Abstract
Recently, the survival of the high-risk population of preterm infants has steadily improved, and the severity of prematurity is a growing threat of gestational-age-related fatal conditions. Posthemorrhagic hydrocephalus (PHH) is the most common but serious neurological complication in premature infants, which can have life-threatening consequences during the acute phase in the neonatal period and life-long psychomotor and cognitive sequelae in their later life. Although neonatologists, pediatric neurologists, and pediatric neurosurgeons have investigated a diversified strategy for several decades, a consensus on the best management of PHH in premature infants still must be reached. Several approaches have tried to reduce the incidence of intraventricular hemorrhage (IVH) and mitigate the effect of IVH-related hydrocephalus. This paper reviews and discusses the clinical feature of PHH in premature infants, general/nonsurgical management of prematurity for IVH prevention, and posthemorrhagic management, and how and when to intervene.
In neonatal brain hemorrhage, a subdural hematoma was previously the most common type of intracranial hemorrhage, probably as a result of adverse obstetric conditions [22]. In the last 10 years, the incidence of preterm birth of children with germinal matrix (GM)-intraventricular-intraparenchymal hemorrhage (IPH) has increased, which was likely due to the improved survival of extremely preterm children [11]. Currently, GM hemorrhage (GMH) is the most frequent and important brain abnormality in the neonatal period because of the increased survival of premature infants with very low birth weight (VLBW <1500 g), who received advanced neonatal intensive care [22]. Approximately 15–20% of preterm infants weighing <1500 g at birth develop GMH [18], which is an independent predictor of worse neonatal outcomes [38]. In preterm infants, the mortality rate with periventricular hemorrhagic infarction ranges between 38% and 60% [64]. Nevertheless, the survival rate increases with sophisticated neonatal care. However, the rates of neurodevelopmental impairment and cerebral palsy are also increasing, becoming detached from the ideal goal of intact survival. Survived preterm birth is deeply associated with not only motor disorders but also intellectual disabilities, communication disorders, attention-deficit/hyper-reactivity disorder, autism spectrum disorders, and so forth [36]. At present, GMH in neonates presents as a great medical, social, and financial challenge for each country.
A correlation has been established between low gestational age (GA) at birth and the incidence and severity of hemorrhage [23]. Clinically, intraventricular hemorrhage (IVH) occurred in 16% of all very preterm infants [72]. At 22 weeks postmenstrual age (PMA), 30% of preterm infants had IVH; conversely, at 28 weeks PMA, only 3% had IVH [72]. IVH may emerge as from being clinically silent to catastrophic, depending on the degree of hemorrhage volume and site. On computed tomography, Papile et al. [57] classified subependymal hemorrhage and IVH in premature infants with BW <1500 g (Table 1). A four-grade classification of GM-IVH based on hemorrhage location and severity was developed : grade 1 is defined by hemorrhage that is confined to the GM; grade 2, the hemorrhage extends into lateral ventricles without ventricular dilatation; grade 3, the ventricular hemorrhage is present in addition to ventricular dilatation; and grade 4, parenchymal hemorrhage is present [57]. A similar grading system by Volpe, which is based on cranial ultrasound scans, is also practical. Grade 1 refers to hemorrhage confined to the subependymal GM, and grade 2 as hemorrhage within the lateral ventricle without ventricular dilation and/or hemorrhage occupying <50% of the ventricle. Grade 3 is defined by ventricular dilation and/or hemorrhage occupying >50% of the ventricle, whereas grade 4 is IVH with hemorrhage on the surrounding parenchyma [82]. In this classification, the parenchymal hemorrhagic lesion should not be described as “extension” of the hemorrhage from a large IVH on grade 4 [80]. Volpe explained that this discontinuity between IVH and IPH is caused by the different origins of bleeding from the GM or by subsequent venous hemorrhagic infarction [79-81]. A detail of the pathogenesis is provided in another article. Both grading systems developed by Papile and Volpe are the most widely accepted, although several others exist [44].
As regards the incidence of IVH in premature infants, a study of 1950 infants weighing ≤2250 g born between 1986 and 1995 was conducted in the USA [68]. The rates of IVH calculated by BW were as follows : 31.5% in <750 g, 29.8% in 751–1000 g, 15.8% in 1001–1250 g, 7.3% in 1251–1500 g, and 1.4% in 1551–2250 g. Recently, the detailed incidences of grade 3–4 IVH (n=7655) arranged by each 100-g increase in BW from 500 g to 1500 g and each GA from 23 to 36 weeks were reported in Israel [40]. According to this study, the incidence rate is 36.4% in 500–599 g, 13.8% in 900–999 g, and 2.6% in 1400–1499 g of BW. For GA, the incidence is 39.0% in infants aged 24 weeks, 18.5% in 27 weeks, 3.5% in 30 weeks, and 0.8% in 33 weeks. The incidence of IVH demonstrated an inverse correlation with GA [72].
The time of hemorrhage onset is day 1 of life in at least 50% of the affected infants, day 2 in 25%, and day 3 in 15%. Therefore, approximately 90% of the lesions can be identified 72 hours after birth [82]. Murphy et al. [54] investigated the short-term outcomes of 248 infants who had BW of <1500 g and were admitted between 1994 and 1997. They clinically identified the incidence of each grade of IVH : grade 1 was observed in 104 (43.5%), grade 2 in 65 (26.2%), grade 3 in 45 (18.1%), and grade 4 in 34 (13.7%) patients. Vassilyadi et al. [78] also analyzed the incidence of IVH in 284 premature infants by grade; 48% had grade 1 IVH, 18% had grade 2, 15% had grade 3, and nearly 19% had grade 4 IVH, which were slightly higher in grade 1 (35% from Volpe [82]) than in grade 2 (40% from Volpe [82]) as reported previously.
In the first 14 days of life by each grade, the death rate was 2.9% in grade 1, 9.2% in grade 2, 17.8% in grade 3, and 29.4% in grade 4 [54]. Of 248 infants, 27 died (mortality rate 10.9%), which was related to the severity of the hemorrhage as Volpe had also mentioned.
The rates of progressive ventricular dilatation (PVD) in each grade of hemorrhage were as follows : 4.0% in grade 1, 11.9% in grade 2, 75.7% in grade 3, and 70.8% in grade 4 [54]. The total PVD rate was 25.3% (56 of 221 infants), which was completely consistent with the observation by Volpe, describing that approximately 25% of survivors developed PVD [82]. Moreover, very preterm infants who develop a severe IVH or a periventricular hemorrhagic infarction are prone to posthemorrhagic ventricular dilatation (PHVD) due to an imbalance in cerebrospinal fluid (CSF) production and reabsorption [19]. Depending on the duration of gestation, the odds of PVD occurrence decreased by 0.85 for each week of increase in gestation [54]. Similarly, the odds ratio (OR) for the IVH grade suggests that with each stepwise increase in the IVH grade, for example, from grade 2 to grade 3, the odds of PVD occurring increased more than fivefold [54].
As regards the natural course of PHVD, Murphy et al. [54] found that 53 (24%) of 221 infants with VLBW had ventricular dilatation but had no progression and 112 (51%) had no ventricular dilation. Hence, physicians must pay special attention to the question of whether any invasive treatments, for example, lumbar or fontanel puncture, surgical interventions to alleviate ventricular dilatation temporarily or permanently, are truly required. Even after comparing perinatal status by gestational weeks, BW, clinical risk index for babies, maximum Pco2, blood pressure, and antenatal steroid use between infants who did develop PVD and those who did not, the strongest predictor of the occurrence of PVD was the severity of IVH [54].
