INTRODUCTION
MATERIALS AND METHODS
Participants
Basic attribute
Hand grip strength
Pulmonary function test
Cough ability and measurement condition
Statistical analysis
RESULTS
Participants characteristics
Table 1.
Values are presented as number (%) or mean±standard deviation.
OPD, obstructive pulmonary disease; RPD, restrictive pulmonary disease; BMI, body mass index; BSA, body surface area; FVC, forced vital capacity; %, percent predicted; FEV1, forced expiratory volume in one second; FEV1/FVC, FEV1 as a percentage of FVC; PEF, peak expiratory flow; V75, FEV in 75%; V50, FEV in 50%; V25, FEV in 25%.
Pulmonary function test
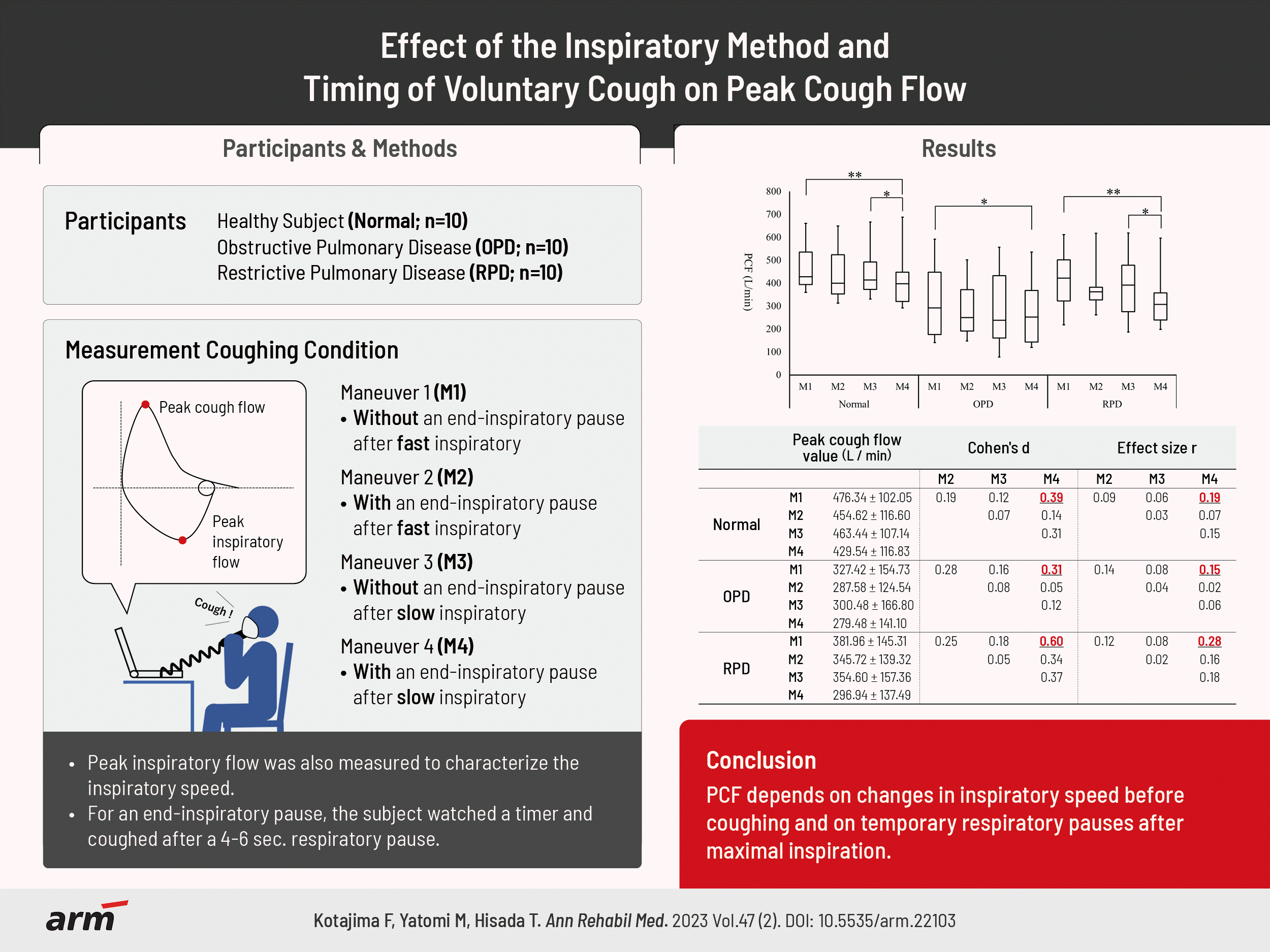
