of RNA-sequencing data for ARHL. We performed differential expression analysis to find common mRNA and lncRNA profiles in the cochleae of aged mice compared to young mice. Gene Ontology (GO) analysis was used for functional exploration. Real-time quantitative reverse-transcription polymerase chain reaction (qRT-PCR) was performed to validate mRNAs and lncRNAs. In addition, we performed trans target prediction analysis with differentially expressed mRNAs and lncRNAs to understand the function of these mRNAs and lncRNAs in ARHL.
Results
We identified 112 common mRNAs and 10 common lncRNAs in the cochleae of aged mice compared to young mice. GO analysis showed that the 112 upregulated mRNAs were enriched in the defense response pathway. When we performed qRT-PCR with 1 mM H2O2-treated House Ear Institute-Organ of Corti 1 (HEI-OC1) cells, the qRT-PCR results were consistent with the RNA-sequencing analysis data. lncRNA-mRNA networks were constructed using the 10 common lncRNAs and 112 common mRNAs in ARHL.
Conclusion
Our study provides a comprehensive understanding of the common mRNA and lncRNA expression profiles in ARHL. Knowledge of ARHL-associated mRNAs and lncRNAs could be useful for better understanding ARHL and these mRNAs and lncRNAs might be a potential therapeutic target for preventing ARHL.
Age-related hearing loss (ARHL), or presbycusis, is a common problem affecting one-third of adults aged ≥65 years [1]. It is characterized by reduced hearing sensitivity and speech understanding, resulting from degeneration of the cochlea, auditory nerves, or both. ARHL can be affected by genetics, metabolism, noise, ototoxic medications, and inflammation [2]. For example, DCLK1, SLC28A3, CEP104, and PCDH20 are genes associated with hearing function in humans [3]. Reactive oxygen species (ROS) are generated by the production of superoxides and hydrogen peroxide (H2O2), and these have the capability of damaging DNA, leading to a loss of proteins and apoptosis in the cochlea [4]. Exposure to noise and ototoxic drugs can lead to cochlear cell degeneration and hearing loss [5]. Inflammation and inflammatory mediators contribute to an increased risk of hearing impairment [6]. ARHL contributes to social isolation, depression, and dementia in aged adults [7]. Therefore, it is important to explore the mechanism of ARHL and identify new therapeutic targets for clinical treatment to prevent hearing loss and improve quality of life.
While ARHL is considered a multifactorial disorder, many studies have focused mostly on protein-coding messenger RNAs (mRNAs). More recently, thousands of non-coding transcripts have been identified through high-throughput sequencing, and some of them have been shown to be functional. Long non-coding RNAs (lncRNAs) are a type of RNA, generally defined as transcripts of more than 200 nucleotides that are not translated into protein [8]. Many studies reported that lncRNAs can control gene regulation via epigenetic modifications, recruit transcription factors, and interact with splicing factors [9]. Some lncRNAs contain miRNA within their sequences and are referred as miRNA host genes lncRNAs (lnc-miRHGs). These lnc-miRHGs have been reported to serve as competing endogenous RNA (ceRNAs), DNA interactors, and protein interactors. [10]. lncRNAs play key roles in biological processes, and alteration of lncRNAs expression levels is associated with diseases such as cancer [9]. Recent studies have established a direct correlation between lncRNA regulation and ARHL in aged mice. The lncRNA Gm44593 can reduce ROS levels by regulating miR-29b/WNK1 signaling in ARHL [11]. The lncRNA AW112010 regulates mitochondrial function and biogenesis, leading to AMP-activated protein kinase (AMPK) activation in ARHL [12]. Downregulated expression of the lncRNA myocardial infarction associated transcript (MIAT) was found in aged hair cells, and MIAT could regulate the expression of SIRT1/PGC-1a by downregulating miR-29b [13]. In addition, upregulated Sirt1 expression by the lncRNA H19 regulates H2O2-induced cochlear hair cell apoptosis by inhibiting miR-653-5p [14]. Although the functions of a few lncRNAs in ARHL have been revealed, the common lncRNAs involved in the ARHL process remain unknown.
In this study, we investigated common mRNA and lncRNA profiles in ARHL through a bioinformatics analysis. In total, 112 common mRNAs and 10 common lncRNAs were identified as being differentially expressed (DE) in the cochleae of three different RNA-sequencing (RNA-seq) datasets. These findings may help identify effective targets for ARHL.
We searched the GEO database (Gene Expression Omnibus; https://www.ncbi.nlm.nih.gov/geo/) for datasets related to ARHL by using the keywords “aging,” “hearing loss,” and “presbycusis.” Three datasets for the cochlea (GSE127204, GSE196870, and PRJNA600057) were downloaded from European Nucleotide Archive (ENA). FASEQ files were downloaded directly from ENA using the IBM Aspera tool (v4.0.0.182279). FASTQ files were trimmed using Trimmomatic (v0.39) [15] to remove adaptors and low-quality reads. The trimmed reads were aligned and quantified to the Gencode vM26 mouse genome (https://www.gencodegenes.org/) [16] using Salmon (v1.4.0) with default settings [17].
Quantified transcript abundances using Salmon were converted to gene-level counts using the tximport (v1.6.3) [18] package in R. Differential expression analysis of mRNA and lncRNA between two conditions was performed using the DESeq2 (v1.30.1) [19] package. We selected the top DE genes (DEGs) of the mRNAs and lncRNAs from the mouse cochlea using the P-values calculated using DESeq2 (Supplementary Tables 1 and 2). Each RNA sequencing dataset was analyzed individually.
Venn diagram and heatmap of DE mRNAs and DE lncRNAs were generated using an online tools provided by VIB and Ghent University (https://bioinformatics.psb.ugent.be/webtools/Venn/) and ComplexHeatmap R package (v 2.6.2) [20]. Genome information on the lncRNAs was generated using the Gviz R package (v1.34.1).
To explore the functions of the DEGs in the cochlea of aged mice, Gene Ontology (GO) enrichment analysis was performed using DAVID Bioinformatics Resources 6.8 (https://david.ncifcrf.gov) using 79 upregulated and 33 downregulated genes. GO analysis involved three categories, namely biological processes (BPs), cellular components (CCs) and molecular function (MF) with P-value <0.05.
To construct the potential lncRNAs-mRNAs network, we performed Pearson correlation coefficient (PCC) analysis using the Hmisc R package (v4.7.0). PCC was calculated between the 10 lncRNAs and 112 mRNAs with cutoff values of |Pearson correlation| >0.9, P<0.05, and the lncRNA-mRNA regulatory network was structured using Cytoscape 3.8.2. Diamond shape represent lncRNAs and circular (ellipse) shape represent mRNAs. Red nodes represent upregulated lncRNAs or mRNAs, and blue nodes represent downregulated lncRNAs or mRNAs.