A small GMH/IVH is usually diagnosed by a neonatologist via screening cranial ultrasound and is carefully monitored whether it does enlarge and ventricular dilatation appeared. The diagnosis of GMH/IVH suspected based on clinical signs alone could be accurately reached in only approximately 50% of patients [27]. Together with serial imaging follow-up, neurological changes in cranial parameters are detected by increased intracranial pressure (ICP). As clinical manifestations, the fontanel is touched full and tense, becoming nonpulsatile. The rapid increase in the head circumference, which is crossing the percentile curve, and separated cranial sutures are also important and most reliable indicators [63]. Along with these parameters, apnea, vomiting, sun setting, bradycardia, decreased spontaneous activity, and sometimes an irritable state may be observed as systemic symptoms [27,46].
To estimate the long-term neurodevelopmental outcomes of preterm infants with IVH and IPH, an observational study published in 1986 by Guzzetta et al. [25] reported the importance of periventricular intraparenchymal echodensities (IPE) of neonatal ultrasound, which was as persuasive as ever. IPE was defined as any periventricular echodensity >1 cm in at least one dimension, and the presence of IPE well correlated with the degree of parenchymal injury and unfavorable outcomes of premature infants with mild-to-severe IVH/IPH [82]. IPE occurred on the same side with larger amounts of intraventricular blood, depending on the asymmetric diversity of injury [25]. In 87% of grade 3 IVH survivors who exhibited major motor deficits, the deficits correlated with the topography of the IPE and thus consisted of either spastic hemiparesis or asymmetric spastic quadriparesis [82]. As described here, the neuropathologic correlation indicated that IPE presumably represented hemorrhagic necrosis of the periventricular tissue. Actually, the outcomes varied with IPE severity. Of the survivors with extensive IPE, all had subsequent major motor deficits, and most of them exhibited cognitive function <80% of normal [25]. Of the 75 infants affected, the mortality rate was high in infants with extensive IPE (30/37; 81%) and those with localized IPE (14/38; 37%) [82]. Thus, with extensive IPE, there is little or no chance of survival with normal neurological and cognitive outcomes. With localized IPE, although major motor deficits are common (12/15; 80%), an appreciable proportion of infants have cognitive functions in the normal range. Careful and quantitative assessments of the ultrasonographic features of IPE are of major value in estimating outcomes [25].
To evaluate long-term outcomes according to each severity of hemorrhage (i.e., quantity of the intraventricular blood), the approximate incidence of definite neurological sequelae was investigated [82] : neurological sequelae occurred in 5% of patients with mild hemorrhage (comparable to grade 1); 15%, moderate (grade 2); 35%, severe (grade 3); and 90%, severe plus apparent periventricular hemorrhagic infarction (grade 4).
Recently, Mukerji et al. [53] conducted a meta-analysis of neurodevelopmental outcomes associated with periventricular/IVH (PIVH) in preterm infants. Their results confirm that severe PIVH leads to worse neurodevelopmental outcomes than mild or no PIVH, which is thought to be a fair statement. An intriguing result was that among survivors, mild PIVH was associated with higher odds of moderate-to-severe neurodevelopmental impairment than the absence of PIVH (OR, 1.48; 95% confidence interval [CI], 1.26–1.73), suggesting that mild PIVH may have independent effects compared with the absence of PIVH [53]. However, the basic question of why mild PIVH without any apparent injury to the brain parenchyma would cause neurologic impairment was raised. Mild PIVH essentially includes hemorrhage limited to the subependymal lining of the GM (grade 1 PIVH) and/or within the ventricle (grade 2 PIVH). The results of this meta-analysis also clarified that the adjusted odds of cerebral palsy and cognitive delay at 18–24 months were higher with severe PIVH but not with mild PIVH. The authors speculated that mild PIVH detected on ultrasound, not magnetic resonance imaging or other measures with high spatial resolution, may have concomitant, but undetected, parenchymal white matter injury, which may contribute to the long-term outcomes of mild PIVH [53].
Payne et al. [59] assessed 1472 infants born at less than 27 weeks GA and described that the neurodevelopmental outcomes at 18–22 months of corrected age of infants with extremely low GA and low-grade PIVH (grades 1–2) are not significantly different from those without hemorrhage.
A goal of the general management of preterm infants is to maintain intact survival. The prevention of GMH/IVH is the first imperative. Once hemorrhage occurs, appropriate treatment of posthemorrhagic hydrocephalus (PHH) is very important as a second step.
For circulatory management in GMH/IVH prevention, the optimal blood pressure for preterm infants should be initially clarified. The lower and upper limits of normal blood pressure for gestational and postnatal age in infants born extremely preterm are unknown [77]. Many neonatologists routinely treat infants whose mean arterial blood pressure is less than that for their GA in weeks [9]. Toyoshima et al. [75] suggested that the vein in the subependymal GM has poor supporting tissues and a thin vascular wall, and both have increased bleeding risk when the venous pressure increases. Therefore, venous congestion is more associated with IVH than with arterial blood pressure. Thus, these hemorrhages cannot be prevented with cardiotonic therapy judged from the blood pressure data alone [75].
An international analysis of 10 neonatal networks that present 11 regions or countries, i.e., Australia/New Zealand, Canada, Israel, Japan, Spain, Sweden, Switzerland, and the United Kingdom, showed that Japan had the lowest rates of mortality and severe IVH of preterm infants [67]. This study analyzed 58004 infants who had BW of <1500 g at 24 (0)–31 (6) weeks of gestation from databases, and the authors compared a composite outcome (mortality or any of grade ≥3 peri-IVH) between each country and all others. Isayama [34] discussed the reason for the lower rates of mortality and severe IVH in Japan. One of the potential reasons is that Japanese neonatologists frequently, twice or more times per day, perform functional echocardiography on extremely preterm infants within 3 days of birth. According to a national survey of the current features of neonatological practice in Japan [52], neonatologists use echocardiography to assess the 1) left ventricular ejection fraction (LVEF), LV end-diastolic diameter, left atrium-to-aortic root ratios, and diameter of the inferior vena cava as indicators of cardiac function and volume status; 2) diameter and shunt flow of the patent ductus arteriosus (PDA) and diastolic flow of the left pulmonary artery for the evaluation of the ductus arteriosus status; 3) blood flow pattern of the anterior cerebral artery, renal artery, and superior mesenteric artery as indicators of the peripheral organ circulation; and 4) shape and motion of the interventricular septum and tricuspid regurgitation for the evaluation of pulmonary hypertension. The early and precise detection of circulatory problems through these echocardiographic findings along with clinical signs may guide the appropriate circulatory management of