Comparison of the mean peak cough flow between the four coughing conditions
Normal
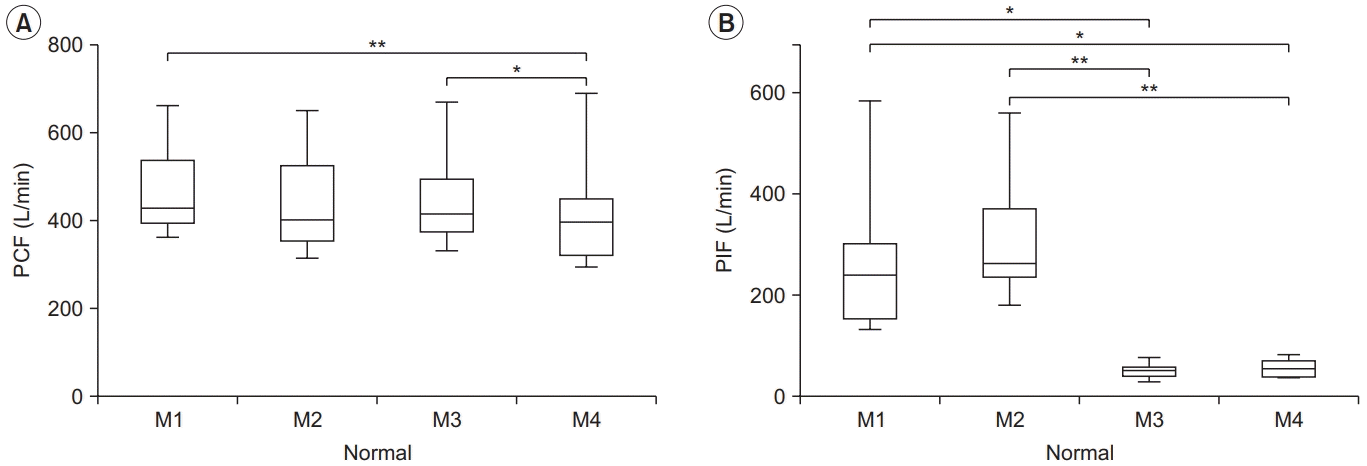 | Fig. 1.Peak cough flow (PCF) (A) and peak inspiratory flow (PIF) (B) for each condition in the normal group. PCF differed significantly between M1–M4 and M3–M4, and PIF differed between M1–M3, M1–M4, M2–M3, and M2–M4. The lower and upper margins of the box represent the lower quartile (Q1) and upper quartile (Q3) of the total score, respectively. The band inside the box indicates the median, and the whiskers range from Q1+1.5 interquartile range (IQR) to Q3+1.5 IQR. The horizontal lines above the box graph depict significant differences using Bonferroni correction (*p<0.05, **p<0.01). M, maneuver. |
Table 2.