The House Ear Institute-Organ of Corti 1 (HEI-OC1) cells were cultured in high glucose Dulbecco’s Modified Eagle Medium (DMEM; Welgene, #11965092) containing 10% fetal bovine serum (Gibco; Thermo Fisher Scientific Inc., #10437028) without antibiotics at 33 °C and 10% CO2.
To confirm individual mRNA and lncRNA, total RNA was extracted from HEI-OC1 cells treated with 1 mM H2O2 for 6 hours using the RNAiso Plus Reagent (Takara) following the manufacturer’s protocol. cDNA was synthesized using the M-MLV Reverse Transcriptase, RNaseH⁺ premix kit EBT-1511 (Elpisbio) with random hexamers according to the manufacture’s protocol. Real-time PCR was performed with TB Green Premix Ex Taq (Tli RNaseH Plus, Takara Bio Inc.) using a Thermal Cycler Dice Real Time System III (Takara Bio Inc.). PCR analyses were as follows: 95 °C for 30 seconds followed by 40 cycles at 95 °C for 5 seconds and 60 °C for 30 seconds. Relative RNA amounts were calculated using the 2–ΔΔCT method. β-actin (ACTB) was used as an internal control for the relative quantitation of mRNAs. Primer details are listed in in Supplementary Table 3.
Data are presented as the mean±standard error of the mean, and statistical analyses were performed using unpaired Student t-test to complete the differential expression analysis of mRNAs and lncRNAs using real-time quantitative reverse-transcription polymerase chain reaction (qRT-PCR). Statistical significance was set at P<0.05.
To identify the important role of mRNA gene expression profiles in ARHL, we used publicly available GEO datasets to analyze the differentially expressed genes (DEGs) in the cochleae of aged mice compared to young mice (Table 1). In total, 112 common mRNAs (79 upregulated and 33 downregulated mRNAs) were identified in all three GEO datasets with a cutoff of |log2FC| ≥ 1.5 and P≤0.05. These were visualized using a Venn diagram (Fig. 1A) and heatmap (Fig. 1B). The upregulated and downregulated genes are listed in Supplementary Table 1. Next, we performed GO analysis with the 112 mRNAs to identify the relevant biological function and enriched signaling pathways using the DAVID website. The details of the GO list are presented in Supplementary Table 4. As shown in Fig. 1C and D, the GO analysis of BPs indicated that the 79 upregulated mRNAs participated in immune system processes, the defense response, the immune response, the innate immune response, and positive regulation of T cell activation. The GO analysis of CCs indicated that the genes participated in the major histocompatibility complex (MHC) class I peptide loading complex, the external side of the plasma membrane, the cell surface, the Golgi apparatus, and symbiont-containing vacuole membranes. The GO analysis of MFs indicated that the genes participated in peptide antigen binding, macromolecular complex binding, TAP2 binding, TAP1 binding, and T cell receptor binding (Fig. 1C). The 33 downregulated mRNAs were involved in the following BPs: modulation of inner ear development, cartilage development, tissue homeostasis, collagen fibril organization, and chondrocyte differentiation. In terms of CCs, genes were associated with the membrane, plasma membrane, postsynaptic density, cytoplasm, and cell projection. In terms of MFs, the genes participated in platelet-derived growth factor binding, identical protein binding, extracellular matrix structural constituent conferring tensile strength, and protein binding (Fig. 1D).
We identified 112 common mRNAs in aged cochleae from the RNA-seq data. Among these encoding defense and immune response genes, Ccl2 (chemokine [C-C motif] ligand 2), Ccl5 (chemokine [C-C motif] ligand 5), and ALOX15 (Arachidonate 15-Lipoxygenase) were upregulated in aged cochleae in the RNA-seq data (Fig. 2A). The inflammation-related gene Alox15 was upregulated in the cochleae of aged C57BL/6 mice [21].
Regarding the regulation of cell communication genes, Col2a1 (collagen type ii alpha 1 chain), Dnm1 (dynamin 1), Eps8l2 (epidermal growth factor receptor kinase substrate 8-like protein 2) and Itsn1 (intersectin 1) were downregulated in aged cochleae in the RNA-seq data (Fig. 2B).
To validate the DE mRNA results, H2O2-treated HEI-OC1 cells were used. H2O2-treated HEI-OC1 cells have been used as an in vitro system to study cellular and molecular mechanisms involved in ARHL [23-26]. We selected three defense responserelated genes and two genes related to the regulation of cell communication and performed qRT-PCR with 1 mM H2O2-treated HEI-OC1 cells. The primer details are presented in Supplementary Table 3. Ccl2, Ccl5, and Ccl7 were significantly upregulated in H2O2-treated HEI-OC1 cells compared with their expression in the control group. Itsn1 and Aak1 were significantly downregulated in H2O2-treated HEI-OC1 cells (Fig. 2C). These qRT-PCR results were consistent with the RNA-seq data. In conclusion, the results showed that the RNA-seq analysis data were reliable, with high specificity for ARHL.
To identify the lncRNAs associated with ARHL, we analyzed three RNA-seq datasets. We identified a total of 10 lncRNAs, including seven upregulated and three downregulated lncRNAs (P<0.05 and |log2FC|>1.0) (Fig. 3A) in the cochlear of aged mice compared to those in young mice. These DE lncRNAs were visualized using Venn diagrams and heatmaps (Fig. 3A and B, Supplementary Table 2). In addition, the genomic information of DE lncRNAs was displayed using the Gviz R package (Fig. 3C).
Some lncRNAs harbor miRNA sequences and act as lnc-miRHGs. Zhang et al. [27] revealed that the extent and specificity of miRNA expression during cochlear aging contributes to the progression of ARHL. Among the identified lncRNAs, Meg3 and Rian were demonstrated to be lnc-miRHGs. Based on genomic information of the identified lncRNAs, MEG3 contains the miR1906-1 and miR770 sequences, and Rian contains the miR1188, miR341, and miR370 sequences. The functions and regulatory mechanisms of these miRNAs in ARHL are unknown. Therefore, these miRNAs require further investigation.
We identified 10 common lncRNAs in aged cochleae in the RNA-seq data. Bar graphs were generated to represent upregulated and downregulated lncRNAs in all RNA-seq datasets (Fig. 4A and B). Among them, Rian has already been reported as an ARHL-associated lncRNA [12]. Consistent with the previous report, Rian expression was significantly downregulated in aged cochleae in our analysis.
To confirm the DE lncRNA results, we performed qRT-PCR validation in H2O2-treated HEI-OC1 cells. As shown in Fig. 4C, the lncRNAs SNHG17, ZFAS1, C4a, SNHG15 and SNHG1 were upregulated in H2O2-treated HEI-OC1 cells, whereas MEG3, 1500004A13Rik, and Rian were downregulated. These qRT-PCR results were all in accordance with the RNA-seq data, except Gm16094 and Gm11131. However, the relationship between these lncRNAs and ARHL regulation remains unknown.
