Abstract
Intraventricular hemorrhage (IVH) is a serious concern for preterm infants and can predispose such infants to brain injury and poor neurodevelopmental outcomes. IVH is particularly common in preterm infants. Although advances in obstetric management and neonatal care have led to a lower mortality rate for preterm infants with IVH, the IVH-related morbidity rate in this population remains high. Therefore, the present review investigated the pathophysiology of IVH and the evidence related to interventions for prevention. The analysis of the pathophysiology of IVH was conducted with a focus on the factors associated with cerebral hemodynamics, vulnerabilities in the structure of cerebral vessels, and host or genetic predisposing factors. The findings presented in the literature indicate that fluctuations in cerebral blood flow, the presence of hemodynamic significant patent ductus arteriosus, arterial carbon dioxide tension, and impaired cerebral venous drainage; a vulnerable or fragile capillary network; and a genetic variant associated with a mechanism underlying IVH development may lead to preterm infants developing IVH. Therefore, strategies focused on antenatal management, such as routine corticosteroid administration and magnesium sulfate use; perinatal management, such as maternal transfer to a specialized center; and postnatal management, including pharmacological agent administration and circulatory management involving prevention of extreme blood pressure, hemodynamic significant patent ductus arteriosus management, and optimization of cardiac function, can lower the likelihood of IVH development in preterm infants. Incorporating neuroprotective care bundles into routine care for such infants may also reduce the likelihood of IVH development. The findings regarding the pathogenesis of IVH further indicate that cerebrovascular status and systemic hemodynamic changes must be analyzed and monitored in preterm infants and that individualized management strategies must be developed with consideration of the risk factors for and physiological status of each preterm infant.
Intraventricular hemorrhage (IVH) is a major concern for preterm infants and is a predisposing factor for brain injury and poor neurodevelopmental outcomes. Improvements in perinatal and neonatal care have increased the survival rate of preterm infants, particularly those born at the gestational age of less than 25 weeks [113]. However, the incidence of IVH in such infants remains approximately 25–30% [18,24]. These findings indicate that determining optimal management strategies for IVH can be challenging for neonatologists. Therefore, obtaining a comprehensive understanding of the pathophysiology of IVH and evidence-based interventions for prevention may enable the development of individualised care for this vulnerable population. The present review investigated the underlying pathogenesis of IVH with support from available evidence on the topic and discussed potential IVH prevention strategies that can be employed during prenatal, perinatal, and early postnatal periods.
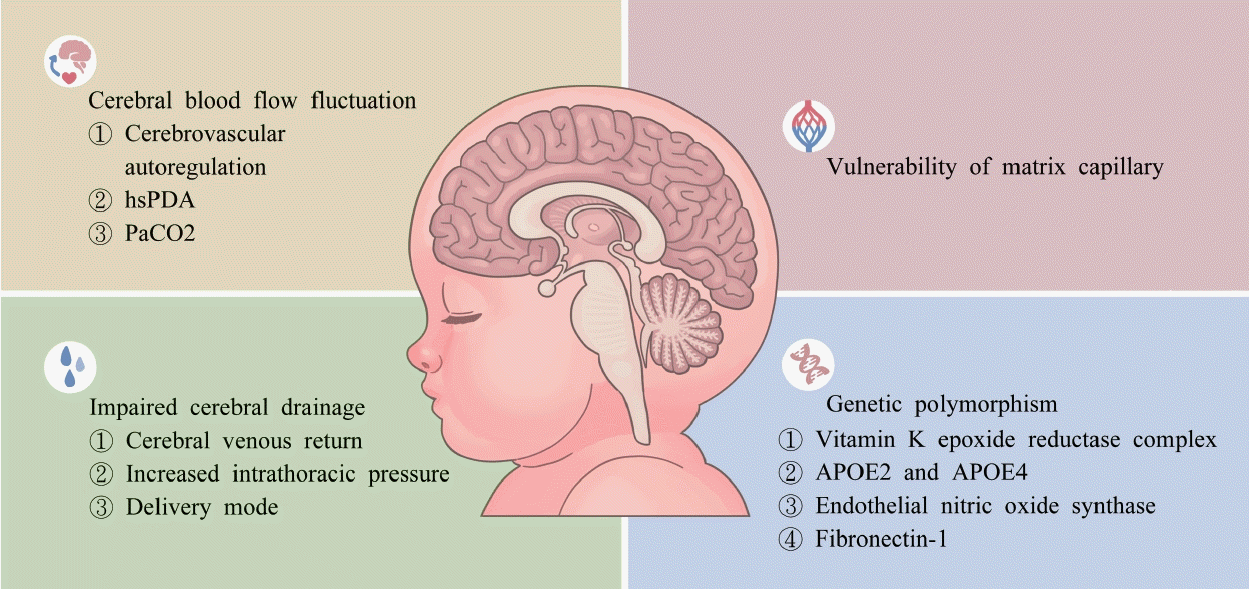
The pathophysiology of IVH in preterm infants is multifactorial and complex. The present review investigated the pathogenesis of IVH focusing on factors associated with cerebral haemodynamics, vulnerabilities in the structure of cerebral vessels, and host or genetic predisposing factors. Fig. 1 presents an overview of the pathogenesis of IVH.
Cerebrovascular autoregulation (CAR) plays an essential role in the maintenance of stable and adequate cerebral flow in the states of hypotension and hypoperfusion or hypertension. Lou et al. [65] examined impaired CAR (measured through 133 Xe clearance) in preterm infants with respiratory distress syndrome (RDS) alone and those with concurrent RDS and asphyxia. Another study used continuous-wave Doppler ultrasound to determine whether preterm infants experience considerable changes in cerebral blood flow (CBF) relative to changes in their mean arterial blood pressure (MABP) [83]. A growing body of evidence has supported the association among cerebral perfusion, CAR capacity, and severe IVH; this association was determined through near-infrared spectroscopy in sick infants [59,60]. Preterm infants have impaired CAR in the first few days after birth [59,116,123]. A study used near-infrared spectroscopy to demonstrate that very low birth weight neonates exhibit a considerable concordant change in both their cerebral intravascular oxygen levels and MABP that is consistent with impaired CAR [112]. Soul et al. [101] reported a high prevalence of cerebral pressure passivity in very low birth weight infants (96.7%; 87 of 90 infants), with the prevalence being higher in hypotensive infants. By contrast, several studies have reported CBF to have no association with MABP in preterm infants receiving high-frequency oscillatory ventilation support or before or after treatment for hypotension, which may indicate that preterm infants have intact CAR [63,78,120]. However, CAR is a dynamic and evolving process. Erratic haemodynamic changes occurring in pressure-passive cerebral circulation may precede IVH in preterm infants [59]. Thewissen et al. [107] reported that hypotensive preterm infants experienced significantly longer periods of cerebral hypoxia and impaired CAR and that these characteristics were associated with early IVH or death. Critically ill preterm infants with pressure-passive circulation were reported to have a higher rate of IVH occurrence than do neonates with effective autoregulation [112]. This indicates that blood pressure extremes are more harmful to vulnerable preterm infants. Vesoulis et al. [117] indicated that extreme MABP measurements (i.e., <23 and >46 mmHg) in preterm infants born at a gestational age of <30 weeks were associated with a significantly higher incidence of severe IVH.
The presence of hemodynamic significant patent ductus arteriosus (hsPDA) has been demonstrated to increase the risk of severe IVH in preterm infants [32,89]. However, findings regarding the effect of hsPDA on cerebral oxygenation and CBF have differed [28,115]. Chock et al. [17] discovered preterm infants without hsPDA to have nonsignificantly lower cerebral pressure-passive index scores than did those with hsPDA. In addition, the presence of hsPDA was reported to partly contribute to changes in CBF [77]. The association between hsPDA and IVH may be the result of complex interaction among hemodynamic instability, coagulation, and hsPDA management [13]. Although the detailed mechanism underlying this association is not well elucidated, the sustained patency of ductus arteriosus in combination with cardiac function immaturity was identified to potentially contribute to the occurrence of IVH [79]. Martini et al. [67] discovered that hsPDA was associated with an increase in cerebrovascular reactivity during the transition period. In preterm infants with hsPDA, a positive association was noted between changes in CBF and the severity of cardiac dysfunction, and the incidence of IVH was reported to be higher in such infants [48].
Arterial carbon dioxide tension (PaCO2) affects vasoreactivity and regulates CBF. Hypocarbia has been determined to be associated with higher risks of lung injury and IVH [31,33]. Permissive hypercarbia may be induced to protect the lungs and is commonly used in the care of preterm infants. However, excessively elevated PaCO2 can led to a 2-fold higher risk of IVH when PaCO2 becomes greater than 60 mmHg [33] and can lead to a 5-fold higher risk of IVH when PaCO2 becomes greater than 75 mmHg [49]. A retrospective study reported that fluctuations in PaCO2 may be a more prominent predisposing factor for severe IVH than the presence of hypercarbia alone is [3]. The positive correlation between the transcutaneous CO2 level and cerebral blood volume in premature infants indicates that such infants experience strong CO2 cerebrovascular reactivity [4]. Other factors, including a lower gestational age [92,118], having undergone dopamine treatment [67], prolonged hyperglycaemia [6], and lower initial haematocrit [51], have been reported to be independently associated with CBF fluctuations and a significant high risk of IVH in preterm infants.