| Coughing M | PCF value (L/min) |
p-valuea) |
Cohen’s d |
Effect size r |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| M2 | M3 | M4 | M2 | M3 | M4 | M2 | M3 | M4 | ||
| Normal | ||||||||||
| M1 | 476.34±102.05 | 0.121 | <0.999 | 0.002** | 0.19 | 0.12 | 0.39 | 0.09 | 0.06 | 0.19 |
| M2 | 454.62±116.60 | - | 0.532 | 0.755 | - | 0.07 | 0.14 | - | 0.03 | 0.07 |
| M3 | 463.44±107.14 | - | - | 0.015* | - | - | 0.31 | - | - | 0.15 |
| M4 | 429.54±116.83 | - | - | - | - | - | - | - | - | - |
| OPD | ||||||||||
| M1 | 327.42±154.73 | 0.074 | 0.485 | 0.020* | 0.28 | 0.16 | 0.31 | 0.14 | 0.08 | 0.15 |
| M2 | 287.58±124.54 | - | <0.999 | <0.999 | - | 0.08 | 0.05 | - | 0.04 | 0.02 |
| M3 | 300.48±166.80 | - | - | <0.999 | - | - | 0.12 | - | - | 0.06 |
| M4 | 279.48±141.10 | - | - | - | - | - | - | - | - | - |
| RPD | ||||||||||
| M1 | 381.96±145.31 | 0.582 | 0.325 | 0.002** | 0.25 | 0.18 | 0.60 | 0.12 | 0.08 | 0.28 |
| M2 | 345.72±139.32 | - | <0.999 | 0.093 | - | 0.05 | 0.34 | - | 0.02 | 0.16 |
| M3 | 354.60±157.36 | - | - | 0.047* | - | - | 0.37 | - | - | 0.18 |
| M4 | 296.94±137.49 | - | - | - | - | - | - | - | - | - |
Obstructive pulmonary disease
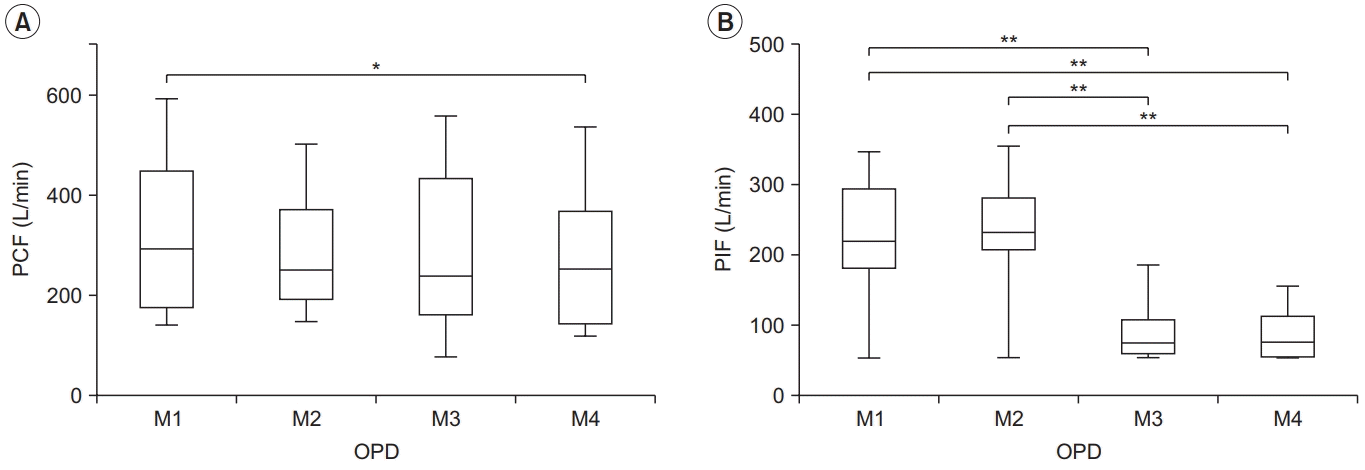 | Fig. 2.Peak cough flow (PCF) (A) and peak inspiratory flow (PIF) (B) for each condition in the obstructive pulmonary disease (OPD) group. PCF differed significantly between M1–M4, and PIF differed between M1–M3, M1–M4, M2–M3, and M2–M4. The lower and upper margins of the box represent the lower quartile (Q1) and upper quartile (Q3) of the total score, respectively. The band inside the box indicates the median, and the whiskers range from Q1+1.5 interquartile range (IQR) to Q3+1.5 IQR. The horizontal lines above the box graph depict significant differences using Bonferroni correction (*p<0.05, **p<0.01). M, maneuver. |