To demonstrate which lncRNAs are involved in ARHL, we chose 10 lncRNAs and 112 mRNAs to construct the lncRNA-mRNA interaction network. We performed trans pattern analysis of the PCCs (P<0.05 and |Pearson correlation| >0.9), based on the RNA-seq analysis data using the Hmisc package. We identified 80 common co-expressed pairs of upregulated and downregulated lncRNAs in the three different RNA-seq datasets (Fig. 5, Supplementary Table 5). These co-expressed pairs were used to build a lncRNA-mRNA co-expression network using Cytoscape. In the co-expression network, C4a and Gm11131 showed the highest number of interactions.
ARHL is a common problem in aging societies worldwide. The primary pathology of ARHL includes the degeneration of hair cells, stria vascularis, and ganglion neurons in the cochlea, as well as the auditory nerve. Hearing loss caused by genetic factors and exposure to oxidative stress, noise, and ototoxic drugs not only affects hearing sensitivity and speech understanding, but also leads to social isolation, depression, and dementia in older adults. Therefore, understanding the mechanism of ARHL is important for its prevention.
In this study, we focused on the common expression profiles of mRNA and lncRNA in ARHL using a bioinformatics approach. From the mRNA profiles of young and aged mice, we identified 112 common mRNAs from three different RNA-seq datasets. Among them, upregulated mRNAs were related to immune system processes, the defense response, the immune response, the innate immune response, and positive regulation of T cell activation (Fig. 1C). These results closely align with those reported by Chen et al. [22], who revealed that a mouse model based on aging, noise exposure, and cisplatin ototoxicity shared hub genes involved in immune system processes, the inflammatory response, and apoptotic processes. Similarly, the 79 upregulated mRNAs identified in this study were involved in defense and immune responses. Increased levels of ROS in aging result in the accumulation of oxidative stress, leading to mitochondrial damage [28]. Oxidative stress is one of the pathologies linked to ARHL, and ROS can modulate immune response and inflammation by inducing oxidative stress [6,29].
We identified 10 common lncRNAs from three different RNA-seq datasets. Among them, Rian had been reported to be downregulated in the cochleae of aged mice. However, the other nine lncRNAs were not known to have any roles in ARHL. Small nucleolar RNA host genes (SNHGs) are a group of long noncoding RNAs, which are reported in many studies as being overexpressed in various cancers. With very few exceptions, the members of the SNHG family are recognized as inducing increased proliferation, cell cycle progression, invasion, and metastasis of cancer cells through interaction with methyltransferase and transcription factors and act as ceRNAs for miRNAs [30]. The lncRNA SNHG17 has been reported to enhance glioma cell proliferation, migration, and invasion by inducing zinc fingers and homeoboxes 1 (ZHX1) expression via inhibiting miR-23b-3p [31]. The lncRNA SNHG1 promotes cervical cancer progression by regulating NIMA-related kinase 2 (NEK2) expression via inhibiting miR-195 [32]. In addition, SNHG1 enhances cell proliferation, migration and invasion in esophageal cancer by regulating cell division cycle 42 (Cdc42) expression via inhibiting miR-195 [33]. lncRNA SNHG15 was increased in human cancers and can contribute to cell proliferation by acting as a sponge for miRNAs [34]. We found that the lncRNAs SNHG1, SNHG15, and SNHG17 were upregulated in both aged cochleae and H2O2-treated HEI-OC1 cells. mir-23b-3p and miR-195, which could act as ceRNAs of SNHG1 and SNHG17, were reported to be downregulated in the cochleae of aged C57BL/6 mice [27]. However, the functional roles of these miRNAs and their target genes in the cochleae are not known. In addition, the biological functions of the miRNA-containing lncRNAs MEG3 and Rian are still unknown (Fig. 3C). We identified a total of 80 trans-regulated target genes for upregulated and downregulated lncRNAs from the aged mouse cochleae (Fig. 5). This is the first step in understanding how lncRNAs regulate ARHL. The predicted target genes were highly enriched in biological processes, including immune system processes, the innate immune response, the defense response and the defense response to viruses. These results indicate that the identified lncRNAs and miRNAs could be associated with ARHL. However, the detailed regulatory mechanism needs to be further explored.
In this paper, we used a bioinformatics approach to identify mRNAs and lncRNAs involved in ARHL from public RNA-seq data generated in different environments with animals from different genetic backgrounds and different ages at the time of testing. Our study could provide a deeper understanding of the function of mRNAs and lncRNAs in ARHL development, as well as potential targets for preventing ARHL.
▪ We identified 112 common messenger RNAs (mRNAs) and 10 common long non-coding RNAs (lncRNAs) from aged cochlear samples in three different RNA-sequencing datasets.
▪ Age-related hearing loss (ARHL)-associated mRNAs were highly enriched in biological processes such as the immune system and defense response.
▪ The lncRNAs Gm11131, C4a, and Gm16094 were the ARHL-related lncRNAs identified with the highest frequency from aged cochlear samples.
ACKNOWLEDGMENTS
This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant no. HR20C0021) and by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (grant no. 2021R1C1C1013710).
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via https://doi.org/10.21053/ceo.2022.01235.
Supplementary Table 1.
List of differentially expressed messenger RNAs (mRNAs) in each dataset. The log2-fold change values and P-values between young and aged samples were obtained using the salmon-DESeq2 pipeline.
Supplementary Table 2.
List of differentially expressed long non-coding RNAs (lncRNAs) in each dataset. The log2-fold change values and P-values between young and aged samples were obtained using the salmon-DESeq2 pipeline.
Supplementary Table 3.
List of primer sequences used for real-time quantitative reverse-transcription polymerase chain reaction.
Supplementary Table 4.
List of the Gene Ontology analysis with upregulated and downregulated messenger RNA (mRNA).
Supplementary Table 5.
List of the trans-regulatory genes of differentially expressed long non-coding RNAs (lncRNAs). Eleven lncRNAs and their trans-regulatory target messenger RNAs (mRNAs) were identified.
REFERENCES
2. Keithley EM. Pathology and mechanisms of cochlear aging. J Neurosci Res. 2020; Sep. 98(9):1674–84.

