1. Buiret G, Pavic M, Pignat JC, Pasquet F. Gelatin-thrombin matrix: a new and simple way to manage recurrent epistaxis in hematology units. Case Rep Otolaryngol. 2013; 2013:851270.

2. Reyre A, Michel J, Santini L, Dessi P, Vidal V, Bartoli JM, et al. Epistaxis: the role of arterial embolization. Diagn Interv Imaging. 2015; 96(7-8):757–73.

3. Tunkel DE, Anne S, Payne SC, Ishman SL, Rosenfeld RM, Abramson PJ, et al. Clinical practice guideline: nosebleed (epistaxis). Otolaryngol Head Neck Surg. 2020; 162(1_suppl):S1–38.

4. Yang X, Yi K, Tian J, Guo Y. [A meta-analysis compare rapid rhino with Merocel for nasal packing]. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2012; 26(14):655–60. Chinese.
5. Moumoulidis I, Draper MR, Patel H, Jani P, Price T. A prospective randomised controlled trial comparing Merocel and Rapid Rhino nasal tampons in the treatment of epistaxis. Eur Arch Otorhinolaryngol. 2006; 263(8):719–22.

6. Badran K, Malik TH, Belloso A, Timms MS. Randomized controlled trial comparing Merocel® and RapidRhino® packing in the management of anterior epistaxis. Clin Otolaryngol. 2005; 30(4):333–7.
7. Nikolaou G, Holzmann D, Soyka MB. Discomfort and costs in epistaxis treatment. Eur Arch Otorhinolaryngol. 2013; 270(8):2239–44.

8. Gudziol V, Mewes T, Mann WJ. Rapid Rhino: a new pneumatic nasal tamponade for posterior epistaxis. Otolaryngol Head Neck Surg. 2005; 132(1):152–5.

9. Pallin DJ, Chng YM, McKay MP, Emond JA, Pelletier AJ, Camargo CA Jr. Epidemiology of epistaxis in US emergency departments, 1992 to 2001. Ann Emerg Med. 2005; 46(1):77–81.

10. Denholm SW, Maynard CA, Watson HG. Warfarin and epistaxis--a case controlled study. J Laryngol Otol. 1993; 107(3):195–6.
11. Abrich V, Brozek A, Boyle TR, Chyou PH, Yale SH. Risk factors for recurrent spontaneous epistaxis. Mayo Clin Proc. 2014; 89(12):1636–43.

12. Liao Z, Guo J, Mi J, Liao W, Chen S, Huang Y, et al. Analysis of bleeding site to identify associated risk factors of intractable epistaxis. Ther Clin Risk Manag. 2021; 17:817–22.

13. Krulewitz NA, Fix ML. Epistaxis. Emerg Med Clin North Am. 2019; 37(1):29–39.

14. Byun H, Chung JH, Lee SH, Ryu J, Kim C, Shin JH. Association of hypertension with the risk and severity of epistaxis. JAMA Otolaryngol Head Neck Surg. 2020; 147(1):1–7.

15. Min HJ, Kang H, Choi GJ, Kim KS. Association between hypertension and epistaxis: systematic review and meta-analysis. Otolaryngol Head Neck Surg. 2017; 157(6):921–7.

16. Ando Y, Iimura J, Arai S, Arai C, Komori M, Tsuyumu M, et al. Risk factors for recurrent epistaxis: importance of initial treatment. Auris Nasus Larynx. 2014; 41(1):41–5.

17. Kallenbach M, Dittberner A, Boeger D, Buentzel J, Kaftan H, Hoffmann K, et al. Hospitalization for epistaxis: a population-based healthcare research study in Thuringia, Germany. Eur Arch Otorhinolaryngol. 2020; 277(6):1659–66.

18. Wang L, Wang X, Ba Y. Clinical analysis of delayed epistaxis following endoscopic sinus surgery. Am J Otolaryngol. 2022; 43(3):103406.

19. Seidel DU, Jacob L, Kostev K, Sesterhenn AM. Risk factors for epistaxis in patients followed in general practices in Germany. Rhinology. 2017; 55(4):312–8.

20. Schmelzer B, Katz S, Vidts G. Long-term efficacy of our surgical approach to turbinate hypertrophy. Am J Rhinol. 1999; 13(5):357–61.

21. Koskinen A, Myller J, Mattila P, Penttilä M, Silvola J, Alastalo I, et al. Long-term follow-up after ESS and balloon sinuplasty: comparison of symptom reduction and patient satisfaction. Acta Otolaryngol. 2016; 136(5):532–6.