The internal cerebral vein is generally used to evaluate the status of cerebral venous drainage. High-grade fluctuation in the internal cerebral vein was reported to be associated with IVH [46], and an increase in the preload, the diastolic pressure of the right ventricle, or intrathoracic pressure was determined to impede central venous return and increase in cerebral venous pressure [88]. Lower superior vena cava flow is caused by considerable shunting of hsPDA [10] and impaired myocardial function [56] and is associated with an increased risk of IVH [57].
High positive pressure ventilation settings can increase intrathoracic pressure and reduce central venous return. In addition, persistent higher mean airway pressure from high-frequency oscillatory ventilation was reported to increase the likelihood of developing IVH [76]. However, a meta-analysis of 17 randomised controlled trials indicated that the rates of infants receiving high-frequency oscillatory ventilation developing IVH did not significantly differ from those of infants receiving conventional ventilation [20]. Furthermore, infants receiving synchronised and volume-targeted ventilation were demonstrated to have a lower risk of severe IVH than did those receiving original pressure-limited ventilation because synchronised and volumetargeted ventilation involves the use of a lower intrathoracic pressure [55]. Pneumothorax in neonates with a gestational age of <28 weeks was reported to be a risk factor for IVH. This may be because increased intrathoracic pressure in combination with a lack of CAR can lead to impaired cerebral venous return [87].
Prolonged labour and excessive stretch force applied by hands or instruments can damage the cerebral venous system and result in intracranial haemorrhage in term neonates [12,84]. Prolonged labour was reported to be a risk factor for IVH [62]. Although several studies have indicated that IVH rates in children delivered using different modes do not differ [93,122], others have reported a positive correlation between vaginal delivery and IVH [21,45], particularly for neonates born at a gestational age of <26 weeks [44].
The capillaries of the germinal matrix have unique vascular infrastructure and a high metabolic demand for rapid angiogenesis. The features of a fragile capillary network include a paucity of pericytes, immature basement membranes, and poor support from muscles or collagen [30,124]. Primitive sinusoid capillaries generally remain undifferentiated in the germinal zone until term [91]. The subependymal matrix is a border zone located between the cerebral small arteries and deep cerebral vein. This location renders this matrix susceptible to hemodynamic instability and focal hypoxic changes [30].
Due to the advances in genomics, a growing body of evidence believed genetic variants to be involved in various mechanisms underlying IVH development. Polymorphisms in the gene encoding vitamin K metabolism (vitamin K epoxide reductase complex 1) [96] and transportation (APOE2 and APOE4) [29] have been demonstrated to influence the risk of IVH in preterm infants. Szpecht et al. [105] reported that endothelial nitric oxide synthase and fibronectin-1 polymorphism genes [103] are respectively associated with a 3.4- and 7-fold higher risk of IVH in preterm infants. However, because the results of genetic studies on IVH have been conflicting, additional studies are required to verify the role of genetic factors in the aetiology and pathogenesis of IVH [104,108].
Antenatal corticosteroid was demonstrated to reduce the severity of RDS and long-term morbidity [68]. Ment et al. [70] conducted a randomised controlled trial and discovered that preterm infants who received antenatal steroids had a lower risk of IVH. Administering a course of corticosteroids to women prior to their anticipated preterm births reduced the occurrence of IVH of all grades and particularly reduced the occurrence of severe IVH in preterm infants born at the gestational age of 22–29 weeks [121]. Antenatal steroids were indicated to have the highest efficacy in reducing the likelihood of IVH development when administered 24 hours to 7 days before birth [36]. Antenatal steroids were also demonstrated to reduce the risk of IVH indirectly by improving RDS and shortening the required duration of mechanical ventilation [42]. Although the mechanism underlying this effect remains unclear, it is likely related to the suppression of cerebral angiogenesis [119].
Tocolytics are widely used to reduce the likelihood of extreme preterm birth. However, findings regarding their effect on long-term neurodevelopmental outcomes in preterm infants have been conflicting. Pinto Cardoso et al. [86] conducted a population-based cohort study and discovered that a composite adverse outcome (death/severe IVH) was significantly lower in preterm infants with tocolytic exposure than in those without. Antenatal exposure of magnesium sulfate lowered the risk of severe intraventricular hemorrhage [7,9]. By contrast, a high dose of magnesium sulfate was suggested to increase the risk of IVH [75]. Mittendorf et al. [74] reported that antenatal exposure to magnesium sulfate exerted a paradoxical dose effect on neuroprotection. The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine continue to support the short-term (usually less than 48 hours) use of magnesium sulfate in obstetric care for fetal neuroprotection before anticipated early preterm (less than 32 weeks of gestation) delivery [127]. Intravenous administration of magnesium sulfate with one dose of 4–8 g in pregnant women who are at risk of preterm delivery within 7 days might benefit the preterm infants [126].
Delayed cord clamping (DCC) of more than 30 seconds at birth is crucial to ensure that a sufficient amount of blood and stem cells can transfuse from the placenta to the newborn [16,81]. DCC can improve cerebral oxygenation at birth [81] and reduce the incidence of iron deficiency anaemia at infancy [53] in term and late preterm infants. Although a meta-analysis revealed that DCC is not associated with a significant reduction in the incidence of IVH [35], some randomised controlled trials have demonstrated that compared with immediate cord clamping, DCC in extremely preterm infants is associated with a lower risk of severe IVH [43,47]. However, implementing DCC during preterm labour can be challenging because the maternal-neonatal condition is often unstable and resuscitation is often required. Umbilical cord milking (UCM) is an alternative method that offers advantages over to DCC because it does not necessitate a delay in resuscitation initiation. Compared with immediate cord clamping, UCM can immediately increase pulmonary blood flow to improve oxygenation [52] and reduce the need for blood transfusion [8,52]. Toledo et al. [109] discovered UCM to be associated with a lower IVH rate than that of immediate cord clamping in preterm infants at the gestational age of 27–32 weeks. However, UCM can cause a rapid increase in CBF and thus can significantly increase the risk of severe IVH compared with DCC, particularly in preterm infants born at a gestational age of 28 weeks or less [8,52]. The available evidence indicates that UCM cannot replace DCC as a placenta transfusion strategy in extremely preterm infants. If immediate resuscitation is necessary, it might be an alternative way to DCC that to lift the clamped umbilical cord high to transfuse cord blood into the neonate by gravity.
Numerous scholars have investigated whether caesarean section in preterm deliveries can reduce the rate of IVH development. Rahman et al. [90] and Ljustina et al. [64] have enrolled preterm infants born at the gestational age of 27–34 weeks and reported caesarean section to have no notable effect on IVH reduction. A retrospective study reported no association between the delivery mode and all forms of birth trauma, including IVH, in preterm deliveries [66]. However, other studies have indicated that elective caesarean section exerted a protective effect on IVH in preterm infants [2,42,45]. Hübner et al. [44] reported that preterm infants born at a gestational age of 24–25 weeks benefitted from antenatal steroid treatment in combination with caesarean section. Gamaleldin et al. [38] reported that IVH was less likely to occur if preterm infants born at less than 27 weeks of gestation were delivered by caesarean section. The American College of Obstetricians and Gynecologists suggested caesarean section might be considered and recommended for preterm infants at gestational age of 23–24 weeks and of 25 weeks, respectively [5]. Elective caesarean section might be considered as a strategy to prevent IVH for extremely preterm infants (who had birth weight less than 1000 g or born at less than 27 weeks of gestation) in Taiwan.