extremely preterm infants [34]. Furthermore, monitoring the end-systolic wall stress (ESWS) of the left ventricle to assess the afterload of the left ventricle, which was adopted by approximately two-thirds of neonatal intensive care units, was reported as very unique in Japan [34]. The LVEF, commonly used for the evaluation of cardiac function, is influenced by the preload, afterload, and heart rate [14] and is not reliable enough to evaluate cardiac function in the unstable circulation of premature infants shortly after birth. Toyoshima et al. [75] originally proposed the efficacy of monitoring of the ESWS of the left ventricle to assess the afterload of the left ventricle and the mean velocity of circumferential fiber shortening (mVcfc), which is an index of LV pump function. By obtaining the stress-velocity relationship (mVcfc and ESWS relationship) (Fig. 1), they found that the incidences of intraventricular and/or pulmonary hemorrhages were higher in infants with an excessive afterload, which decreased LV function. The poor LV function that results from the excessive afterload indicated by a high ESWS was treated not by catecholamine, which increases afterload, but vasodilators as exemplified by nitroglycerin or milrinone, which mitigates afterload, diuretic, and sedation [69]. This could effectively prevent IVH [75]. A high ESWS accompanied by poor cardiac function is considered a risk factor for IVH due to afterload mismatch [75].
By using ultrasound, Ikeda et al. [30] uniquely reported that a high-grade perfusion waveform fluctuation in the ICV of extremely low birth weight infants was associated with IVH. The blood flowing from the veins in bilateral subependymal GM merges into the ICV. Hence, by focusing on venous Doppler measurement, not on arterial assessment, to predict IVH in preterm infants, they measured perfusion waveforms on the ICV via the anterior fontanel, which were classified into four patterns, starting with grade 0 if they showed a steady flow waveform and increasing up to grade 3 as the magnitude of fluctuation increased (Fig. 2). The incidence of IVH was high in low GA infants who exhibited high-grade fluctuations in the perfusion waveform of the ICV, whereas mature infants rarely exhibited high-grade fluctuations in the ICV, which were never observed even with increased central venous pressure, such as in heart failure [31]. Some plausible pathogenesis of the relationship between high-grade fluctuations in the perfusion waveform of the ICV and IVH were discussed. From the venous side, the jugular venous system has no valve. Hence, fluctuations in central venous pressure are transmitted intracranially, especially in premature infants. High-grade fluctuations in the perfusion waveform for the ICV may indicate the influence of the central venous pressure on microcirculation at the site of the IVH [31]. From the arterial side, cerebral autoregulation failure in premature infants is suspected. Premature and mature infants may have susceptibility differences based on the ability to maintain a relatively constant cerebral blood flow despite fluctuations in arterial blood pressure. A high-grade fluctuation in the perfusion waveform of the ICV above grade 2, suggesting that the waveform fluctuates, but the minimum speed is less than half the maximum speed and never drops to 0 cm/s, may predict the occurrence of subsequent hemorrhage from the GM and IVH. Therefore, antihypertension therapy, vasodilator use, and PDA treatment are considered.
Additionally, to prevent GMH/IVH, different clinical management aspects have been reported, including the effect of mechanical ventilation [3,4], efficacy of routine antenatal infection screening of chorioamnionitis or ureaplasma [29], antenatal maternal corticosteroid [7,8,10,24,49,66,70,71,73,74,87], regardless of the delivery mode [62], advantageous care of the umbilical cord at delivery [2,28,37,60,61], and usefulness of indomethacin administration [1,5,6,21,26,50,51,58,65]. These are meticulously discussed elsewhere.
Serial tapping of the CSF by LP (or ventricular puncture) has previously been used with some degree of efficacy. de Vries et al. [16] reported that two or three LPs reduced the need for surgery in up to one-quarter of all cases. Thus, the use of LPs in the immediate period after the identification of significant ventricular dilatation can help in temporary or total avoidance of surgical intervention [19]. However, a significant volume of the CSF must be removed daily (at least 10 mL/kg) for 2–3 weeks, and this can be burdensome for the infant and the treating physician. Moreover, a Cochrane review concluded that this treatment failed to reduce disability or the need for ventriculoperitoneal (VP) shunt surgery and was further associated with an increased risk of infection [86]. In 2004, Mazzola et al. [48] analyzed a randomized controlled study and three non-randomized studies to establish the optimal treatment strategies for the efficacy of serial LP as a Pediatric Hydrocephalus Systematic Review and Evidence-Based Guidelines Task Force. They advised against the routine use of serial LP to reduce the need for shunt placement or avoid the progression of hydrocephalus in premature infants [48]. The recommendation was level I and had high clinical certainty. Taken together, the routine use of LPs for avoiding future shunting in PHH is no longer recommended.
Pharmacological therapy with diuretics such as acetazolamide and furosemide, which reduce CSF production, has also been widely used as a non-invasive therapy to treat patients with PHH and avoid the need for a VP shunt [20]. Acetazolamide, a carbonic anhydrase inhibitor, decreases CSF production by >50%, and furosemide, a loop diuretic, also decreases CSF production [85]. The medical regimen consisted of furosemide 1 mg/kg/day intravenously or orally and acetazolamide starting at 20 mg/kg/day increasing by 10 mg/kg/day up to 100 mg/kg/day. Despite pervasive daily treatment, a few clinical trials have determined the effect of these diuretics. A randomized controlled trial of 177 infants by the International PHVD Drug Trial Group [33] analyzed the outcomes of 151 infants. Death or shunt placement occurred in 49 of 75 infants who received diuretics plus standard therapy, compared with 35 of 76 who received standard therapy alone. In addition, 1-year data of 129 infants were analyzed for mortality and morbidity; 84% (52/62) of the infants who received drug therapy died or had a disability or impairment at 1 year compared with 60% (40/67) of those who received standard therapy (relative risk, 1.40; 95% CI, 1.12–1.76; p=0.012), suggesting that the use of acetazolamide and furosemide in preterm infants with PHVD is associated with a higher rate of shunt placement and increased neurological morbidity. Although infants who need diuretics to manage CSF accumulation have clinically much severe CSF malabsorption in their hydrocephalic conditions, a Cochrane review also concluded that acetazolamide and furosemide therapy is neither effective nor safe and cannot be recommended in treating PHVD [85].
Numerous surgical management methods of PHH have been reported so far. Most of them fall into three main categories : 1) early CSF diversions as a temporizing measure such as external ventricular drainage, ventricular access device, and ventriculo-subgaleal shunt; 2) efforts to decrease intraventricular blood clot by which CFS absorption conceivably improve, i.e., neuroendoscopic lavage of the hematoma, trial of drainage, irrigation and fibrinolytic therapy); and 3) permanent shunt such as VP shunt, ventriculoatrial shunt, and endoscopic third ventriculostomy. Many treatment choices have both merits and demerits. The indication, evaluation, and argument of this surgical management are discussed elsewhere.