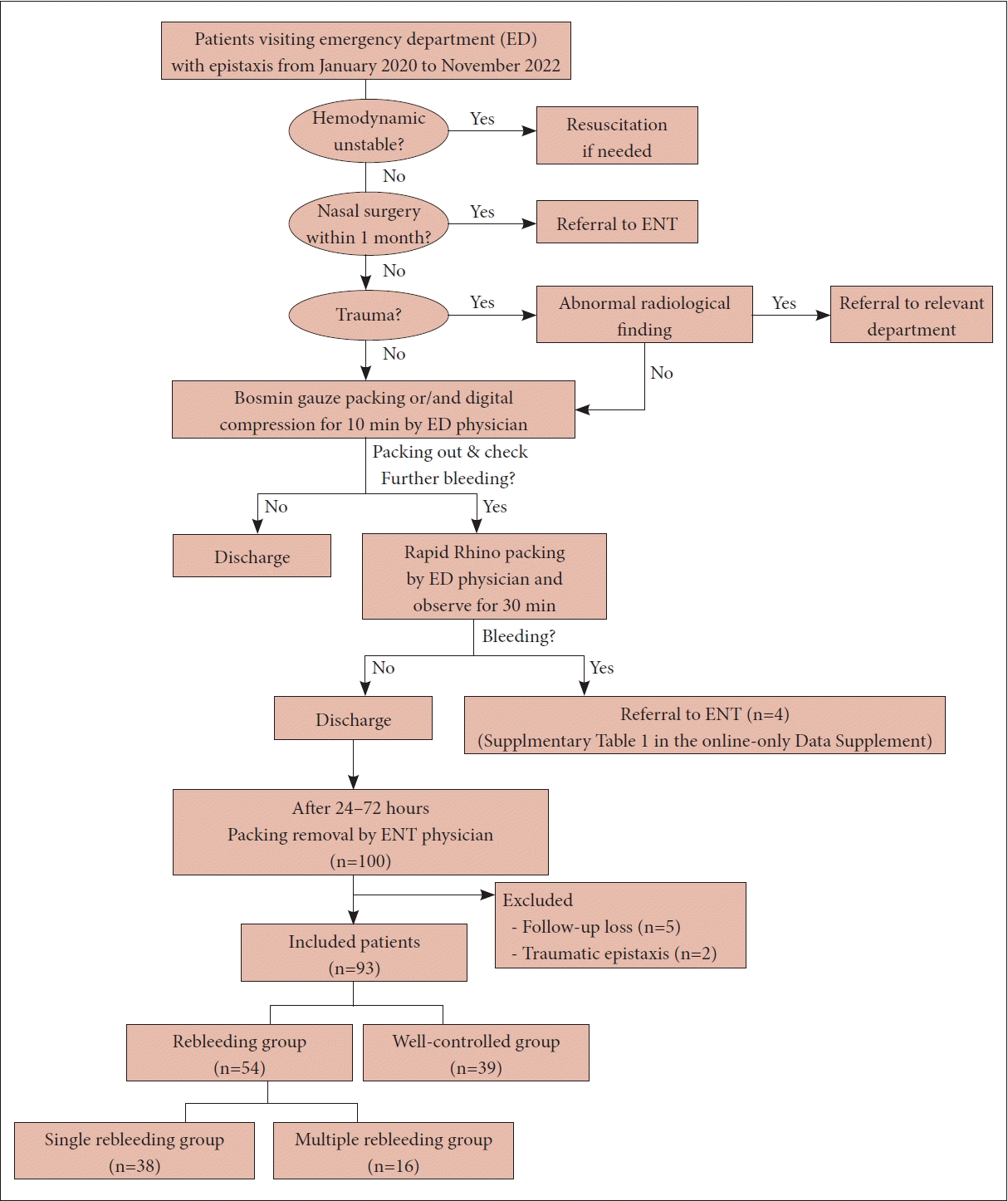
22. Kim SJ, Lee SJ, Go YJ, Park S, Bae JH. Characteristics and risk factors of recurrent epistaxis in geriatric patients. Korean J Otorhinolaryngol-Head Neck Surg. 2021; 64(8):548–53.

23. Mackeith S, Hettige R, Falzon A, Draper M. The relationship between pressure and volume when using Rapid Rhino(r) packs in the management of epistaxis. Rhinology. 2011; 49(4):470–3.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download