3. Di Stazio M, Morgan A, Brumat M, Bassani S, Dell’Orco D, Marino V, et al. New age-related hearing loss candidate genes in humans: an ongoing challenge. Gene. 2020; Jun. 742:144561.

4. Ames BN. Mitochondrial decay, a major cause of aging, can be delayed. J Alzheimers Dis. 2004; Apr. 6(2):117–21.

5. Steyger PS, Cunningham LL, Esquivel CR, Watts KL, Zuo J. Editorial: cellular mechanisms of ototoxicity. Front Cell Neurosci. 2018; Mar. 12:75.

6. Menardo J, Tang Y, Ladrech S, Lenoir M, Casas F, Michel C, et al. Oxidative stress, inflammation, and autophagic stress as the key mechanisms of premature age-related hearing loss in SAMP8 mouse Cochlea. Antioxid Redox Signal. 2012; Feb. 16(3):263–74.

7. Loughrey DG, Kelly ME, Kelley GA, Brennan S, Lawlor BA. Association of age-related hearing loss with cognitive function, cognitive impairment, and dementia: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg. 2018; Feb. 144(2):115–26.

9. Bhat SA, Ahmad SM, Mumtaz PT, Malik AA, Dar MA, Urwat U, et al. Long non-coding RNAs: mechanism of action and functional utility. Noncoding RNA Res. 2016; Nov. 1(1):43–50.

10. Sun Q, Song YJ, Prasanth KV. One locus with two roles: microRNA-independent functions of microRNA-host-gene locus-encoded long noncoding RNAs. Wiley Interdiscip Rev RNA. 2021; May. 12(3):e1625.

11. Li Q, Zang Y, Sun Z, Zhang W, Liu H. Long noncoding RNA Gm44593 attenuates oxidative stress from age-related hearing loss by regulating miR-29b/WNK1. Bioengineered. 2022; Jan. 13(1):573–82.

12. Su Z, Xiong H, Pang J, Lin H, Lai L, Zhang H, et al. LncRNA AW112010 promotes mitochondrial biogenesis and hair cell survival: implications for age-related hearing loss. Oxid Med Cell Longev. 2019; Oct. 2019:6150148.

