Introduction
In benign cases that need surgery, if not bilateral disease, lobectomy is the basic principle of surgery. Even after lobectomy in low-risk differentiated thyroid cancer, local recurrence and survival rates have been reported to be lower than those of total thyroidectomy; hence, the operation indication for lobectomy was expanded in the 2015 American Thyroid Association (ATA) guidelines. Consequently, the incidence of thyroid lobectomy has been gradually increasing.
1) Lobectomy has residual thyroid tissue that secretes thyroid hormone after surgery. Therefore, normal thyroid function can likely be maintained without taking thyroid hormone medication, and many patients need not to take the medication. However, hypothyroidism after lobectomy is not uncommon; although its incidence varies depending on the definition criteria for hypothyroidism, the judgment period, and the follow-up period, the incidence has been reported to be up to 5.6-46%.
2-4) Therefore, even after lobectomy, approximately 20-47% of patients need to take thyroid hormone medications.
2-4)
Until now, studies on hypothyroidism after lobectomy have focused on the frequency, risk factors, and clinical features of hypothyroidism. The risk factors for hypothyroidism after lobectomy are preoperative high thyroid-stimulating hormone (TSH) levels, positive antithyroid peroxidase (anti-TPO) antibody, and lymphocytic infiltration in the thyroid gland.
5-7) These are patient-related factors that cannot be managed. Moreover, even in patients without these risk factors, postoperative hypothyroidism is not uncommon. To date, no prospective studies have explored the extent of thyroid lobectomy, that is, the effect of isthmusectomy on hypothyroidism. Although conventional lobectomy is an operation performed to remove a lobe, including the isthmus (unilateral lobectomy plus isthmusectomy is performed to remove the isthmus together), the essential rationale and oncological safety for isthmusectomy are not clear. We hypothesized that, for patients without preoperative hypothyroidism, the greater the amount of thyroid tissue left after surgery, the better normal thyroid function would be maintained. This study aimed to determine whether saving the isthmus to maintain thyroid function helps to reduce hypothyroidism after lobectomy.
Go to :

Materials and Methods
From January 2016 to June 2018, all patients who met the indications and exclusion criteria were considered subjects. In total, 144 consecutive patients who underwent lobectomy with the conventional open method by one surgeon (YS Jeon) from one institution were retrospectively enrolled, and their data were analyzed. The criteria for selection of the target patients were patients with normal preoperative TSH levels (0.27-4.2 μIU/mL) and those with benign thyroid nodules, follicular neoplasm, or low-risk papillary thyroid cancer (PTC) that did not require suppression of TSH. Low-risk PTC was defined as a tumor size of <2 cm, intrathyroidal lesions, no or less than five micrometastases, and no distant or lateral lymph node metastasis. Patients who met the following conditions were excluded from the study: (1) hyperthyroidism or hypothyroidism (TSH <0.2 or >4.2 μIU/mL), (2) a history of taking thyroid hormone medications or antithyroid drugs, (3) a history of cervical radiation therapy or thyroid surgery, (4) planned subtotal or total thyroidectomy, (5) endoscopic/robotic thyroidectomy, (6) need for completion of thyroidectomy due to postoperative high-risk surgical pathology, (7) short postoperative follow-up (<1 year), (8) agenesis of the isthmus (patients born without the isthmus), and (9) presence of malignancies in the isthmus or difficulty in securing the margin due to close proximity to the isthmus.
The isthmus was defined as the thyroid anterior to the trachea and bounded by both lateral margins of the trachea, and isthmus-saving or isthmus-removing lobectomy was performed to preserve or remove the isthmus along the tracheal margin (
Fig. 1). For resection along the correct isthmus margin, the strap muscle was first dissected from the thyroid gland. After exposing the isthmus, it was marked with a marking pen along the tracheal margin. The superior, lateral, and inferior parts of the thyroid were resected. Then, after cutting Berry’s ligament, the isthmus was finally resected or saved along the marked line. No PTC patients had suspicious lymph node findings in the central neck area on preoperative ultrasound or CT, and prophylactic central lymph node dissection was performed in all cases.