In addition to closing hsPDA, indomethacin has been suggested to prevent IVH in preterm infants because of its effects on blood vessels. Whether indomethacin or other nonsteroidal anti-inflammatory drugs can reduce the incidence and severity of IVH in preterm infants remains the topic of debate. A multicentre controlled trial demonstrated that low-dose prophylactic indomethacin administered between 6 and 12 hours after birth reduced the incidence or severity of IVH and had no notable adverse effects [71]. Kalani et al. [50] discovered that early ibuprofen administration had a similar IVH-preventive effect. Two recent meta-analyses revealed that indomethacin prophylaxis was associated with a lower rate of severe IVH [1,125]. In contrast, several retrospective studies and clinical trials reported that prophylactic indomethacin not only demonstrated no IVH-preventive effect but also increased the risks of impaired renal function and bleeding tendency [61,72,82]. Indomethacin might appear to be a promising candidate, but proper patient selection is the crux.
Perinatal transition from foetal to postnatal circulation causes considerable hemodynamic stress on the cardiovascular system. Preterm infants experience more extreme upheaval during the first few days of life than full-term neonates do because of their premature organ function. The disruption of placental circulation forces the malfunctional myocardium of preterm infants to experience an abruptly increased systemic arterial resistance as well as an increased afterload [19,106]. The presence of a left-to-right shunt of patent ductus arteriosus (PDA) can cause the left ventricle to experience volume overload and unstable CBF [17,77]. The incompetence of premature myocardial function can result in impaired cerebral venous drainage [56]. These hemodynamic changes may be worsened by the immature cerebrovascular regulation of preterm infants [59]. Therefore, precise circulatory management aimed at hemodynamic alteration is necessary to prevent IVH.
Blood pressure being at either extreme in preterm infants can be hazardous and is associated with IVH [117]. Extremely preterm infants have varying rates of hypotension (15–50%) [25]. Although no clear definition for normal blood pressure for preterm infants has been established, many studies and neonatologists have defined hypotension in preterm infants as an MABP value less than gestational age (in weeks) [15,27]. However, blood pressure is not a favourable indicator of cerebral or systemic perfusion. Therefore, administration of inotropic drugs or volume expansion for hypotensive preterm infants with clinical signs of favourable perfusion may be unnecessary [26]. Advances in cerebrovascular monitoring have indicated that optimal MABP may be defined as favourable cerebral oxygenation or a minimum cerebrovascular reactivity index value, as determined through near-infrared spectroscopy. Such a definition would prevent the overuse of inotropic drugs and unintentional aggregation of the severity of IVH [22,114]. Several studies indicate that hydrocortisone therapy is increasingly being used for refractory hypotensive preterm infants because such infants are proposed to have an inadequate hypothalamic-pituitary-adrenal axis response to stress during the first week of life [99,110].
Whether conservative monitoring or aggressively treating PDA leads to more favourable results remains the topic of debate. Those who support conservative management on the basis of frequent spontaneous closure occurring [98] lack evidence regarding such management’s association with decreased morbidity [73]. However, adverse events were verified to occur both in cases of medical and surgical closure [95]. Nevertheless, early screening and treatment of PDA were reported to be associated with a decrease in mortality and morbidity in preterm infants [54,94]. Most neonatologists decide to treat or not to treat PDA on the basis of clinical signs and sonographic evidence of PDA-related cardiac dysfunction and organ hypoperfusion. Early targeted therapy for PDA in properly selected preterm infants may improve clinical short-term and long-term outcomes [40].
Traditionally, pharmacological closure of hsPDA has been completed using an intravenous cyclooxygenase inhibitor. Such treatment has a closure rate of 70–80% [73]. A study, however, indicated that a high dose of oral ibuprofen may be the most effective alternative to such treatment and have fewer side effects [73]. Intravenous acetaminophen has gradually come to be considered a treatment of choice because its closure rate is similar to that of cyclooxygenase inhibitors and does not affect renal function or increase the risk of necrotising enterocolitis [80]. Surgical ligation and transcatheter closure of PDA can be used as back-up therapy for patients who fail medical treatment and continue to exhibit cardiopulmonary compromise [102].
The cardiac function of preterm infants is not suited to cope with an increased afterload and preload alteration. An animal model revealed the foetal myocardium to have a lower compliance than that of a mature heart [37]. The preterm heart exhibited low ventricle-arterial coupling during the early days of life. In addition, hsPDA was demonstrated to contribute to overload in the immature left ventricle and consistently lower contractility [11]. Cardiac dysfunction and uncoupling under conditions of an increased afterload have been reported to impede cerebral venous return and contribute to IVH in preterm infants [56,57]. The stress-velocity relationship was reported to be an effective indicator of cardiac function and is calculated from end-systolic wall stress (an index of left ventricle afterload) and the left ventricle rate-corrected mean velocity of circumferential fibre shortening (an index of left ventricle contractility). Serial echocardiographic assessment of the stress-velocity relationship can be employed to determine whether contractility has been improved by the administration of inotropic drugs and maintain the afterload at an acceptable level by using vasodilators [111].
Optimising management in the immediate post-delivery period—the first 72 hours of life—to reduce the incidence of IVH has been extensively investigated in the literature [14,100]. Widely accepted practices related to such management include (1) maintaining a supine midline with a neutral head position [58]; (2) tilting the incubator to 10° to 30° to avoid the head-down position [58]; (3) ensuring minimal handling, including suction [69]; (4) avoiding rapid flushes and blood withdrawal through intravenous or arterial routes [97]; (5) avoiding routine endotracheal suction [69]; and (6) initiating additional intervention for pain or stress relief in the form of non-nutritive sucking and oral breast milk or sucrose [41]. Several studies have indicated that neonatal neuroprotective care bundles that incorporate these practices can reduce the incidence and severity of IVH; however, studies that have applied such practices have obtained inconsistent results [23,34,39,85]. Therefore, additional studies are required to develop optimal management practices.
IVH in infants can contribute to complex and multifactorial interactions. Although advances in obstetric management and neonatal care have led to lower mortality in such infants, the incidence rate of IVH remains unchanged, which is a cause of considerable concern. Therefore, prevention strategies focused on both general care and individualised management must be reviewed.
The routine administration of antenatal corticosteroids, the use of magnesium sulphate, and maternal transfer to a specialised centre can lead to more favourable outcomes, including a lower likelihood of developing IVH, in preterm infants. In addition, incorporating a neuroprotective care bundle into the routine care provided to preterm infants can potentially reduce the rate of IVH development.
The interaction between the cerebrovascular status and systemic hemodynamic changes in preterm infants must be analysed and monitored. Management strategies should be individualised on the basis of specific risk factors for and physiological status of each preterm infant. In addition, the integration of a multifaceted prevention programme could lead to a reduction in the incidence of IVH.
Notes
References
1. Al-Matary A, Abu Shaheen A, Abozaid S. Use of prophylactic indomethacin in preterm infants: a systematic review and meta-analysis. Front Pediatr. 10:760029. 2022.