13. Hao S, Wang L, Zhao K, Zhu X, Ye F. Rs1894720 polymorphism in MIAT increased susceptibility to age-related hearing loss by modulating the activation of miR-29b/SIRT1/PGC-1α signaling. J Cell Biochem. 2019; Apr. 120(4):4975–86.
14. Xie W, Shu T, Peng H, Liu J, Li C, Wang M, et al. LncRNA H19 inhibits oxidative stress injury of cochlear hair cells by regulating miR-653-5p/SIRT1 axis. Acta Biochim Biophys Sin (Shanghai). 2022; Mar. 54(3):332–9.

15. Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 2014; Aug. 30(15):2114–20.

16. Harrow J, Frankish A, Gonzalez JM, Tapanari E, Diekhans M, Kokocinski F, et al. GENCODE: the reference human genome annotation for The ENCODE Project. Genome Res. 2012; Sep. 22(9):1760–74.

17. Patro R, Duggal G, Love MI, Irizarry RA, Kingsford C. Salmon provides fast and bias-aware quantification of transcript expression. Nat Methods. 2017; Apr. 14(4):417–9.

18. Soneson C, Love MI, Robinson MD. Differential analyses for RNA-seq: transcript-level estimates improve gene-level inferences. F1000Res. 2015; Dec. 4:1521.

19. Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014; Dec. 15(12):550.

20. Gu Z, Eils R, Schlesner M. Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics. 2016; Sep. 32(18):2847–9.

21. Su Z, Xiong H, Liu Y, Pang J, Lin H, Zhang W, et al. Transcriptomic analysis highlights cochlear inflammation associated with age-related hearing loss in C57BL/6 mice using next generation sequencing. PeerJ. 2020; Aug. 8:e9737.

22. Chen P, Hao JJ, Li MW, Bai J, Guo YT, Liu Z, et al. Integrative functional transcriptomic analyses implicate shared molecular circuits in sensorineural hearing loss. Front Cell Neurosci. 2022; Mar. 16:857344.

23. Lin H, Xiong H, Su Z, Pang J, Lai L, Zhang H, et al. Inhibition of DRP-1-dependent mitophagy promotes cochlea hair cell senescence and exacerbates age-related hearing loss. Front Cell Neurosci. 2019; Dec. 13:550.

24. Qi F, Zhang R, Chen J, Zhao F, Sun Y, Du Z, et al. Down-regulation of Cav1.3 in auditory pathway promotes age-related hearing loss by enhancing calcium-mediated oxidative stress in male mice. Aging (Albany NY). 2019; Aug. 11(16):6490–502.

25. Park C, Thein P, Kalinec G, Kalinec F. HEI-OC1 cells as a model for investigating prestin function. Hear Res. 2016; May. 335:9–17.

26. Kalinec GM, Park C, Thein P, Kalinec F. Working with auditory HEIOC1 cells. J Vis Exp. 2016; Sep. (115):54425.

27. Zhang Q, Liu H, McGee J, Walsh EJ, Soukup GA, He DZ. Identifying microRNAs involved in degeneration of the organ of corti during age-related hearing loss. PLoS One. 2013; Apr. 8(4):e62786.

28. Orrenius S. Reactive oxygen species in mitochondria-mediated cell death. Drug Metab Rev. 2007; 39(2-3):443–55.

29. Yu J, Wang Y, Liu P, Li Q, Sun Y, Kong W. Mitochondrial DNA common deletion increases susceptibility to noise-induced hearing loss in a mimetic aging rat model. Biochem Biophys Res Commun. 2014; Oct. 453(3):515–20.

30. Zimta AA, Tigu AB, Braicu C, Stefan C, Ionescu C, Berindan-Neagoe I. An emerging class of long non-coding rna with oncogenic role arises from the snorna host genes. Front Oncol. 2020; Apr. 10:389.

31. Ge BH, Li GC. Long non-coding RNA SNHG17 promotes proliferation, migration and invasion of glioma cells by regulating the miR23b-3p/ZHX1 axis. J Gene Med. 2020; Nov. 22(11):e3247.