Although the research was performed in the 1990s, a study by Murphy et al. [54] focusing on the natural history of posthemorrhagic ventriculomegaly in VLBW infants was highly suggestive. They investigated 248 VLBW infants with IVH, and 221 survived for >14 days. Each grade of IVH was distributed as follows : 43.5% for grade 1, 26.2% for grade 2, 18.1% for grade 3, and 13.7% for grade 4. Of these 221 survivor infants, no ventricular dilatation was documented in 112 infants (51%). In the remaining infants, 56 (25%) were found to have PVD on serial cranial ultrasound scans after IVH. Notably, 53 infants (24%) showed ventricular dilatation but no progression. This means that cases of spontaneous arrest of ventricular dilatation were not fewer than expected. Therefore, physicians must determine whether ventricular dilatation in an infant with IVH becomes progressive. If progressive, the decision about when and how the infant should be intervened is agonizing because intervention-related complications in a very low birth-weight body are not negligible.
To compare differences in neurodevelopmental outcomes by the timing of intervention for preterm infants with PHH, Leijser et al. [45] observed 127 preterm infants with management policies divided into two, namely, “early approach” (EA) and “late approach” (LA). EA (n=78) represents a treatment strategy based on ventricular measurements using ultrasound exceeding the normal (ventricular index [VI] <+2 standard deviation [SD] / anterior horn width [AHW] <6 mm) described by Levene [47] and Davies et al. [15] Consequently, patients received interventions with initial temporizing procedures before the development of symptoms. Temporizing procedures included CSF drainage from LPs or ventricular reservoir, if needed, followed by a permanent neurosurgical intervention such as VP shunt placement. By contrast, LA (n=49) was based on clinical signs of ICP, mostly with immediate permanent neurosurgical intervention. As a result, 49 infants (63%) in the EA group and 24 (49%) in the LA group underwent intervention. While the EA group mostly had LPs as an initial intervention, most of the infants in the LA group received VP shunt as an initial intervention [45]. Surprisingly, the overall rate of VP shunt placement was lower in the EA group than in the LA group (20% vs. 92%; p<0.001). Although reservoir-related complications were slightly more common in the EA group, substantially more shunt-related complications occurred in the LA group, particularly infection and the need for revision. Interventionrelated death occurred only in the LA group, and 11 infants in the EA group and 20 in the LA group died between 10 days and 6 weeks of birth [45]. Furthermore, at 18–24 months of corrected age, the majority of survivors in the EA group (55/62) had a normal range (within 1 SD of mean) of cognitive and motor outcome scores, regardless of the need for intervention [44]. By contrast, most survivors in the LA group had moderate-to-severe abnormal outcomes (14/27) and cognitive and motor scores were lower in survivors with intervention than in those without intervention [45]. Accordingly, the authors speculated that late intervention during the development of chronic ventricular dilatation and pressure onto the immature white matter caused an irreversible injury of the premature brain, suggesting interference of cerebral perfusion due to brain distortion, ischemia, free radical formation, inflammation [84], and so forth, even if the CSF pressure returned to normal by subsequent surgical interventions [45]. Studies using near-infrared spectroscopy and Doppler techniques have shown that cerebral perfusion [39], oxygenation, and blood flow [76] are progressively impaired with worsening ventricular dilatation, paralleled with increasing abnormality of amplitude-integrated electroencephalography background [56] and delayed latencies of evoked potentials [17]. These neurochemical and neurophysiological changes occur even before signs of increased ICP develop and normalize following CSF drainage [17,43,55,56]. Hence, early intervention may prevent white matter damage, which is associated with future cognitive and motor impairment and eventually cerebral palsy in premature infants with IVH.
The Early versus Late Ventricular Intervention Study socalled “ELVIS” trial was also the most recent randomized controlled trial [16]. In this study, 126 preterm infants ≤34 weeks of gestation with PHVD after grades 3–4 hemorrhage were randomized to two groups : low threshold (LT) (VI >97 percentile and AHW >6 mm) or high threshold (HT) (VI >97 percentile + 4 mm and AHW >10 mm) groups. To reduce VI and the eventual need for a VP shunt, CSF tapping by LPs up to a maximum of three times, followed by taps from a ventricular reservoir, were tried. Contrary to expectations, no significant difference in the composite main outcome of VP shunt or death was shown (30% in the LT group versus 37% in the HT group, p=0.45). VP shunt was inserted in 12/64 (19%) of the infants in the LT and 14/62 (23%) infants in the HT group. In addition, 58% of infants in the LT group and 7% in the HT group required shunt revision (p<0.01). Conclusively, no significant difference in VP shunt placement or death in infants with PHVD was found between the LT and HT groups [16].
This ELVIS trial had later updates on (A) the assessment of additional brain injury and ventricular volume using termequivalent age magnetic resonance imaging [13] and (B) death or severe neurodevelopmental disability at 2 years [12]. These substudies clarified that (A) more brain injuries, including not only the white matter but also the gray matter and cerebellum, and larger ventricular volumes, were observed in the HT group than in the LT group using Kidokoro score [42], and (B) the LT group with VP shunt had cognitive and motor scores measured by Bayley cognitive/motor score similar to those without VP shunt (p=0.3 for both), whereas in the HT group, those with VP shunt had significantly lower scores than those without VP shunt (p=0.01 and p=0.004, respectively), suggesting that earlier intervention was associated with a lower odds of death or severe neurodevelopmental disability at 24 months corrected age in preterm infants with progressive PHVD [12].
The design criteria of other studies that discussed the timing of intervention in preterm infants with progressive PHVD are as follows : VI >97th percentile + 4 mm [35,41,84], AHW >4 mm, thalamo-occipital distance >26 mm, third ventricle width >3 mm [84], fronto-occipital horn ratio >0.55 plus 2 of 3 clinical manifestations, namely, bradycardia, split suture, bulging fontanel [83], and ICP signs [45]. As an inherent bias, because of severe prematurity, physicians are more compelled to intervene late with a HT inevitably. Meanwhile, from the findings of several randomized controlled trials, intervention at an early stage with a lower threshold before becoming symptomatic toward a high ICP may be recommended more than that one at the late stage with a HT. Therefore, early draining of adequate amounts of CSF before becoming symptomatic may yield favorable outcomes in the management of PHH in preterm infants.
The management strategy for PHH in premature infants still has controversies even though the morbidity and mortality of those infants, in recent decades, have gradually decreased. The ultimate goal is intact survival. Further studies are needed to elucidate how the risk of IVH could be minimized and the potential of immature infants’ brain could be maximized.
Notes
References
1. Abu-Shaweesh JM, Almidani E. PDA: does it matter? Int J Pediatr Adolesc Med. 7:9–12. 2020; Erratum in : Int J Pediatr Adolesc Med 7 : 212, 2020.