2. Alotaibi WSM, Alsaif NS, Ahmed IA, Mahmoud AF, Ali K, Hammad A, et al. Reduction of severe intraventricular hemorrhage, a tertiary singlecenter experience: incidence trends, associated risk factors, and hospital policy. Childs Nerv Syst. 36:2971–2979. 2020.

3. Altaany D, Natarajan G, Gupta D, Zidan M, Chawla S. Severe intraventricular hemorrhage in extremely premature infants: are high carbon dioxide pressure or fluctuations the culprit? Am J Perinatol. 32:839–844. 2015.

4. Aly S, El-Dib M, Lu Z, El Tatawy S, Mohamed M, Aly H. Factors affecting cerebrovascular reactivity to CO2 in premature infants. J Perinat Med. 47:979–985. 2019.

5. American College of Obstetricians and Gynecologists; Society for Maternal-Fetal Medicine. Obstetric care consensus No. 6: periviable birth. Obstet Gynecol. 130:e187–e199. 2017.
6. Auerbach A, Eventov-Friedman S, Arad I, Peleg O, Bdolah-Abram T, BarOz B, et al. Long duration of hyperglycemia in the first 96 hours of life is associated with severe intraventricular hemorrhage in preterm infants. J Pediatr. 163:388–393. 2013.

7. Ayed M, Ahmed J, More K, Ayed A, Husain H, AlQurashi A, et al. Antenatal magnesium sulfate for preterm neuroprotection: a single-center experience from Kuwait Tertiary NICU. Biomed Hub. 7:80–87. 2022.

8. Balasubramanian H, Ananthan A, Jain V, Rao SC, Kabra N. Umbilical cord milking in preterm infants: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 105:572–580. 2020.

9. Bansal V, Desai A. Efficacy of antenatal magnesium sulfate for neuroprotection in extreme prematurity: a comparative observational study. J Obstet Gynaecol India. 72:36–47. 2022.

10. Bates S, Odd D, Luyt K, Mannix P, Wach R, Evans D, et al. Superior vena cava flow and intraventricular haemorrhage in extremely preterm infants. J Matern Fetal Neonatal Med. 29:1581–1587. 2016.

11. Baumgartner S, Olischar M, Wald M, Werther T, Berger A, Waldhor T, et al. Left ventricular pumping during the transition-adaptation sequence in preterm infants: impact of the patent ductus arteriosus. Pediatr Res. 83:1016–1023. 2018.

12. Borna H, Rad SM, Borna S, Mohseni SM. Incidence of and risk factors for birth trauma in Iran. Taiwan J Obstet Gynecol. 49:170–173. 2010.

13. Brunner B, Hoeck M, Schermer E, Streif W, Kiechl-Kohlendorfer U. Patent ductus arteriosus, low platelets, cyclooxygenase inhibitors, and intraventricular hemorrhage in very low birth weight preterm infants. J Pediatr. 163:23–28. 2013.

14. Castrodale V, Rinehart S. The golden hour: improving the stabilization of the very low birth-weight infant. Adv Neonatal Care. 14:9–14. quiz 15-16. 2014.
15. Cayabyab R, McLean CW, Seri I. Definition of hypotension and assessment of hemodynamics in the preterm neonate. J Perinatol 29 Suppl. 2:S58–S62. 2009.