32. Ji YY, Meng M, Miao Y. lncRNA SNHG1 promotes progression of cervical cancer through miR-195/NEK2 axis. Cancer Manag Res. 2020; Nov. 12:11423–33.
33. Chen Y, Sheng HG, Deng FM, Cai LL. Downregulation of the long noncoding RNA SNHG1 inhibits tumor cell migration and invasion by sponging miR-195 through targeting Cdc42 in oesophageal cancer. Kaohsiung J Med Sci. 2021; Mar. 37(3):181–91.
34. Tong J, Ma X, Yu H, Yang J. SNHG15: a promising cancer-related long noncoding RNA. Cancer Manag Res. 2019; Jul. 11:5961–9.
Fig. 1.
Identification of differentially expressed messenger RNA (mRNA) profiles in age-related hearing loss. A Venn diagram (A) and heatmap (B) showing the number of significantly differentially expressed mRNA profiles in the cochleae of aged mice compared to young mice. Red indicates upregulation and blue represents downregulation. Significantly enriched Gene Ontology terms of upregulated (C) and downregulated (D) mRNAs. MHC, major histocompatibility complex.
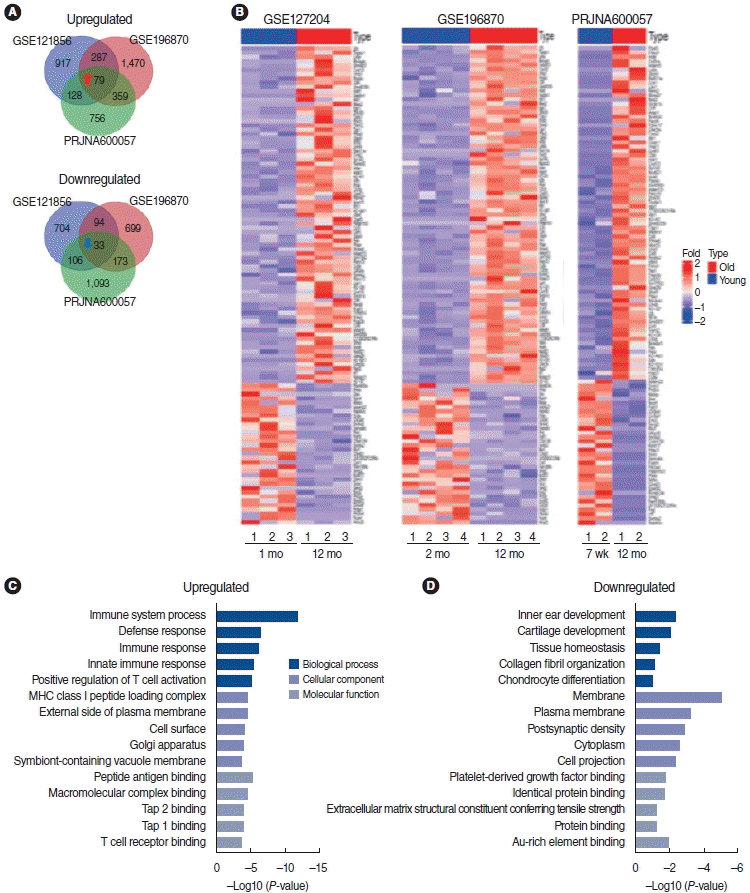
Fig. 2.