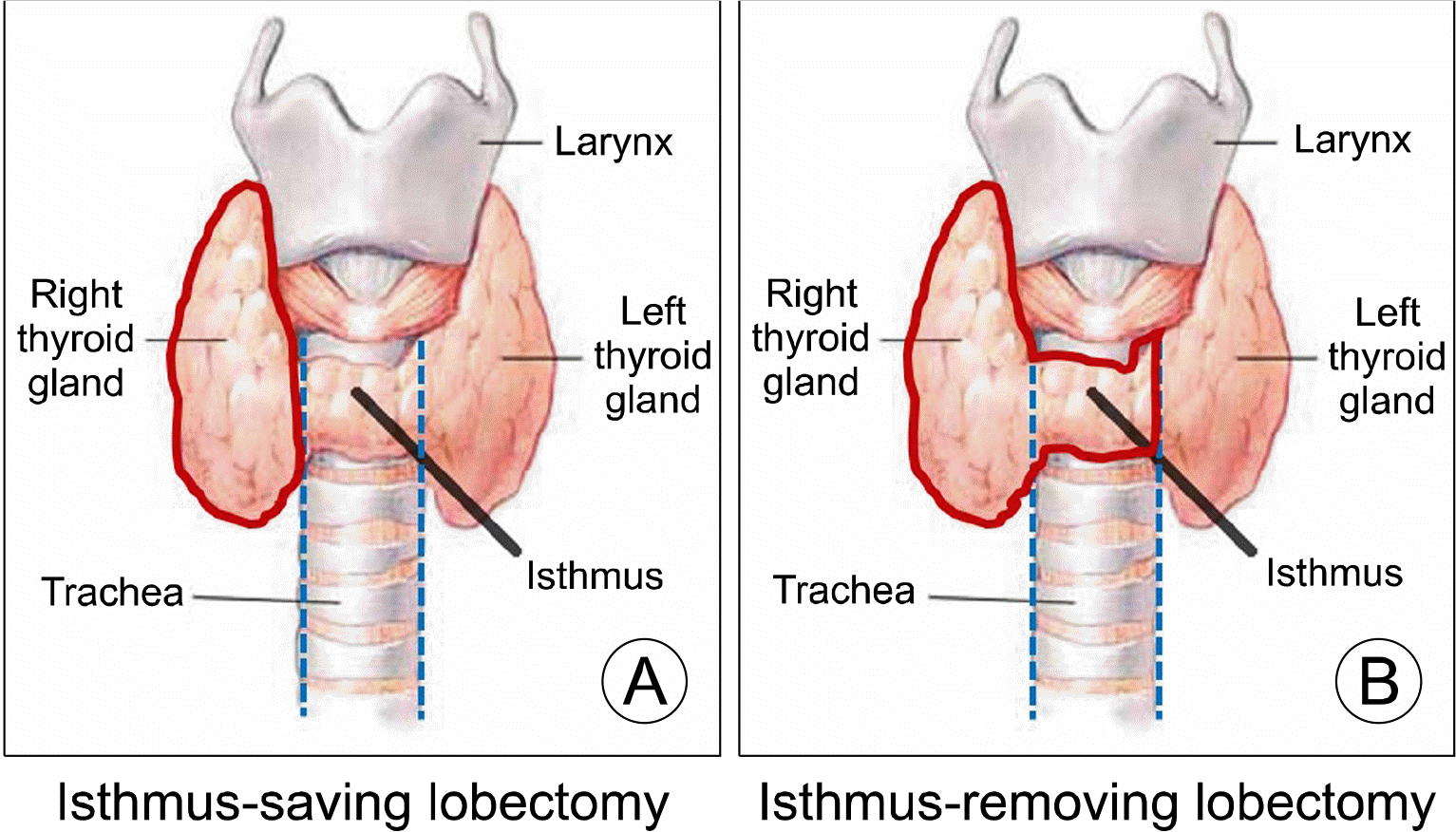 | Fig. 1Isthmus-saving right lobectomy and isthmus-removing right lobectomy. 
|
Patients were classified into an experimental group, an isthmus-saving lobectomy group (IS group), in which the isthmus was preserved, and a control group, referred to as the isthmus-removing lobectomy group (IR group), in which the isthmus was removed together. Patients were not randomly assigned but divided into two groups according to the presence or absence of isthmus lesions. If there was a benign tumor in the isthmus of the lobe to be operated on or if the cancer was close to the isthmus, isthmus-removing lobectomy was performed. If not, isthmus- saving lobectomy was performed. When patients have a pyramidal lobe, it may be difficult to identify for various reasons, such as adhesion during the next operation; hence, it was removed from both groups. Thyroid function tests were performed at 3, 6, 12, and 24 months after surgery. The results were then compared between the IS group and the IR group. Further-more, postoperative levothyroxine medications for hypothyroidism, other postoperative complications, and clinicopathological data were compared.
As the TSH test provides the most important value in this study, it was performed as a radioimmunoassay to improve its accuracy. Commercial immunoradiometric assay kits were used for TSH measurement (TSH IRMA, BRAHMS, Hennigsdorf, Germany). The normal range for TSH in our institution is 0.27-4.2 μIU/mL. Overt hypothyroidism was defined biochemically as a decrease in free thyroxine (T4) concentration and a TSH level >10 μIU/mL. Subclinical hypothyroidism was defined as a case in which the free T4 concentration was normal and the TSH concentration slightly elevated to <10 μIU/mL. No thyroid hormone medication (levothyroxine) was initially administered in any patients. During the follow-up period, levothyroxine was administered to patients with overt hypothyroidism, those with TSH levels >10 μIU/mL, and those with subclinical hypothyroidism who complained of obvious symptoms of hypothyroidism (fatigue, edema, and weight gain) (
Fig. 2). If a patient is diagnosed with asymptomatic subclinical hypothyroidism, it may be temporary hypothyroidism. Therefore, the thyroid function test was performed again after 2 months to evaluate whether it progressed to overt hypothyroidism without administering levothyroxine. Two months after the administration of thyroid hormone medications, a thyroid function test was performed to adjust the drug dose if necessary. Patients with normal thyroid function who desired to take levothyroxine were instructed to take levothyroxine and were excluded from the study to reduce bias. Furthermore, if the postoperative histopathological results were not diagnosed as low risk, the patients were asked to take thyroid hormone again for TSH suppression and were excluded from the study. None of the patients wanted to take thyroid hormone medication after the surgery, and two patients who took levothyroxine for TSH suppression without completion of thyroidectomy were excluded from this study because postoperative pathology revealed that they were intermediate-risk patients. This study was approved by the Institutional Review Board (IRB No. 2020-09- 022). The t test or the Mann‒Whitney U test was used to compare continuous variables. The chi-square test, Fisher’s exact test or Pearson’s correlation analysis was used to compare categorical variables. The analysis was conducted using SPSS Version 20.0, and statistical significance was accepted for p values of <0.05.