2. Al-Wassia H, Shah PS. Efficacy and safety of umbilical cord milking at birth: a systematic review and meta-analysis. JAMA Pediatr. 169:18–25. 2015.

3. Aly H, Hammad TA, Essers J, Wung JT. Is mechanical ventilation associated with intraventricular hemorrhage in preterm infants? Brain Dev. 34:201–205. 2012.

4. Aly H, Massaro AN, Patel K, El-Mohandes AA. Is it safer to intubate premature infants in the delivery room? Pediatrics. 115:1660–1665. 2005.

5. Bada HS, Green RS, Pourcyrous M, Leffler CW, Korones SB, Magill HL, et al. Indomethacin reduces the risks of severe intraventricular hemorrhage. J Pediatr. 115:631–637. 1989.

6. Bandstra ES, Montalvo BM, Goldberg RN, Pacheco I, Ferrer PL, Flynn J, et al. Prophylactic indomethacin for prevention of intraventricular hemorrhage in premature infants. Pediatrics. 82:533–542. 1988.

7. Been JV, Degraeuwe PL, Kramer BW, Zimmermann LJ. Antenatal steroids and neonatal outcome after chorioamnionitis: a meta-analysis. BJOG. 118:113–122. 2011.

8. Bolt RJ, van Weissenbruch MM, Lafeber HN, Delemarre-van de Waal HA. Glucocorticoids and lung development in the fetus and preterm infant. Pediatr Pulmonol. 32:76–91. 2001.

9. Cayabyab R, McLean CW, Seri I. Definition of hypotension and assessment of hemodynamics in the preterm neonate. J Perinatol 29 Suppl. 2:S58–S62. 2009.

10. Chapman K, Holmes M, Seckl J. 11β-hydroxysteroid dehydrogenases: intracellular gate-keepers of tissue glucocorticoid action. Physiol Rev. 93:1139–1206. 2013.

11. Christian EA, Jin DL, Attenello F, Wen T, Cen S, Mack WJ, et al. Trends in hospitalization of preterm infants with intraventricular hemorrhage and hydrocephalus in the United States, 2000-2010. J Neurosurg Pediatrics. 17:260–269. 2016.

12. Cizmeci MN, Groenendaal F, Liem KD, van Haastert IC, BenaventeFernández I, van Straaten HLM, et al. Randomized controlled early versus late ventricular intervention study in posthemorrhagic ventricular dilatation: outcome at 2 years. J Pediatr. 226:28–35.e3. 2020.
13. Cizmeci MN, Khalili N, Claessens NHP, Groenendaal F, Liem KD, Heep A, et al. ELVIS study group. Assessment of brain injury and brain volumes after posthemorrhagic ventricular dilatation: a nested substudy of the randomized controlled ELVIS trial. J Pediatr. 208:191–197.e2. 2019.
14. Colan SD, Borow KM, Neumann A. Left ventricular end-systolic wall stress-velocity of fiber shortening relation: a load-independent index of myocardial contractility. J Am Coll Cardiol. 4:715–724. 1984.

15. Davies MW, Swaminathan M, Chuang SL, Betheras FR. Reference ranges for the linear dimensions of the intracranial ventricles in preterm neonates. Arch Dis Child Fetal Neonatal Ed. 82:F218–F223. 2000.

16. de Vries LS, Groenendaal F, Liem KD, Heep A, Brouwer AJ, van ‘t Verlaat E, et al. Treatment thresholds for intervention in posthaemorrhagic ventricular dilation: a randomised controlled trial. Arch Dis Child Fetal Neonatal Ed. 104:F70–F75. 2019.

17. De Vries LS, Pierrat V, Minami T, Smet M, Casaer P. The role of short latency somatosensory evoked responses in infants with rapidly progressive ventricular dilatation. Neuropediatrics. 21:136–139. 1990.

18. du Plessis AJ. The role of systemic hemodynamic disturbances in prematurity-related brain injury. J Child Neurol. 24:1127–1140. 2009.

19. El-Dib M, Limbrick DD Jr, Inder T, Whitelaw A, Kulkarni AV, Warf B, et al. Management of post-hemorrhagic ventricular dilatation in the infant born preterm. J Pediatr. 226:16–27.e3. 2020.
20. Ellenbogen JR, Waqar M, Pettorini B. Management of post-haemorrhagic hydrocephalus in premature infants. J Clin Neurosci. 31:30–34. 2016.

21. Fowlie PW, Davis PG, McGuire W. Prophylactic intravenous indomethacin for preventing mortality and morbidity in preterm infants. Cochrane Database Syst Rev. (7):CD000174. 2010.

22. Ghazi-Birry HS, Brown WR, Moody DM, Challa VR, Block SM, Reboussin DM. Human germinal matrix: venous origin of hemorrhage and vascular characteristics. AJNR Am J Neuroradiol. 18:219–229. 1997.
23. Gilard V, Tebani A, Bekri S, Marret S. Intraventricular hemorrhage in very preterm infants: a comprehensive review. J Clin Med. 9:2447. 2020.

24. Gilstrap LC, Christensen R, Clewell WH, D’Alton ME, Davidson EC Jr, Escobedo MB, et al. Effect of corticosteroids for fetal maturation on perinatal outcomes. NIH consensus development panel on the effect of corticosteroids for fetal maturation on perinatal outcomes. JAMA. 273:413–418. 1995.

25. Guzzetta F, Shackelford GD, Volpe S, Perlman JM, Volpe JJ. Periventricular intraparenchymal echodensities in the premature newborn: critical determinant of neurologic outcome. Pediatrics. 78:995–1006. 1986.

26. Hamrick SEG, Sallmon H, Rose AT, Porras D, Shelton EL, Reese J, et al. Patent ductus arteriosus of the preterm infant. Pediatrics. 146:e20201209. 2020.