Restrictive pulmonary disease
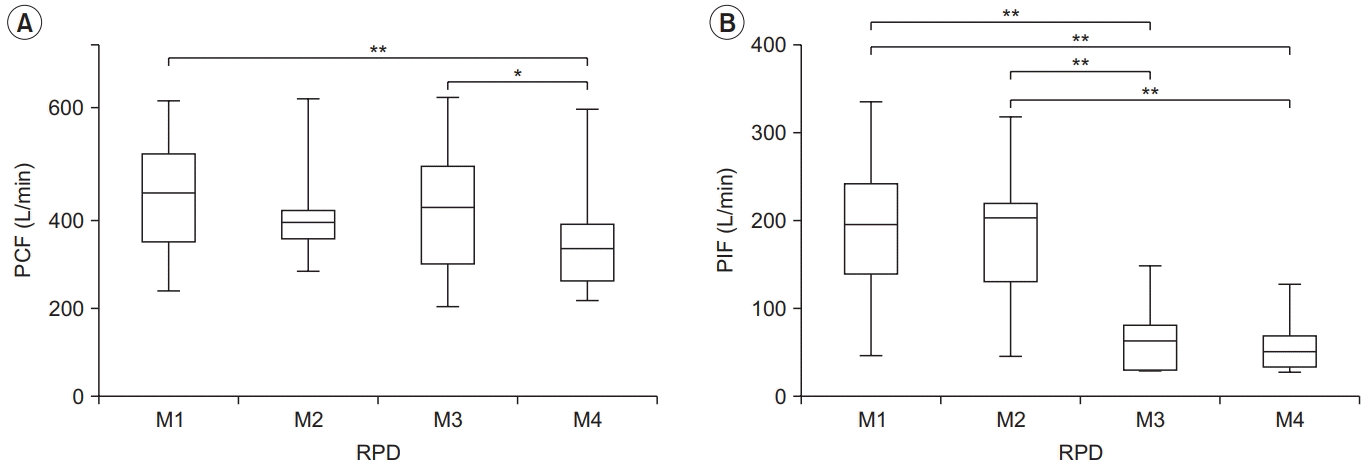 | Fig. 3.Peak cough flow (PCF) (A) and peak inspiratory flow (PIF) (B) for each condition in the restrictive pulmonary disease (RPD) group. PCF differed significantly between M1–M4 and M3–M4, and PIF differed between M1–M3, M1– M4, M2–M3, and M2–M4. The lower and upper margins of the box represent the lower quartile (Q1) and upper quartile (Q3) of the total score, respectively. The band inside the box indicates the median, and the whiskers range from Q1+1.5 interquartile range (IQR) to Q3+1.5 IQR. The horizontal lines above the box graph depict significant differences using Bonferroni correction (*p<0.05, **p<0.01). M, maneuver. |
Comparison of the mean peak inspiratory flow between the four coughing conditions
Table 3.
| Coughing M | PIF value (L/min) |
p-valuea) |
||
|---|---|---|---|---|
| M2 | M3 | M4 | ||
| Normal | ||||
| M1 | 264.42±169.66 | <0.999 | 0.031* | 0.026* |
| M2 | 305.64±109.73 | - | <0.001** | <0.001** |
| M3 | 50.28±15.35 | - | - | <0.999 |
| M4 | 51.54±23.27 | - | - | - |
| OPD | ||||
| M1 | 225.60±82.50 | <0.999 | 0.001** | 0.002** |
| M2 | 223.02±87.00 | - | 0.004** | 0.004** |
| M3 | 88.62±42.33 | - | - | <0.999 |
| M4 | 86.64±41.65 | - | - | - |
| RPD | ||||
| M1 | 185.82±86.90 | <0.999 | 0.004** | 0.006** |
| M2 | 182.46±82.95 | - | 0.006** | 0.006** |
| M3 | 66.84±37.83 | - | - | <0.999 |
| M4 | 57.78±28.60 | - | - | - |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download