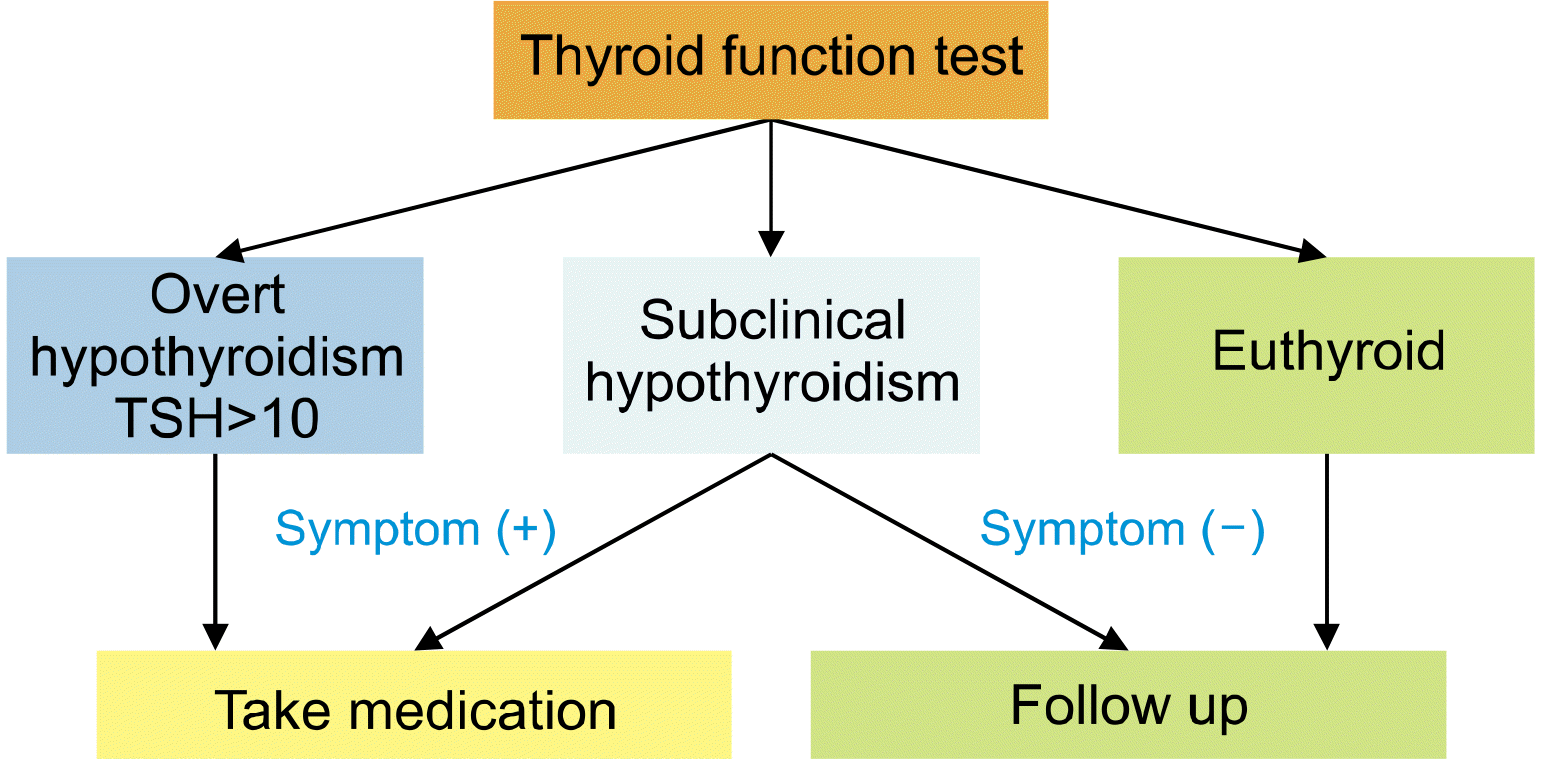 | Fig. 2As a follow-up flow of the patient, medication was administered if overt hypothyroidism or subclinical hypothyroidism with obvious symptoms occurred. 