27. Hill A : Neurological and Neuromuscular Disorders in MacDonald MG, Seshia MMK, Mullett MD (eds) : Avery’s Neonatology Pathophysiology & Management of the Newborn, ed 6. Philadelphia : Lippincott William & Wilkins, 2005, pp1384-1409.
28. Hosono S, Mugishima H, Takahashi S, Takahashi S, Masaoka N, Yamamoto T, et al. One-time umbilical cord milking after cord cutting has same effectiveness as multiple-time umbilical cord milking in infants born at <29 weeks of gestation: a retrospective study. J Perinatol. 35:590–594. 2015.

29. Huang J, Meng J, Choonara I, Xiong T, Wang Y, Wang H, et al. Antenatal infection and intraventricular hemorrhage in preterm infants: a metaanalysis. Medicine (Baltimore). 98:e16665. 2019.
30. Ikeda T, Amizuka T, Ito Y, Mikami R, Matsuo K, Kawamura N, et al. Changes in the perfusion waveform of the internal cerebral vein and intraventricular hemorrhage in the acute management of extremely lowbirth-weight infants. Eur J Pediatr. 174:331–338. 2015.

31. Ikeda T, Ito Y, Mikami R, Matsuo K, Kawamura N, Yamoto A, et al. Fluctuations in internal cerebral vein and central side veins of preterm infants. Pediatr Int. 63:1319–1326. 2021.

32. Ikeda T, Ito Y, Mikami R, Matsuo K, Kawamura N, Yamoto A. Hemodynamics of infants with strong fluctuations of internal cerebral vein. Pediatr Int. 61:475–481. 2019.

33. International PHVD Drug Trial Group. International randomised controlled trial of acetazolamide and furosemide in posthaemorrhagic ventricular dilatation in infancy. Lancet. 352:433–440. 1998.
34. Isayama T. The clinical management and outcomes of extremely preterm infants in Japan: past, present, and future. Transl Pediatr. 8:199–211. 2019.

35. Johnson A, Wincott E, Grant A, Elbourne D. Randomised trial of early tapping in neonatal posthaemorrhagic ventricular dilatation: results at 30 months. Arch Dis Child Fetal Neonatal Ed. 71:F147. 1994.

36. Johnson S, Marlow N. Preterm birth and childhood psychiatric disorders. Pediatr Res. 69(5 Pt 2):11R–18R. 2011.

37. Katheria AC, Truong G, Cousins L, Oshiro B, Finer NN. Umbilical cord milking versus delayed cord clamping in preterm infants. Pediatrics. 136:61–69. 2015.

38. Kazan S, Güra A, Uçar T, Korkmaz E, Ongun H, Akyuz M. Hydrocephalus after intraventricular hemorrhage in preterm and low-birth weight infants: analysis of associated risk factors for ventriculoperitoneal shunting. Surg Neurol. 2(64 Suppl):S77–S81. discussion S81. 2005.

39. Kempley ST, Gamsu HR. Changes in cerebral artery blood flow velocity after intermittent cerebrospinal fluid drainage. Arch Dis Child. 69(1 Spec No):74–76. 1993.

40. Kenet G, Kuperman AA, Strauss T, Brenner B. Neonatal IVH--mechanisms and management. Thromb Res 127 Suppl. 3:S120–S122. 2011.
41. Kennedy CR, Ayers S, Campbell MJ, Elbourne D, Hope P, Johnson A. Randomized, controlled trial of acetazolamide and furosemide in posthemorrhagic ventricular dilation in infancy: follow-up at 1 year. Pediatrics. 108:597–607. 2001.

42. Kidokoro H, Neil JJ, Inder TE. New MR imaging assessment tool to define brain abnormalities in very preterm infants at term. AJNR Am J Neuroradiol. 34:2208–2214. 2013.

43. Klebermass-Schrehof K, Rona Z, Waldhör T, Czaba C, Beke A, Weninger M, et al. Can neurophysiological assessment improve timing of intervention in posthaemorrhagic ventricular dilatation? Arch Dis Child Fetal Neonatal Ed. 98:F291–F297. 2013.

44. Kuban K, Teele RL. Rationale for grading intracranial hemorrhage in premature infants. Pediatrics. 74:358–363. 1984.

45. Leijser LM, Miller SP, van Wezel-Meijler G, Brouwer AJ, Traubici J, van Haastert IC, et al. Posthemorrhagic ventricular dilatation in preterm infants: when best to intervene? Neurology. 90:e698–e706. 2018.
46. Leonard JR, Limbrick DD Jr. Intraventricular Hemorrhage and PostHemorrhagic Hydrocephalus in Albright AL, Pollack IF, Adelson PD (eds) : Principles and Practice of Pediatric Neurosurgery, ed 3. New York: Thieme Medical Publishers, Inc;2015. p. 137–144.
47. Levene MI. Measurement of the growth of the lateral ventricles in preterm infants with real-time ultrasound. Arch Dis Child. 56:900–904. 1981.

48. Mazzola CA, Choudhri AF, Auguste KI, Limbrick DD Jr, Rogido M, Mitchell L, et al. Pediatric hydrocephalus: systematic literature review and evidence-based guidelines. Part 2: management of posthemorrhagic hydrocephalus in premature infants. J Neurosurg Pediatr 14 Suppl. 1:8–23. 2014.

49. McGoldrick E, Stewart F, Parker R, Dalziel SR. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst Rev. (12):CD004454. 2020.

50. Ment LR, Duncan CC, Ehrenkranz RA, Kleinman CS, Taylor KJ, Scott DT, et al. Randomized low-dose indomethacin trial for prevention of intraventricular hemorrhage in very low birth weight neonates. J Pediatr. 112:948–955. 1988.

51. Ment LR, Oh W, Ehrenkranz RA, Phillip AG, Vohr B, Allan W, et al. Low-dose indomethacin therapy and extension of intraventricular hemorrhage: a multicenter randomized trial. J Pediatr. 124:951–955. 1994.

52. Miyata M, Toyoshima K, Yoda H, Murase M, Kawato H, Yamamoto K, et al. Extensive use of vasodilator agents and functional echocardiography to monitor extremely-low-birth-weight infants in Japan. J Neonatal Perinatal Med. 9:261–269. 2016.

53. Mukerji A, Shah V, Shah PS. Periventricular/intraventricular hemorrhage and neurodevelopmental outcomes: a meta-analysis. Pediatrics. 136:1132–1143. 2015.

54. Murphy BP, Inder TE, Rooks V, Taylor GA, Anderson NJ, Mogridge N, et al. Posthaemorrhagic ventricular dilatation in the premature infant: natural history and predictors of outcome. Arch Dis Child Fetal Neonatal Ed. 87:F37–F41. 2002.

55. Norooz F, Urlesberger B, Giordano V, Klebermasz-Schrehof K, Weninger M, Berger A, et al. Decompressing posthaemorrhagic ventricular dilatation significantly improves regional cerebral oxygen saturation in preterm infants. Acta Paediatr. 104:663–669. 2015.