16. Chen X, Li X, Chang Y, Li W, Cui H. Effect and safety of timing of cord clamping on neonatal hematocrit values and clinical outcomes in term infants: a randomized controlled trial. J Perinatol. 38:251–257. 2018.

17. Chock VY, Ramamoorthy C, Van Meurs KP. Cerebral autoregulation in neonates with a hemodynamically significant patent ductus arteriosus. J Pediatr. 160:936–942. 2012.

18. Christian EA, Jin DL, Attenello F, Wen T, Cen S, Mack WJ, et al. Trends in hospitalization of preterm infants with intraventricular hemorrhage and hydrocephalus in the United States, 2000-2010. J Neurosurg Pediatr. 17:260–269. 2016.

19. Ciccone MM, Scicchitano P, Zito A, Gesualdo M, Sassara M, Calderoni G, et al. Different functional cardiac characteristics observed in term/preterm neonates by echocardiography and tissue doppler imaging. Early Hum Dev. 87:555–558. 2011.
20. Cools F, Offringa M, Askie LM. Elective high frequency oscillatory ventilation versus conventional ventilation for acute pulmonary dysfunction in preterm infants. Cochrane Database Syst Rev. (3):CD000104. 2015.

21. Costa STB, Costa P, Graca AM, Abrantes M; Portuguese National Registry of very low birth weight infants. Delivery mode and neurological complications in very low birth weight infants. Am J Perinatol. 2022; [Epub ahead of print].

22. da Costa CS, Czosnyka M, Smielewski P, Austin T. Optimal mean arterial blood pressure in extremely preterm infants within the first 24 hours of life. J Pediatr. 203:242–248. 2018.

23. de Bijl-Marcus K, Brouwer AJ, De Vries LS, Groenendaal F, Wezel-Meijler GV. Neonatal care bundles are associated with a reduction in the incidence of intraventricular haemorrhage in preterm infants: a multicentre cohort study. Arch Dis Child Fetal Neonatal Ed. 105:419–424. 2020.

24. de Figueiredo Vinagre LE, de Siqueira Caldas JP, Martins Marba ST, Procianoy RS, de Cassia Silveira R, Santiago Rego MA, et al. Temporal trends in intraventricular hemorrhage in preterm infants: a Brazilian multicenter cohort. Eur J Paediatr Neurol. 39:65–73. 2022.

25. Dempsey EM. What should we do about low blood pressure in preterm infants. Neonatology. 111:402–407. 2017.

26. Dempsey EM, Al Hazzani F, Barrington KJ. Permissive hypotension in the extremely low birthweight infant with signs of good perfusion. Arch Dis Child Fetal Neonatal Ed. 94:F241–244. 2009.

27. Dempsey EM, Barrington KJ. Diagnostic criteria and therapeutic interventions for the hypotensive very low birth weight infant. J Perinatol. 26:677–681. 2006.

28. Dix L, Molenschot M, Breur J, de Vries W, Vijlbrief D, Groenendaal F, et al. Cerebral oxygenation and echocardiographic parameters in preterm neonates with a patent ductus arteriosus: an observational study. Arch Dis Child Fetal Neonatal Ed. 101:F520–F526. 2016.

29. Dzietko M, Schulz S, Preuss M, Haertel C, Stein A, Felderhoff-Mueser U, et al. Apolipoprotein E gene polymorphisms and intraventricular haemorrhage in infants born preterm: a large prospective multicentre cohort study. Dev Med Child Neurol. 61:337–342. 2019.

30. Egesa WI, Odoch S, Odong RJ, Nakalema G, Asiimwe D, Ekuk E, et al. Germinal matrix-intraventricular hemorrhage: a tale of preterm infants. Int J Pediatr. 2021:6622598. 2021.

31. Erickson SJ, Grauaug A, Gurrin L, Swaminathan M. Hypocarbia in the ventilated preterm infant and its effect on intraventricular haemorrhage and bronchopulmonary dysplasia. J Paediatr Child Health. 38:560–562. 2002.

32. Evans N, Kluckow M. Early ductal shunting and intraventricular haemorrhage in ventilated preterm infants. Arch Dis Child Fetal Neonatal Ed. 75:F183–186. 1996.

33. Fabres J, Carlo WA, Phillips V, Howard G, Ambalavanan N. Both extremes of arterial carbon dioxide pressure and the magnitude of fluctuations in arterial carbon dioxide pressure are associated with severe intraventricular hemorrhage in preterm infants. Pediatrics. 119:299–305. 2007.

34. Ferreira DM, Girao ALA, AVS ES, Chaves EMC, de Almeida PC, Freire VS, et al. Application of a bundle in the prevention of peri-intraventricular hemorrhage in preterm newborns. J Perinat Neonatal Nurs. 34:E5–E11. 2020.

35. Fogarty M, Osborn DA, Askie L, Seidler AL, Hunter K, Lui K, et al. Delayed vs early umbilical cord clamping for preterm infants: a systematic review and meta-analysis. Am J Obstet Gynecol. 218:1–18. 2018.

36. Fortmann I, Mertens L, Boeckel H, Gruttner B, Humberg A, Astiz M, et al. A timely administration of antenatal steroids is highly protective against intraventricular hemorrhage: an observational multicenter cohort study of very low birth weight infants. Front Pediatr. 10:721355. 2022.

37. Friedman WF. The intrinsic physiologic properties of the developing heart. Prog Cardiovasc Dis. 15:87–111. 1972.

38. Gamaleldin I, Harding D, Siassakos D, Draycott T, Odd D. Significant intraventricular hemorrhage is more likely in very preterm infants born by vaginal delivery: a multi-centre retrospective cohort study. J Matern Fetal Neonatal Med. 32:477–482. 2019.

39. Gross M, Engel C, Trotter A. Evaluating the effect of a neonatal care bundle for the prevention of intraventricular hemorrhage in preterm infants. Children (Basel). 8:257. 2021.

40. Hamrick SEG, Sallmon H, Rose AT, Porras D, Shelton EL, Reese J, et al. Patent ductus arteriosus of the preterm infant. Pediatrics. 146:e20201209. 2020.

41. Hatfield LA, Murphy N, Karp K, Polomano RC. A systematic review of behavioral and environmental interventions for procedural pain management in preterm infants. J Pediatr Nurs. 44:22–30. 2019.

42. Helwich E, Rutkowska M, Bokiniec R, Gulczynska E, Hozejowski R. Intraventricular hemorrhage in premature infants with Respiratory Distress Syndrome treated with surfactant: incidence and risk factors in the prospective cohort study. Dev Period Med. 21:328–335. 2017.
43. Hemmati F, Sharma D, Namavar Jahromi B, Salarian L, Farahbakhsh N. Delayed cord clamping for prevention of intraventricular hemorrhage in preterm neonates: a randomized control trial. J Matern Fetal Neonatal Med. 35:3633–3639. 2022.