|
Go to :

Results
Among the 144 patients, 100 were in the IS group, and 44 were in the IR group (
Table 1). No differences were observed in sex, age, or body mass index between the two groups. In total, 30 patients (20.8%) showed autoAb (anti-Tg Ab, anti-TPO Ab)-positive results before the surgery, and there was no difference between the two groups. The mean preoperative TSH level of all patients was 1.91±1.0 μIU/mL. A total of 87 (60.4%), 35 (24.3%), and 22 (15.3%) patients had preoperative TSH levels of <2, 2-3, and 3-4.2 μIU/mL, respectively. At 3, 6, 12, and 24 months after surgery, the mean TSH levels were 3.9, 4.1, 3.2, and 3.0 μIU/mL, respectively. While there was no difference in preoperative TSH levels between the two groups, the TSH level was significantly lower in the IS group at 3 and 6 months after the surgery (p=0.049 and 0.026, respectively). Overall, 122 patients (84.7%) were diagnosed with PTC by fine-needle aspiration before surgery and then underwent surgery. Furthermore, 22 patients (15.2%) were diagnosed with benign nodules or follicular neoplasms. Among those 22 patients, 2 were diagnosed with follicular thyroid cancer after surgery. All patients with PTC underwent prophylactic central lymph node dissection, and there was no difference in the number of retrieved lymph nodes or metastatic lymph nodes between the IS group and the IR group. In the postoperative pathological results, 23 patients (16.0%) had Hashimoto thyroiditis. Right lobectomy was performed in 74 patients, and left lobectomy was performed in 70 patients.
Table 1
Clinicopathological characteristics
|
IS group (n=100) |
IR group (n=44) |
p value |
|
Sex (F:M) |
72:28 |
37:7 |
0.119 |
|
Age (years) |
47.2±1.1 |
47.5±1.9 |
0.885 |
|
Body mass index (m2/kg) |
23.4±0.3 |
23.5±0.5 |
0.848 |
|
Positive of preoperative auto antibody (n) |
22 (22.0%) |
8 (18.2%) |
0.630 |
|
Preoperative TSH (μIU/mL) |
1.8±0.1 |
2.0±0.1 |
0.671 |
|
Postoperative TSH (μIU/mL) |
|
|
|
|
3 months |
3.6±0.2 |
4.5±0.5 |
0.049 |
|
6 months |
3.6±0.2 |
5.2±0.7 |
0.026 |
|
12 months |
3.1±0.2 |
3.7±0.4 |
0.419 |
|
24 months |
3.0±0.1 |
3.1±0.3 |
0.275 |
|
Tumor size (cm) |
1.0.±0.1 |
0.9±0.1 |
0.740 |
|
Diagnosis (n) |
|
|
0.243 |
|
Papillary cancer |
83 |
39 |
|
|
Benign |
15 |
5 |
|
|
Follicular cancer |
2 |
0 |
|
|
Retrieved lymph node (n) |
5.1±0.3 |
6.1±0.6 |
0.141 |
|
Metastatic lymph node (n) |
0.5±0.1 |
0.7±0.2 |
0.444 |
|
Thyroiditis (n) |
14 (14.0%) |
9 (20.5%) |
0.330 |

After the surgery, 11 (7.6%) and 40 (27.7%) patients were diagnosed with overt hypothyroidism and subclinical hypothyroidism, respectively. A total of 24 patients (16.7%) took thyroid hormones after the surgery, among whom 11 patients took medication for overt hypothyroidism or a TSH level of >10 μIU/mL, and 13 patients took medication for subclinical hypothyroidism with obvious symptoms. A comparison of the IS group and the IR group revealed the presence of 67 (67%) and 26 (59%) euthyroid patients in the thyroid function tests after the surgery, respectively. There were more patients with euthyroid status in the IS group, but the difference was not statistically significant (p=0.361) (
Fig. 3). However, postoperative overt hypothyroidism was found in 5 patients (5/100, 5.0%) in the IS group and 6 patients (6/44, 13.6%) in the IR group (p=0.04), and the numbers of patients who took medication after the surgery, 10 (10/100, 10%) and 14 (14/44, 31.8%), were significantly lower in the IS group (p=0.01) (
Table 2). In another previous study, preoperative TSH>2 was the strongest risk factor for hypothyroidism after lobectomy (odds ratio 5.517).