56. Olischar M, Klebermass K, Hengl B, Hunt RW, Waldhoer T, Pollak A, et al. Cerebrospinal fluid drainage in posthaemorrhagic ventricular dilatation leads to improvement in amplitude-integrated electroencephalographic activity. Acta Paediatr. 98:1002–1009. 2009.

57. Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr. 92:529–534. 1978.

58. Park J, Yoon SJ, Han J, Song IG, Lim J, Shin JE, et al. Patent ductus arteriosus treatment trends and associated morbidities in neonates. Sci Rep. 11:10689. 2021.

59. Payne AH, Hintz SR, Hibbs AM, Walsh MC, Vohr BR, Bann CM, et al. Neurodevelopmental outcomes of extremely low-gestational-age neonates with low-grade periventricular-intraventricular hemorrhage. JAMA Pediatr. 167:451–459. 2013.

60. Rabe H, Gyte GM, Díaz-Rossello JL, Duley L. Effect of timing of umbilical cord clamping and other strategies to influence placental transfusion at preterm birth on maternal and infant outcomes. Cochrane Database Syst Rev. 9:CD003248. 2019.

61. Rabe H, Jewison A, Fernandez Alvarez R, Crook D, Stilton D, Bradley R, et al. Milking compared with delayed cord clamping to increase placental transfusion in preterm neonates: a randomized controlled trial. Obstet Gynecol. 117(2 Pt 1):205–211. 2011.

62. Riskin A, Riskin-Mashiah S, Bader D, Kugelman A, Lerner-Geva L, Boyko V, et al. Delivery mode and severe intraventricular hemorrhage in single, very low birth weight, vertex infants. Obstet Gynecol. 112:21–28. 2008.

63. Robinson S. Neonatal posthemorrhagic hydrocephalus from prematurity: pathophysiology and current treatment concepts. J Neurosurg Pediatr. 9:242–258. 2012.

64. Roze E, Kerstjens JM, Maathuis CG, ter Horst HJ, Bos AF. Risk factors for adverse outcome in preterm infants with periventricular hemorrhagic infarction. Pediatrics. 122:e46–e52. 2008.

65. Schmidt B, Davis P, Moddemann D, Ohlsson A, Roberts RS, Saigal S, et al. Long-term effects of indomethacin prophylaxis in extremely-lowbirth-weight infants. N Engl J Med. 344:1966–1972. 2001.

66. Sentilhes L, Sénat MV, Ancel PY, Azria E, Benoist G, Blanc J, et al. Prevention of spontaneous preterm birth: guidelines for clinical practice from the French College of Gynaecologists and Obstetricians (CNGOF). Eur J Obstet Gynecol Reprod Biol. 210:217–224. 2017.

67. Shah PS, Lui K, Sjörs G, Mirea L, Reichman B, Adams M, et al. Neonatal outcomes of very low birth weight and very preterm neonates: an international comparison. J Pediatr. 177:144–152.e6. 2016.
68. Sheth RD. Trends in incidence and severity of intraventricular hemorrhage. J Child Neurol. 13:261–264. 1998.

69. Simons SH, van Dijk M, van Lingen RA, Roofthooft D, Duivenvoorden HJ, Jongeneel N, et al. Routine morphine infusion in preterm newborns who received ventilatory support: a randomized controlled trial. JAMA. 290:2419–2427. 2003.

70. Skoll A, Boutin A, Bujold E, Burrows J, Crane J, Geary M, et al. No. 364-antenatal corticosteroid therapy for improving neonatal outcomes. J Obstet Gynaecol Can. 40:1219–1239. 2018.

71. Society for Maternal-Fetal Medicine (SMFM), Reddy UM, Deshmukh U, Dude A, Harper L, Osmundson SS. Society for Maternal-Fetal Medicine Consult Series #58: use of antenatal corticosteroids for individuals at risk for late preterm delivery: replaces SMFM Statement #4, implementation of the use of antenatal corticosteroids in the late preterm birth period in women at risk for preterm delivery, August 2016. Am J Obstet Gynecol. 225:B36–B42. 2021.
72. Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC, et al. Neonatal outcomes of extremely preterm infants from the NICHD neonatal research network. Pediatrics. 126:443–456. 2010.

73. Sweet DG, Carnielli V, Greisen G, Hallman M, Ozek E, Te Pas A, et al. European consensus guidelines on the management of respiratory distress syndrome - 2019 update. Neonatology. 115:432–450. 2019.

74. Takahashi T, Fee EL, Takahashi Y, Saito M, Yaegashi N, Usuda H, et al. Betamethasone phosphate reduces the efficacy of antenatal steroid therapy and is associated with lower birthweights when administered to pregnant sheep in combination with betamethasone acetate. Am J Obstet Gynecol. 226:564.e1–564.e14. 2022.

75. Toyoshima K, Kawataki M, Ohyama M, Shibasaki J, Yamaguchi N, Hoshino R, et al. Tailor-made circulatory management based on the stress-velocity relationship in preterm infants. J Formos Med Assoc. 112:510–517. 2013.

76. van Alfen-van der Velden AA, Hopman JC, Klaessens JH, Feuth T, Sengers RC, Liem KD. Cerebral hemodynamics and oxygenation after serial CSF drainage in infants with PHVD. Brain Dev. 29:623–629. 2007.

77. Vargo L, Seri I. New NANN practice guideline: the management of hypotension in the very-low-birth-weight infant. Adv Neonatal Care. 11:272–278. 2011.
78. Vassilyadi M, Tataryn Z, Shamji MF, Ventureyra EC. Functional outcomes among premature infants with intraventricular hemorrhage. Pediatr Neurosurg. 45:247–255. 2009.

79. Vesoulis ZA, Mathur AM. Cerebral autoregulation, brain injury, and the transitioning premature infant. Front Pediatr. 5:64. 2017.

80. Volpe JJ. Brain injury in the premature infant. Neuropathology, clinical aspects, pathogenesis, and prevention. Clin Perinatol. 24:567–587. 1997.
81. Volpe JJ. Edward B. Neuhauser lecture. Current concepts of brain injury in the premature infant. AJR Am J Roentgenol. 153:243–251. 1989.

82. Volpe JJ : Intracranial Hemorrhage: Germinal Matrix-Intraventricular Hemorrhage of the Premature Infant in Volpe JJ (ed) : Neurology of the newborn, ed 4. Philadelphia : W.B. Saunders Company, 2001, pp428-493.
83. Wellons JC 3rd, Shannon CN, Holubkov R, Riva-Cambrin J, Kulkarni AV, Limbrick DD Jr, et al. Shunting outcomes in posthemorrhagic hydrocephalus: results of a Hydrocephalus Clinical Research Network prospective cohort study. J Neurosurg Pediatr. 20:19–29. 2017.