44. Hübner ME, Ramirez R, Burgos J, Dominguez A, Tapia JL. Mode of delivery and antenatal steroids and their association with survival and severe intraventricular hemorrhage in very low birth weight infants. J Perinatol. 36:832–836. 2016.

45. Humberg A, Härtel C, Paul P, Hanke K, Bossung V, Hartz A, et al. Delivery mode and intraventricular hemorrhage risk in very-low-birth-weight infants: observational data of the German Neonatal Network. Eur J Obstet Gynecol Reprod Biol. 212:144–149. 2017.

46. Ikeda T, Ito Y, Mikami R, Matsuo K, Kawamura N, Yamoto A, et al. Fluctuations in internal cerebral vein and central side veins of preterm infants. Pediatr Int. 63:1319–1326. 2021.

47. Jelin AC, Zlatnik MG, Kuppermann M, Gregorich SE, Nakagawa S, Clyman R. Clamp late and maintain perfusion (CLAMP) policy: delayed cord clamping in preterm infants. J Matern Fetal Neonatal Med. 29:1705–1709. 2016.

48. Jim WT, Chiu NC, Chen MR, Hung HY, Kao HA, Hsu CH, et al. Cerebral hemodynamic change and intraventricular hemorrhage in very low birth weight infants with patent ductus arteriosus. Ultrasound Med Biol. 31:197–202. 2005.

49. Kaiser JR, Gauss CH, Pont MM, Williams DK. Hypercapnia during the first 3 days of life is associated with severe intraventricular hemorrhage in very low birth weight infants. J Perinatol. 26:279–285. 2006.

50. Kalani M, Shariat M, Khalesi N, Farahani Z, Ahmadi S. A comparison of early ibuprofen and indomethacin administration to prevent intraventricular hemorrhage among preterm infants. Acta Med Iran. 54:788–792. 2016.
51. Karagol BS, Calisici E, Zeybek C, Unay B, Yuksel S. The impact of initial hematocrit values after birth on peri-/intraventricular hemorrhage in extremely low birth weight neonates. Childs Nerv Syst. 38:109–114. 2022.

52. Katheria AC, Szychowski JM, Essers J, Mendler MR, Dempsey EM, Schmolzer GM, et al. Early cardiac and cerebral hemodynamics with umbilical cord milking compared with delayed cord clamping in infants born preterm. J Pediatr. 223:51–56.e1. 2020.

53. Kc A, Malqvist M, Rana N, Ranneberg LJ, Andersson O. Effect of timing of umbilical cord clamping on anaemia at 8 and 12 months and later neurodevelopment in late pre-term and term infants; a facility-based, randomized-controlled trial in Nepal. BMC Pediatr. 16:35. 2016.

54. Khanafer-Larocque I, Soraisham A, Stritzke A, Al Awad E, Thomas S, Murthy P, et al. Intraventricular hemorrhage: risk factors and association with patent ductus arteriosus treatment in extremely preterm neonates. Front Pediatr. 7:408. 2019.

55. Klingenberg C, Wheeler KI, McCallion N, Morley CJ, Davis PG. Volume-targeted versus pressure-limited ventilation in neonates. Cochrane Database Syst Rev. 10:CD003666. 2017.

56. Kluckow M. The pathophysiology of low systemic blood flow in the preterm infant. Front Pediatr. 6:29. 2018.

57. Kluckow M, Evans N. Low superior vena cava flow and intraventricular haemorrhage in preterm infants. Arch Dis Child Fetal Neonatal Ed. 82:F188–194. 2000.

58. Kochan M, Leonardi B, Firestine A, McPadden J, Cobb D, Shah TA, et al. Elevated midline head positioning of extremely low birth weight infants: effects on cardiopulmonary function and the incidence of periventricular-intraventricular hemorrhage. J Perinatol. 39:54–62. 2019.

59. Kooi EMW, Richter AE. Cerebral autoregulation in sick infants: current insights. Clin Perinatol. 47:449–467. 2020.
60. Kooi EMW, Verhagen EA, Elting JWJ, Czosnyka M, Austin T, Wong FY, et al. Measuring cerebrovascular autoregulation in preterm infants using near-infrared spectroscopy: an overview of the literature. Expert Rev Neurother. 17:801–818. 2017.

61. Kumar Nair PA, Pai MG, Gazal HA, Da Costa DE, Al Khusaiby SM. Indomethacin prophylaxis for intraventricular hemorrhage in very low birth weight babies. Indian Pediatr. 41:551–558. 2004.
62. Leviton A, Fenton T, Kuban KC, Pagano M. Labor and delivery characteristics and the risk of germinal matrix hemorrhage in low birth weight infants. J Child Neurol. 6:35–40. 1991.

63. Lightburn MH, Gauss CH, Williams DK, Kaiser JR. Cerebral blood flow velocities in extremely low birth weight infants with hypotension and infants with normal blood pressure. J Pediatr. 154:824–828. 2009.

64. Ljustina S, Berisavac M, Kovacević-Vukolić L, VelickovićAleksić V, Marković N. Analysis of intracranial hemorrhage grade in preterm singleton pregnancies delivered vaginally or by cesarean section. Vojnosanit Pregl. 70:255–258. 2013.

65. Lou HC, Lassen NA, Friis-Hansen B. Impaired autoregulation of cerebral blood flow in the distressed newborn infant. J Pediatr. 94:118–121. 1979.

66. Luca A, Vinturache A, Ilea C, Avasiloaiei A, Paduraru L, Carauleanu A, et al. Birth trauma in preterm spontaneous vaginal and cesarean section deliveries: a 10-years retrospective study. PLoS One. 17:e0275726. 2022.

67. Martini S, Czosnyka M, Smielewski P, Iommi M, Galletti S, Vitali F, et al. Clinical determinants of cerebrovascular reactivity in very preterm infants during the transitional period. Pediatr Res. 92:135–141. 2022.

68. McGoldrick E, Stewart F, Parker R, Dalziel SR. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst Rev. 12:CD004454. 2020.

69. McLendon D, Check J, Carteaux P, Michael L, Moehring J, Secrest JW, et al. Implementation of potentially better practices for the prevention of brain hemorrhage and ischemic brain injury in very low birth weight infants. Pediatrics. 111(4 Pt 2):e497–503. 2003.

70. Ment LR, Oh W, Ehrenkranz RA, Philip AG, Duncan CC, Makuch RW. Antenatal steroids, delivery mode, and intraventricular hemorrhage in preterm infants. Am J Obstet Gynecol. 172:795–800. 1995.

71. Ment LR, Oh W, Ehrenkranz RA, Philip AG, Vohr B, Allan W, et al. Lowdose indomethacin and prevention of intraventricular hemorrhage: a multicenter randomized trial. Pediatrics. 93:543–550. 1994.