8) The ATA guidelines recommend that low-risk PTC patients maintain their TSH level at <2 μIU/mL after surgery. Therefore, a subgroup analysis was conducted on only the patients with preoperative TSH levels of <2 μIU/mL to determine how many patients maintained TSH<2 after surgery. There were 66 patients (75.9%) in the IS group and 21 patients (24.1%) in the IR group (
Fig. 4). However, 17 patients (17/66, 25.8%) in the IS group and 2 patients (2/21, 9.5%) in the IR group had their TSH levels maintained at <2 μIU/mL even after surgery, with the difference being statistically significant (p=0.04). When only 22 patients with a TSH level of 3-4.2 μIU/mL before the surgery were analyzed, we found 14 patients (63.6%) in the IS group and 8 patients (36.3%) in the IR group (
Fig. 5). Postoperative overt hypothyroidism was diagnosed in 1 patient (1/14, 7.1%) in the IS group and 2 patients (2/8, 25.0%) in the IR group. The number of patients who took medications after the surgery was also significantly lower in the IS group, with 3 (3/14, 21.4%) in the IS group and 5 (5/8, 62.5%) in the IR group (p=0.04 and 0.02, respectively). Even when only patients without thyroiditis were examined, there were significantly fewer cases of hypothyroidism and medications in the IS group than in the IR group. However, there was no difference between the two groups regarding patients with thyroiditis. No differences were observed between the two groups in terms of complications, such as recurrent laryngeal nerve palsy, hypoparathyroidism, and postoperative bleeding. The average follow-up period after the surgery was 32.5 months (range: 12-57 months).
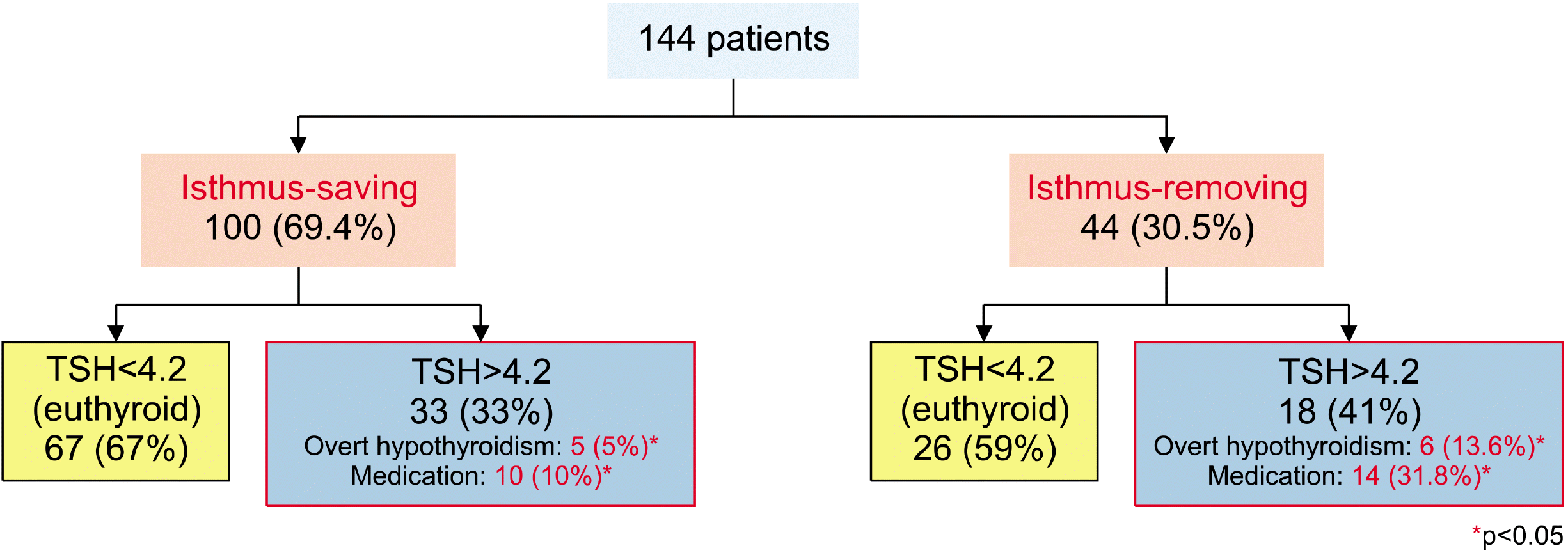 | Fig. 3As a result of all patients, the incidence of overt hypothyroidism and medi-cation was lower in the isthmus-saving lobectomy group. 