Bar graphs representing the upregulated differentially expressed messenger RNAs (mRNAs) (A) and downregulated differentially expressed mRNAs (B) in the cochleae of aged mice. (C) Validation of five differentially expressed mRNAs in H2O2-treated House Ear Institute-Organ of Corti 1 (HEI-OC1) cells using quantitative polymerase chain reaction. Statistical significance is indicated by asterisks (*P<0.05, **P<0.01, ***P<0.001).
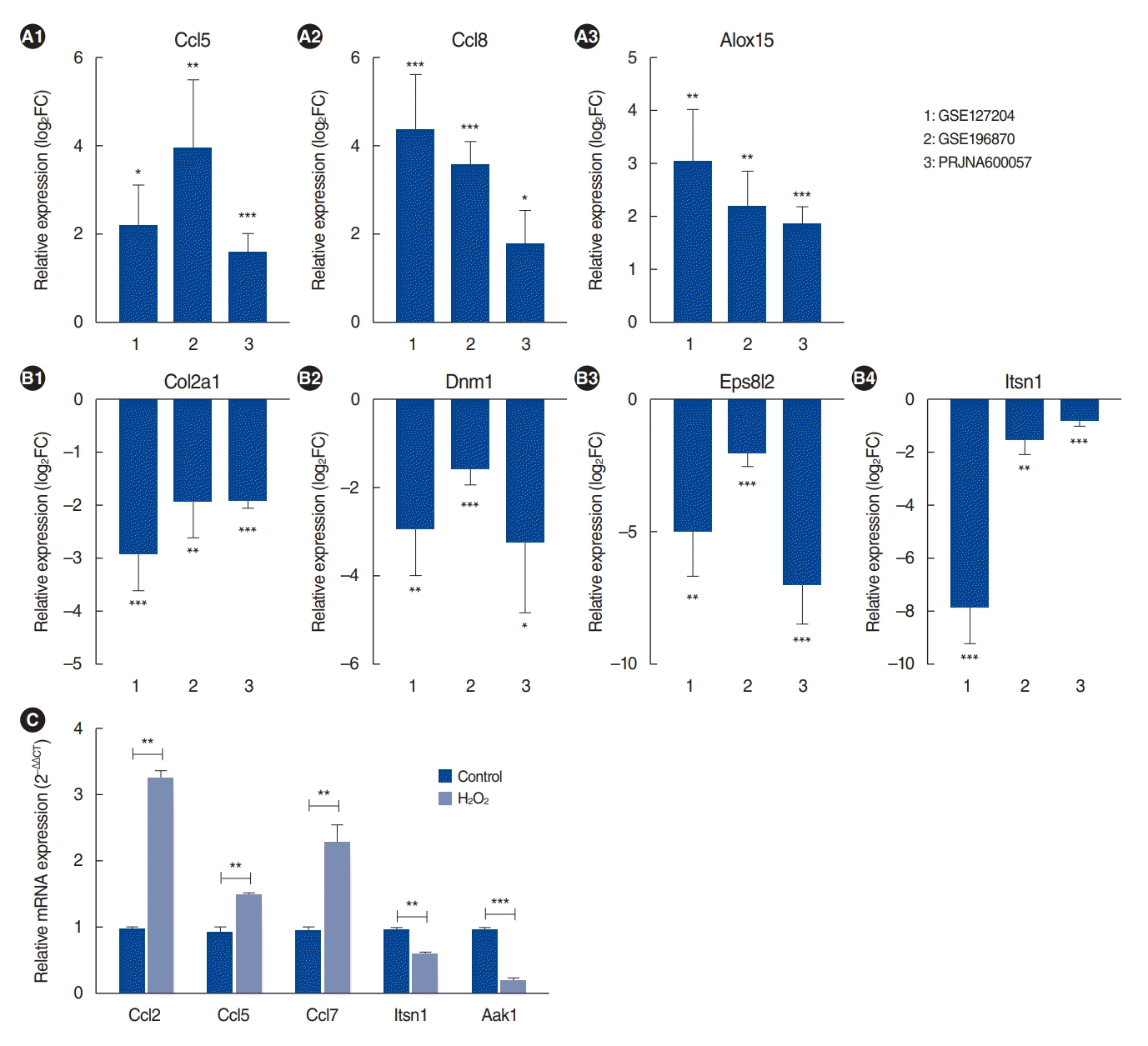
Fig. 3.
Identification of differentially expressed long non-coding RNA (lncRNA) profiles in age-related hearing loss. A Venn diagram (A) and heatmap (B) showing the number of the differentially expressed lncRNA profiles in the cochleae of aged mice compared to young mice. Red indicates upregulation and blue represents downregulation. (C) The identified lncRNAs in their genomic context. Genomic information was visualized with the Gviz package.
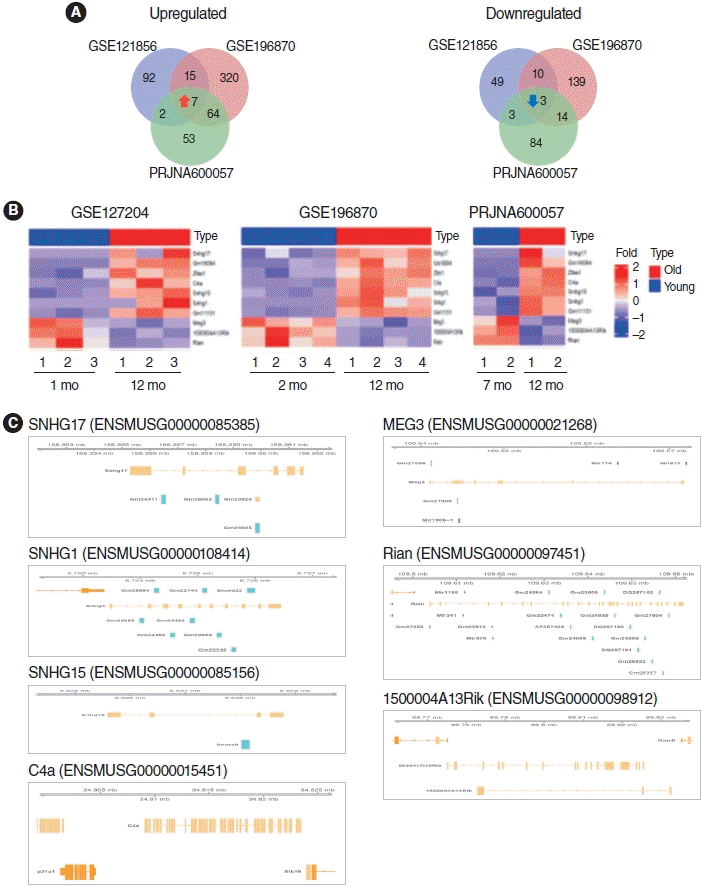
Fig. 4.
Bar graphs representing the upregulated differentially expressed long non-coding RNAs (lncRNAs) (A) and downregulated differentially expressed lncRNAs (B) in the cochleae of aged mice. (C) Validation of 10 differentially expressed lncRNAs in H2O2-treated House Ear InstituteOrgan of Corti 1 (HEI-OC1) cells using quantitative polymerase chain reaction. Statistical significance is indicated by asterisks (*P<0.05, **P<0.01, ***P<0.001). NS, not significant.