72. Miller CJ, Prusakov P, Magers J, Speaks S, Sacic H, Escobar K, et al. Effects of prophylactic indomethacin on intraventricular hemorrhage and adverse outcomes in neonatal intensive care unit. J Perinatol. 42:1644–1648. 2022.

73. Mitra S, Florez ID, Tamayo ME, Mbuagbaw L, Vanniyasingam T, Veroniki AA, et al. Association of placebo, indomethacin, ibuprofen, and acetaminophen with closure of hemodynamically significant patent ductus arteriosus in preterm infants: a systematic review and meta-analysis. JAMA. 319:1221–1238. 2018.

74. Mittendorf R, Besinger R, Santillan M, Gianopoulos J. When used in the circumstance of preterm labor, is there a paradoxical effect of varying exposures to magnesium sulfate (MgSO4) on the developing human brain? Am J Obstet Gynecol. 193(6 Suppl):S65. 2005.

75. Mittendorf R, Dambrosia J, Dammann O, Pryde PG, Lee KS, Ben-Ami TE, et al. Association between maternal serum ionized magnesium levels at delivery and neonatal intraventricular hemorrhage. J Pediatr. 140:540–546. 2002.

76. Moriette G, Paris-Llado J, Walti H, Escande B, Magny JF, Cambonie G, et al. Prospective randomized multicenter comparison of high-frequency oscillatory ventilation and conventional ventilation in preterm infants of less than 30 weeks with respiratory distress syndrome. Pediatrics. 107:363–372. 2001.

77. Mullaart RA, Hopman JC, Rotteveel JJ, Stoelinga GB, De Haan AF, Daniels O. Cerebral blood flow velocity and pulsation in neonatal respiratory distress syndrome and periventricular hemorrhage. Pediatr Neurol. 16:118–125. 1997.

78. Noone MA, Sellwood M, Meek JH, Wyatt JS. Postnatal adaptation of cerebral blood flow using near infrared spectroscopy in extremely preterm infants undergoing high-frequency oscillatory ventilation. Acta Paediatr. 92:1079–1084. 2003.

79. Noori S, Seri I. Hemodynamic antecedents of peri/intraventricular hemorrhage in very preterm neonates. Semin Fetal Neonatal Med. 20:232–237. 2015.

80. Ohlsson A, Shah PS. Paracetamol (acetaminophen) for patent ductus arteriosus in preterm or low birth weight infants. Cochrane Database Syst Rev. 4:CD010061. 2018.

81. Okulu E, Haskologlu S, Guloglu D, Kostekci E, Erdeve O, Atasay B, et al. Effects of umbilical cord management strategies on stem cell transfusion, delivery room adaptation, and cerebral oxygenation in term and late preterm infants. Front Pediatr. 10:838444. 2022.
82. Pan I, Shah PA, Singh J, Kelly KN, Bondi DS. Comparison of neonatal outcomes with and without prophylaxis with indomethacin in premature neonates. J Pediatr Pharmacol Ther. 26:478–483. 2021.

83. Panerai RB, Kelsall AW, Rennie JM, Evans DH. Cerebral autoregulation dynamics in premature newborns. Stroke. 26:74–80. 1995.

84. Parker LA. Part 1: early recognition and treatment of birth trauma: injuries to the head and face. Adv Neonatal Care. 5:288–297. quiz 298-300. 2005.
85. Persad N, Kelly E, Amaral N, Neish A, Cheng C, Fan CS, et al. Impact of a "Brain Protection Bundle" in reducing severe intraventricular hemorrhage in preterm infants <30 weeks GA: a retrospective single centre study. Children (Basel). 8:983. 2021.

86. Pinto Cardoso G, Houivet E, Marchand-Martin L, Kayem G, Sentilhes L, Ancel PY, et al. Association of intraventricular hemorrhage and death with tocolytic exposure in preterm infants. JAMA Netw Open. 1:e182355. 2018.

87. Pishva N, Parsa G, Saki F, Saki M, Saki MR. Intraventricular hemorrhage in premature infants and its association with pneumothorax. Acta Med Iran. 50:473–476. 2012.
88. Piteaud I, Abdennour L, Icke C, Stany I, Lescot T, Puybasset L. Superior vena cava syndrome: cause of secondary raise of intracranial pressure after traumatic brain injury. Ann Fr Anesth Reanim. 27:850–853. 2008.
89. Poryo M, Boeckh JC, Gortner L, Zemlin M, Duppre P, Ebrahimi-Fakhari D, et al. Ante-, peri- and postnatal factors associated with intraventricular hemorrhage in very premature infants. Early Hum Dev. 116:1–8. 2018.

90. Rahman S, Ullah M, Ali A, Afridi N, Bashir H, Amjad Z, et al. Fetal outcomes in preterm cesarean sections. Cureus. 14:e27607. 2022.

91. Raybaud C. Normal and abnormal embryology and development of the intracranial vascular system. Neurosurg Clin N Am. 21:399–426. 2010.

92. Rhee CJ, Fraser CD 3rd, Kibler K, Easley RB, Andropoulos DB, Czosnyka M, et al. The ontogeny of cerebrovascular pressure autoregulation in premature infants. J Perinatol. 34:926–931. 2014.

93. Riskin A, Riskin-Mashiah S, Bader D, Kugelman A, Lerner-Geva L, Boyko V, et al. Delivery mode and severe intraventricular hemorrhage in single, very low birth weight, vertex infants. Obstet Gynecol. 112:21–28. 2008.

94. Rozé JC, Cambonie G, Marchand-Martin L, Gournay V, Durrmeyer X, Durox M, et al. Association between early screening for patent ductus arteriosus and in-hospital mortality among extremely preterm infants. JAMA. 313:2441–2448. 2015.

95. Sankar MN, Bhombal S, Benitz WE. PDA: to treat or not to treat. Congenit Heart Dis. 14:46–51. 2019.

96. Schreiner C, Suter S, Watzka M, Hertfelder HJ, Schreiner F, Oldenburg J, et al. Genetic variants of the vitamin K dependent coagulation system and intraventricular hemorrhage in preterm infants. BMC Pediatr. 14:219. 2014.

97. Schulz G, Keller E, Haensse D, Arlettaz R, Bucher HU, Fauchere JC. Slow blood sampling from an umbilical artery catheter prevents a decrease in cerebral oxygenation in the preterm newborn. Pediatrics. 111:e73–76. 2003.

98. Semberova J, Sirc J, Miletin J, Kucera J, Berka I, Sebkova S, et al. Spontaneous closure of patent ductus arteriosus in infants ≤1500 g. Pediatrics. 140:e20164258. 2017.

99. Seri I. Management of hypotension and low systemic blood flow in the very low birth weight neonate during the first postnatal week. J Perinatol 26 Suppl. 1:S8–S13. discussion S22-S23. 2006.