|
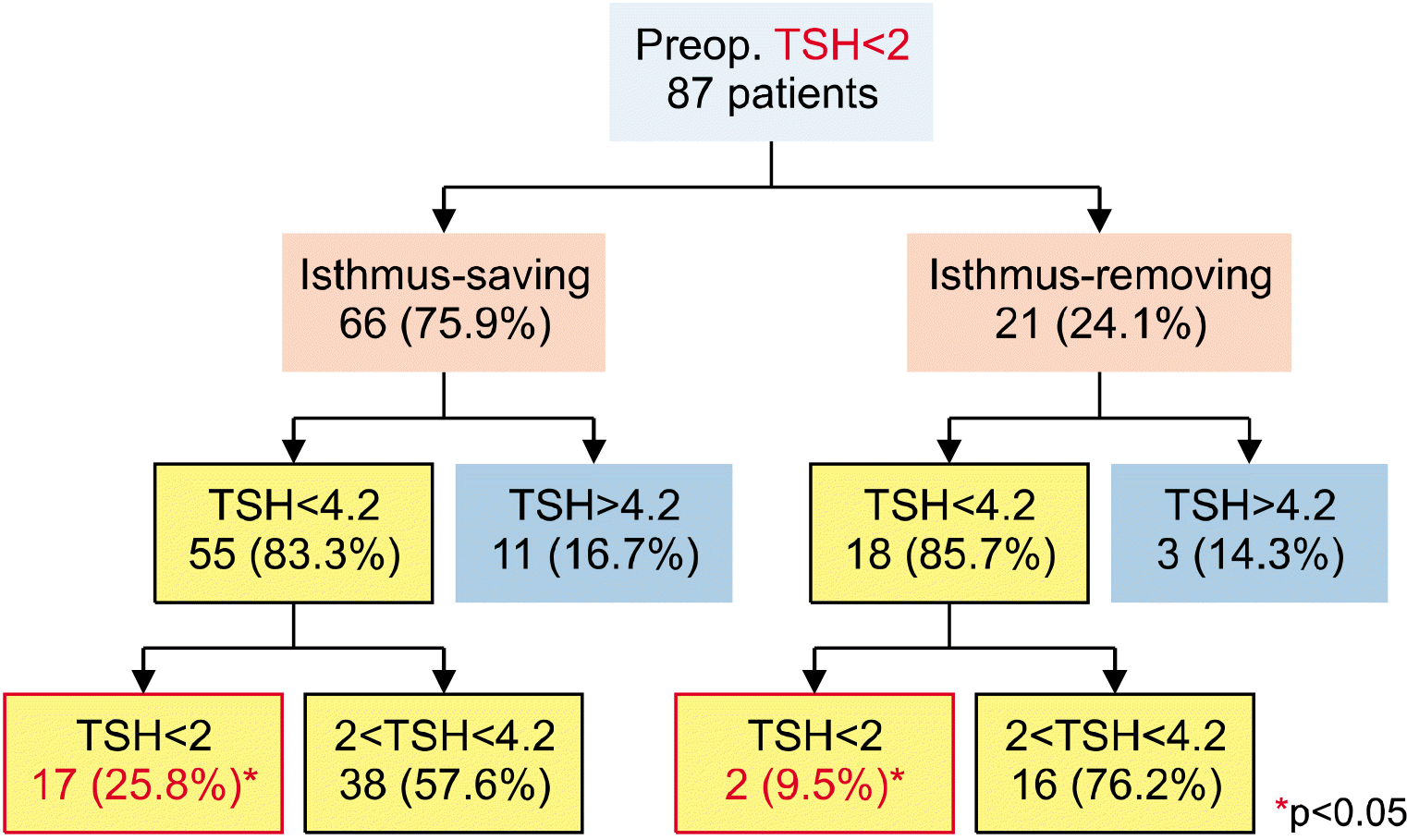 | Fig. 4As a result of patients with a preoperative TSH of <2, the number of patients with a TSH <2 was higher in the isthmus-saving lobectomy group postoperatively. 
|
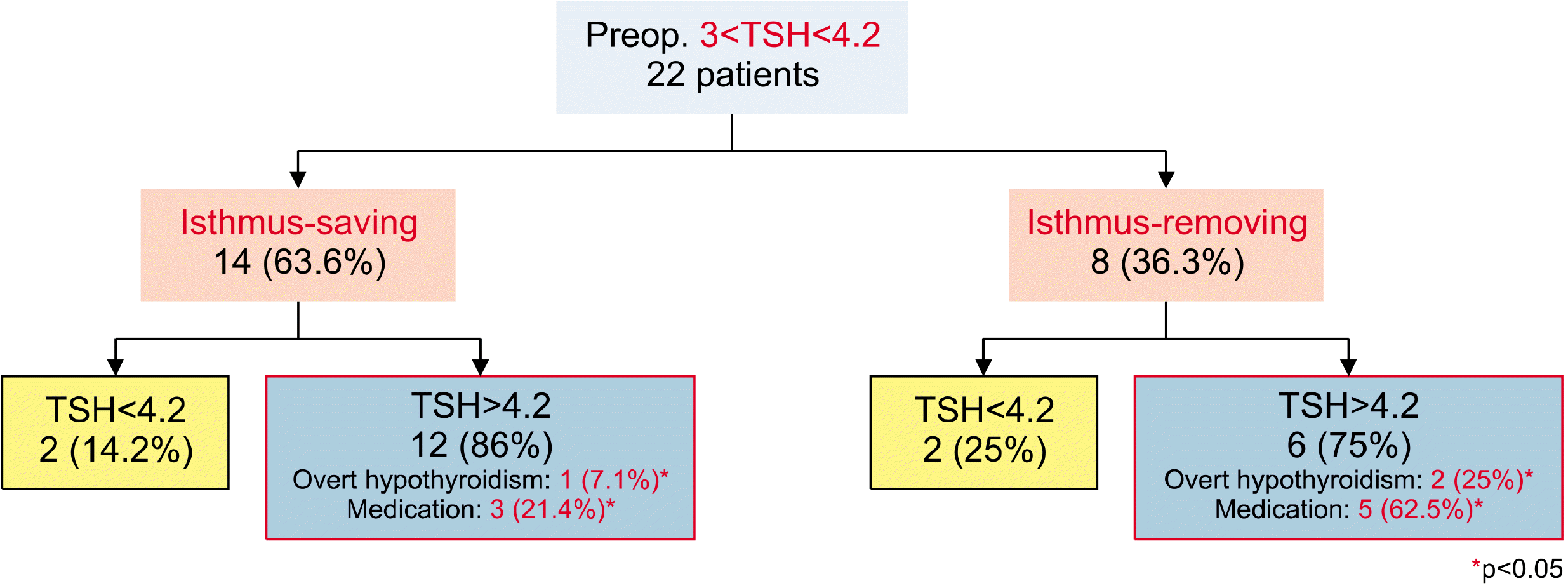 | Fig. 5As a result of patients with preoperative TSH of ≥3 and <4.2, the incidence of hypothyroidism and medication was less in the isthmus-saving lobectomy group. 