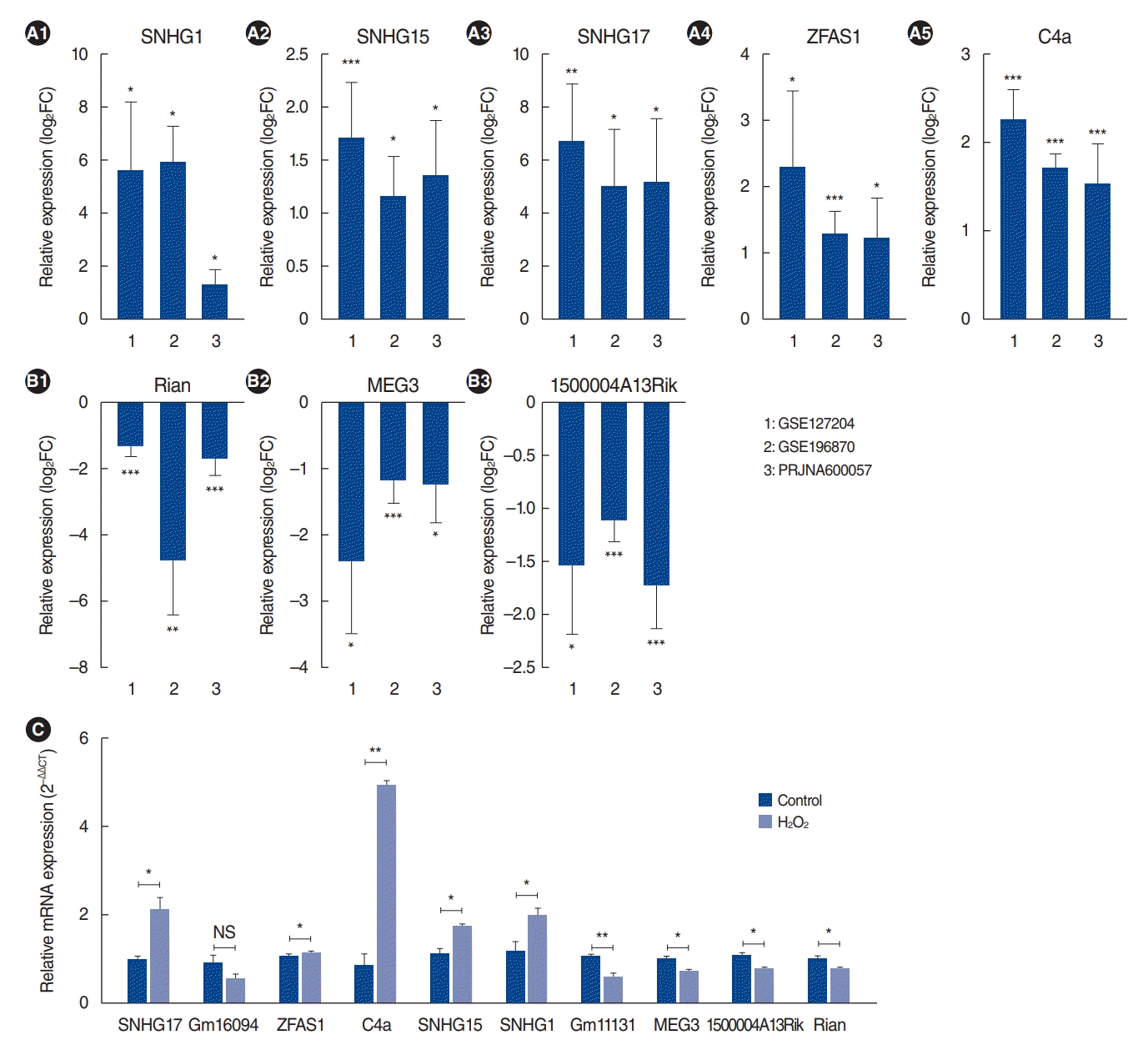
Fig. 5.
Construction of the long non-coding RNA (lncRNA)-messenger RNA (mRNA) network for age-related hearing loss, consisting of upregulated lncRNAs and downregulated lncRNAs with their trans-target differentially expressed mRNA. The diamonds and circles represent lncRNA and mRNAs. Red nodes represent upregulated lncRNAs or mRNAs, and blue nodes represent downregulated lncRNAs or mRNAs.
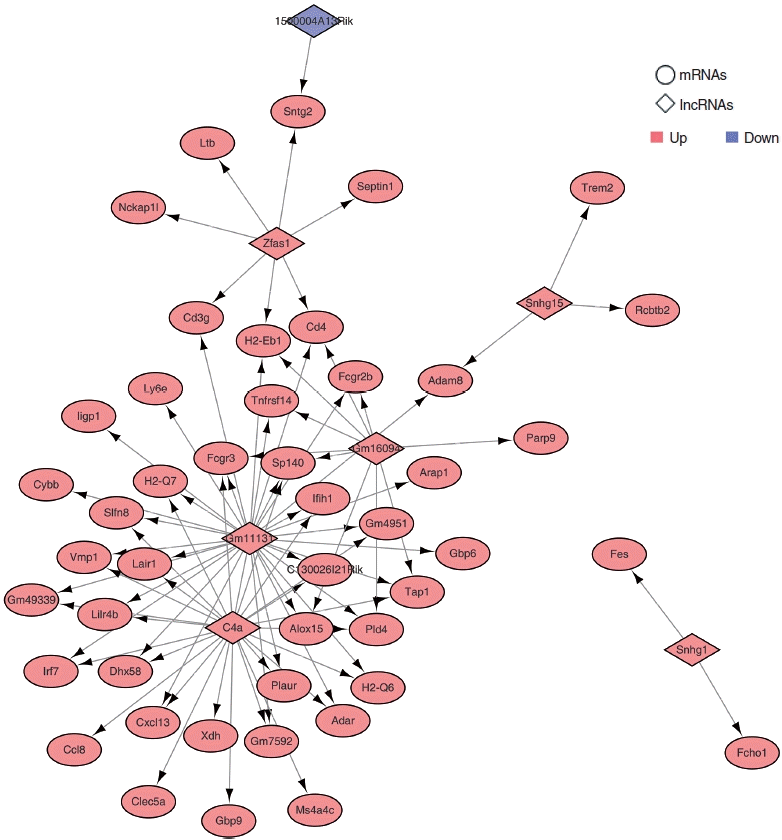
Table 1.
Selected data sets from NCBI GEO database
| Accession number | Platform | Strain | Tissue | Condition |
Dataset |
Reference | |
|---|---|---|---|---|---|---|---|
| Young | Old | ||||||
| GSE127204 | RNAseq | C57BL/6 | Cochlea | 1 mo–12 mo | GSM3630279 GSM3630280 | GSM3630282 GSM3630283 | PMID: 32879802[21] |
| GSM3630281 | GSM3630284 | ||||||
| GSE196870 | RNAseq | C57BL/6 | Cochlea | 2 mo–12 mo | GSM5903362 GSM5903363 | GSM5903370 GSM5903371 | PMID: 35370561[22] |
| GSM5903364 GSM5903365 | GSM5903372 GSM5903373 | ||||||
| PRJNA600057 | RNAseq | C57BL/6 | Cochlea | 7 wk–12 mo | SRR10882835 SRR10882836 | SRR10882833 SRR10882834 | |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download