100. Shah V, Hodgson K, Seshia M, Dunn M, Schmolzer GM. Golden hour management practices for infants <32 weeks gestational age in Canada. Paediatr Child Health. 23:e70–e76. 2018.

101. Soul JS, Hammer PE, Tsuji M, Saul JP, Bassan H, Limperopoulos C, et al. Fluctuating pressure-passivity is common in the cerebral circulation of sick premature infants. Pediatr Res. 61:467–473. 2007.

102. Su BH, Lin HY, Chiu HY, Tsai ML, Chen YT, Lu IC. Therapeutic strategy of patent ductus arteriosus in extremely preterm infants. Pediatr Neonatol. 61:133–141. 2020.

103. Szpecht D, Al-Saad SR, Karbowski LM, Kosik K, Kurzawińska G, Szymankiewicz M, et al. Role of fibronectin-1 polymorphism genes with the pathogenesis of intraventricular hemorrhage in preterm infants. Childs Nerv Syst. 36:1729–1736. 2020.

104. Szpecht D, Gadzinowski J, Seremak-Mrozikiewicz A, Kurzawińska G, Drews K, Szymankiewicz M. The significance of polymorphisms in genes encoding Il-1β, Il-6, TNFα, and Il-1RN in the pathogenesis of intraventricular hemorrhage in preterm infants. Childs Nerv Syst. 33:1905–1916. 2017.

105. Szpecht D, Gadzinowski J, Seremak-Mrozikiewicz A, Kurzawińska G, Szymankiewicz M. Role of endothelial nitric oxide synthase and endothelin-1 polymorphism genes with the pathogenesis of intraventricular hemorrhage in preterm infants. Sci Rep. 7:42541. 2017.

106. Takahashi Y, Harada K, Kishkurno S, Arai H, Ishida A, Takada G. Postnatal left ventricular contractility in very low birth weight infants. Pediatr Cardiol. 18:112–117. 1997.

107. Thewissen L, Naulaers G, Hendrikx D, Caicedo A, Barrington K, Boylan G, et al. Cerebral oxygen saturation and autoregulation during hypotension in extremely preterm infants. Pediatr Res. 90:373–380. 2021.

108. Thornburg CD, Erickson SW, Page GP, Clark EAS, DeAngelis MM, Hartnett ME, et al. Genetic predictors of severe intraventricular hemorrhage in extremely low-birthweight infants. J Perinatol. 41:286–294. 2021.

109. Toledo JD, Rodilla S, Perez-Iranzo A, Delgado A, Maazouzi Y, Vento M. Umbilical cord milking reduces the risk of intraventricular hemorrhage in preterm infants born before 32 weeks of gestation. J Perinatol. 39:547–553. 2019.

110. Tomotaki S, Iwanaga K, Hanaoka S, Tomotaki H, Matsukura T, Niwa F, et al. Antenatal glucocorticoids reduce the incidence of refractory hypotension in low birthweight infants during the early neonatal period, but do not affect it beyond this time. Am J Perinatol. 38:1057–1061. 2021.

111. Toyoshima K, Kawataki M, Ohyama M, Shibasaki J, Yamaguchi N, Hoshino R, et al. Tailor-made circulatory management based on the stress-velocity relationship in preterm infants. J Formos Med Assoc. 112:510–517. 2013.

112. Tsuji M, Saul JP, du Plessis A, Eichenwald E, Sobh J, Crocker R, et al. Cerebral intravascular oxygenation correlates with mean arterial pressure in critically ill premature infants. Pediatrics. 106:625–632. 2000.

113. van Beek PE, Groenendaal F, Broeders L, Dijk PH, Dijkman KP, van den Dungen FAM, et al. Survival and causes of death in extremely preterm infants in the Netherlands. Arch Dis Child Fetal Neonatal Ed. 106:251–257. 2021.

114. van Bel F, Mintzer JP. Monitoring cerebral oxygenation of the immature brain: a neuroprotective strategy? Pediatr Res. 84:159–164. 2018.

115. van der Laan ME, Roofthooft MT, Fries MW, Berger RM, Schat TE, van Zoonen AG, et al. A hemodynamically significant patent ductus arteriosus does not affect cerebral or renal tissue oxygenation in preterm infants. Neonatology. 110:141–147. 2016.

116. Verma PK, Panerai RB, Rennie JM, Evans DH. Grading of cerebral autoregulation in preterm and term neonates. Pediatr Neurol. 23:236–242. 2000.

117. Vesoulis ZA, Flower AA, Zanelli S, Rambhia A, Abubakar M, Whitehead HV, et al. Blood pressure extremes and severe IVH in preterm infants. Pediatr Res. 87:69–73. 2020.

118. Vesoulis ZA, Liao SM, Mathur AM. Gestational age-dependent relationship between cerebral oxygen extraction and blood pressure. Pediatr Res. 82:934–939. 2017.

119. Vinukonda G, Dummula K, Malik S, Hu F, Thompson CI, Csiszar A, et al. Effect of prenatal glucocorticoids on cerebral vasculature of the developing brain. Stroke. 41:1766–1773. 2010.

120. Wardle SP, Yoxall CW, Weindling AM. Determinants of cerebral fractional oxygen extraction using near infrared spectroscopy in preterm neonates. J Cereb Blood Flow Metab. 20:272–279. 2000.

121. Wei JC, Catalano R, Profit J, Gould JB, Lee HC. Impact of antenatal steroids on intraventricular hemorrhage in very-low-birth weight infants. J Perinatol. 36:352–356. 2016.

122. Wolf HT, Weber T, Schmidt S, Norman M, Varendi H, Piedvache A, et al. Mode of delivery and adverse short- and long-term outcomes in vertex-presenting very preterm born infants: a European populationbased prospective cohort study. J Perinat Med. 49:923–931. 2021.

123. Wong FY, Leung TS, Austin T, Wilkinson M, Meek JH, Wyatt JS, et al. Impaired autoregulation in preterm infants identified by using spatially resolved spectroscopy. Pediatrics. 121:e604–e611. 2008.

124. Xu H, Hu F, Sado Y, Ninomiya Y, Borza DB, Ungvari Z, et al. Maturational changes in laminin, fibronectin, collagen IV, and perlecan in germinal matrix, cortex, and white matter and effect of betamethasone. J Neurosci Res. 86:1482–1500. 2008.

125. Yao SL, Smit E, Odd D. The effectiveness of interventions to prevent intraventricular haemorrhage in premature infants: a systematic review and network meta-analysis. J Neonatal Perinatal Med. 16:5–20. 2023.

126. Committee Opinion No. 455. magnesium sulfate before anticipated preterm birth for neuroprotection. Obstet Gynecol. 115:669–671. 2010.
127. Committee Opinion No 652. magnesium sulfate use in obstetrics. Obstet Gynecol. 127:e52–e53. 2016.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download