|
Table 2
Postoperative hypothyroidism and incidence of medication start between isthmus saving and isthmus removing group
|
IS group |
IR group |
p value |
|
Total patients (n=144) |
n=100 (69.4%) |
n=44 (30.5%) |
|
|
Euthyroid |
67 (67%) |
26 (59%) |
0.36 |
|
Overt hypothyroidism |
5 (5%) |
6 (13.6%) |
0.04 |
|
Medication start |
10 (10%) |
14 (31.8%) |
0.01 |
|
Preoperative TSH<2 patients (n=87) |
n=66 (75.9%) |
n=21 (24.1%) |
|
|
Euthyroid |
55 (83.3%) |
18 (85.7%) |
0.79 |
|
TSH<2 |
17 (25.8%) |
2 (9.5%) |
0.04 |
|
Medication start |
3 (4.5%) |
2 (9.5%) |
0.39 |
|
Preoperative 3<TSH<4.2 patients (n=22) |
n=14 (63.6%) |
n=8 (36.3%) |
|
|
Euthyroid |
2 (14.2%) |
2 (25%) |
0.53 |
|
Overt hypothyroidism |
1 (7.1%) |
2 (25%) |
0.04 |
|
Medication start |
3 (21.4%) |
5 (62.5%) |
0.02 |

Go to :

Discussion
The symptoms of hypothyroidism, such as chronic fatigue, loss of appetite, weight gain, feeling cold, constipation, dry skin, changes in menstrual cycle, or excessive menstruation, may debase quality of life. In our retrospective study, isthmus-saving lobectomy reduced the incidence of postoperative hypothyroidism and medication intake and helped to maintain a low TSH level postoperatively when compared with isthmus-removing lobectomy.
The advantage of total thyroidectomy is that it removes thyroid lobes; thus, the recurrence rate is low. Furthermore, recurrence can be detected early using a thyroglobulin test, and an additional advantage is that adjuvant radioactive iodine therapy can be admini-stered if necessary. Nevertheless, the risk of complications after surgery, such as hypoparathyroidism and recurrent laryngeal nerve injury, is more frequent than lobectomy, and all patients must take thyroid hormone medications for the rest of their life because of hypothyroidism. In contrast, the advantages of lobectomy are short operative time, rapid recovery, and fewer complications, such as hypoparathyroidism.
4)
Thus, the frequency of lobectomy, a more conservative treatment, has been increasing.
1) In the low-risk PTC group, there is no evidence of postoperative disease in 78-91% of cases, and prognosis after surgical treatment is known to be very good.
1) Earlier, after thyroid lobectomy, most patients received TSH suppression therapy to reduce recurrence. However, the benefit of TSH suppression in patients with low-risk thyroid cancer is controversial.
9,10) There is no clear evidence for TSH suppression and little evidence to guide TSH targets or the use of thyroid hormone in low-risk patients who have undergone lobectomy in the ATA guidelines.
1) In addition to the questionable effects, TSH suppression lowers bone density, especially in postmenopausal women, and the side effects may be quite serious. Furthermore, it has adverse effects on cardiovascular functions, such as arrhythmia, and particularly exerts a negative impact on the psychological well-being of the patient. Therefore, there is an increasing number of cases in the low-risk group without thyroid hormone treatment for TSH suppression after lobectomy.
10,11)
Although lobectomy has several advantages, a subset of patients undergoing thyroid lobectomy may develop hypothyroidism, and it may therefore not offer freedom from thyroid hormone replacement. Hypothy-roidism occurs at a significant frequency of 7-49% after lobectomy, with an overall risk of approximately 22%.
4,8,12) Such a wide range of incidence of hypothyroidism could be attributed to the different definitions of hypothyroidism, patient characteristics between studied populations, follow-up duration, the timing of thyroid hormone supplementation, and surgical techniques. Hypothyroidism is generally diagnosed within the first 6 months after surgery, but it may also be diagnosed after 6 months. The risk factors for hypothyroidism include high TSH levels before surgery, positive anti-TPO antibody, and Hashimoto thyroiditis. Factors with conflicting results include old age, antithyroglobulin antibody, weight of the resected thyroid tissue, and iodine deficiency.