84. Whitelaw A, Jary S, Kmita G, Wroblewska J, Musialik-Swietlinska E, Mandera M, et al. Randomized trial of drainage, irrigation and fibrinolytic therapy for premature infants with posthemorrhagic ventricular dilatation: developmental outcome at 2 years. Pediatrics. 125:e852–e858. 2010.

85. Whitelaw A, Kennedy CR, Brion LP. Diuretic therapy for newborn infants with posthemorrhagic ventricular dilatation. Cochrane Database Syst Rev. 2001:CD002270. 2001.

Fig. 1.
Stress-velocity relationship (mVcfc-ESWS) cited from the figure provided by Toyoshima et al. [75] with permission from Elsevier. Significant correlations were found between ESWS and mVcfc in both groups (mVcfc = 3.76 × ESWS-0.4; p<0.01, R=0.56). Group 1 : infants with complications (pulmonary hemorrhage, intraventricular hemorrhage, and periventricular leukomalacia; n=9). Group 2 : infants without complications (n=24). Systolic blood pressure and mean blood pressure changed over time, with no differences between the groups. mVcfc : mean velocity of circumferential fiber shortening, ESWS : end-systolic wall stress.
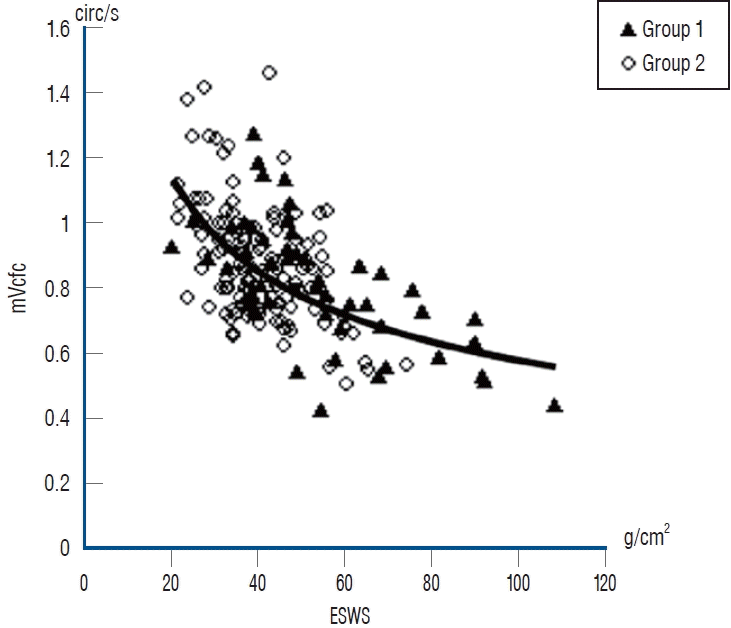
Fig. 2.
Classification of the perfusion waveforms of the internal cerebral vein on ultrasonography into four patterns cited from the figure provided by Ikeda et al. [32] with permission from John Wiley and Sons : (A) grade 0 (low grade), steady flow waveform with a constant perfusion speed; (B) grade 1 (low grade), the waveform fluctuates, but the minimum speed is never less than half the maximum speed; (C) grade 2 (high grade), the waveform fluctuates, and although the minimum speed is less than half the maximum speed, it never drop to 0 cm/s; (D) grade 3 (high grade), the waveform fluctuates, with the speed sometimes dropping to 0 cm/s.
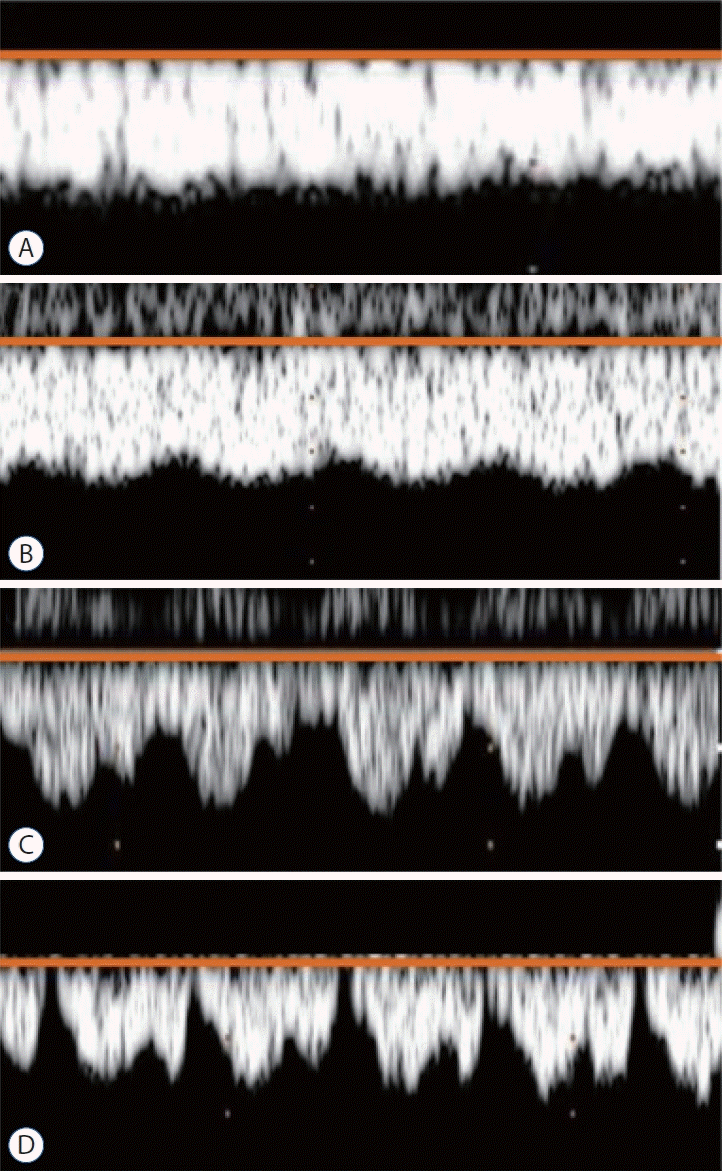
Table 1.
Classification of premature hemorrhage in preterm infants
| Based on CT scan by Papile et al. [57] | Based on ultrasound scan by Volpe [82] | |
|---|---|---|
| Grade 1 | Subependymal hemorrhage | GM hemorrhage with no or minimal IVH (10% of ventricular area on PSV) |
| Grade 2 | IVH without ventricular dilatation | IVH (10–50% of ventricular area on PSV) |
| Grade 3 | IVH wit ventricular dilatation | IVH (>50% of ventricular area on PSV, usually distends LV) |
| Grade 4 | IVH with parenchymal hemorrhage | IVH with hemorrhage on the surrounding parenchyma |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download