4,8,13) If a patient has these risk factors, goiter with an enlarged thyroid volume due to hypothyroidism may occur. In this goiter case, since the function of secreting hormones from the thyroid tissue is lowered, the remaining thyroid volume may have a lower correlation with maintaining normal thyroid function. According to a study by Moon et al.,
14) as a predictive factor for hypothyroidism after hemithyroidectomy, a higher preoperative TSH level and lower the remnant thyroid volume correlated with a higher risk of hypothyroidism (p<0.001 and p=0.0014). We hypothesized that, in patients without hypothyroidism, greater amounts of thyroid tissue left after surgery would be associated with better maintenance of normal thyroid function.
To date, the basic technique for lobectomy has been to remove the isthmus. However, the rationale for isthmus-removing lobectomy is unclear and controversial. For example, during complete thyroidectomy after isthmus-saving lobectomy, surgery could be difficult due to adhesion in the isthmus region, and pretracheal lymph node dissection could also be difficult. In addition, saving the isthmus may be aesthetically unpleasing in a thin patient, especially if the isthmus is thick, and the patient might feel a lump of tissue over the trachea. However, in this study, there was no difficulty in performing pretracheal lymph node dissection during isthmus-saving lobectomy, and there was no difference in the number of retrieved lymph nodes between the IS group and the IR group. Therefore, there were no major problems in central lymph node dissection during isthmus-saving lobectomy. Moreover, even when complete thyroidectomy was performed after isthmus-saving lobectomy, the operation was not difficult due to severe adhesion in the trachea and could be safely performed. Since contralateral tracheoesophageal grooves or recurrent laryngeal nerves were not exposed during the isthmus-saving or isthmus-removing lobectomy, there was no risk of contralateral nerve palsy or parathyroid gland injury during the operation. In tumors confined to the isthmus, there were no complications, such as recurrent laryngeal nerve palsy, hypocalcemia, or hematoma, after isthmusectomy.
15) In only one of 144 patients, the isthmus grew contralaterally over the trachea. It should be noted that this can happen in rare cases if too much isthmus is left.
To date, there are no prospective studies on the effect of the extent of lobectomy depending on whether an isthmectomy is performed for hypothyroidism. According to the results of this study, euthyroidism was observed in 67% of patients in the IS group and 59% of patients in the IR group, with no significant difference. However, the proportion of patients taking levothyroxine for overt and subclinical hypothyroidism with definite symptoms was significantly higher in the IR group. Even in a specific patient group whose preoperative TSH levels were between 3 and 4 μIU/mL, taking medication was significantly reduced in the IS group. A conventional lobectomy is an operation to remove one lobe, including the isthmus. However, isthmus-saving lobectomy can be considered first because the evidence for isthmusectomy is not clear, there were no major technical problems during isthmus-saving lobectomy or completion thyroidectomy, and it helped to maintain the patient’s thyroid function by preserving the isthmus. Moreover, in isthmus-saving lobectomy, if there is no tumor in the isthmus and the free resection margin of the cancer can be secured, it is believed that there is no negative effect on oncologic results. The benefits of maintaining thyroid function after surgery may include a reduction in symptoms due to hypothyroidism, the frequency of taking medications, and the frequency of unnecessary thyroid function tests, which will help improve the quality of life of patients after surgery.
This paper is the first study to show that isthmus-saving lobectomy helps to maintain euthyroid function and keep TSH levels low postoperatively. Nevertheless, this study has some limitations. (1) It was not a randomized controlled trial. (2) The number of enrolled patients was small, and power analysis was not performed to determine the sample size needed. (3) In the case of subclinical hypothyroidism, the criteria for medication depended on the patient’s subjective symptoms. Thus, there may have been bias, and further validation is required by performing a prospective multicenter randomized controlled trial on a large number of patients; such a study is currently in progress. Moreover, rather than administering the drug based on the patient’s subjective symptoms, it would be better to decide whether to start taking the medication based on an objective indicator, such as the hypothyroidism index. Furthermore, in future research, we intend to measure the volume of the isthmus and remnant thyroid and verify whether the quality of life after surgery is better by comparing the group of patients who maintain normal thyroid function without taking thyroid hormone medications and the group of patients taking medications for hypothyroidism. In a prospective multicenter randomized controlled trial, if isthmus-saving lobectomy can significantly reduce the frequencies of postoperative hypothyroidism and medication intake, isthmus-saving lobectomy could be considered first in low-risk patients without lesions in the isthmus and in patients with benign thyroid nodules.
In conclusion, isthmus-saving lobectomy reduced the incidence of postoperative hypothyroidism and medication intake and helped to maintain a low TSH level when compared with isthmus-removing lobectomy.
Go to :








 PDF
PDF Citation
Citation Print
Print





 XML Download
XML Download