Abstract
Background/Aims
Methods
Results
ACKNOWLEDGEMENTS
Notes
Financial support: The study was supported by Mochida Pharmaceutical Co., Ltd. Mochida Pharmaceutical Co., Ltd. provided funding to support the provision of multimatrix mesalazine (Shire US Inc.) and time-dependent controlled-release mesalazine (Kyorin Pharmaceutical Co., Ltd.).
Conflict of interest: Haruhiko Ogata has received consulting, grant, or lecture fees from Mochida Pharmaceutical Co., Ltd., JIMRO, Takeda Pharmaceutical, Mitsubishi Tanabe Pharma, Kyorin Pharmaceutical, Otsuka Pharmaceutical, Astellas Pharma, Eisai, Zeria Pharmaceutical, AbbVie G.K., EA Pharma, and Boston Scientific Japan K.K. Akihiro Ohori has received lecture fees from Mitsubishi Tanabe Pharma and Zeria Pharmaceutical. Seiichi Mizushima and Atsushi Hagino are employees of Mochida Pharmaceutical Co., Ltd. Toshifumi Hibi is editor-in-chief of Intestinal Research and has received consulting, grant, lecture, or manuscript preparation fees from Mochida Pharmaceutical Co., Ltd., AbbVie G.K., EA Pharma, AstraZeneca K.K., JIMRO, Mitsubishi Tanabe Pharma, Eisai, Takeda Pharmaceutical, Zeria Pharmaceutical, Janssen Pharmaceutical K.K., Astellas Pharma, and Otsuka Pharmaceutical.
References
Fig. 1
Patient disposition. aMultiple options are allowed as reasons for discontinuation. FAS, full analysis set; PPS, per protocol set.
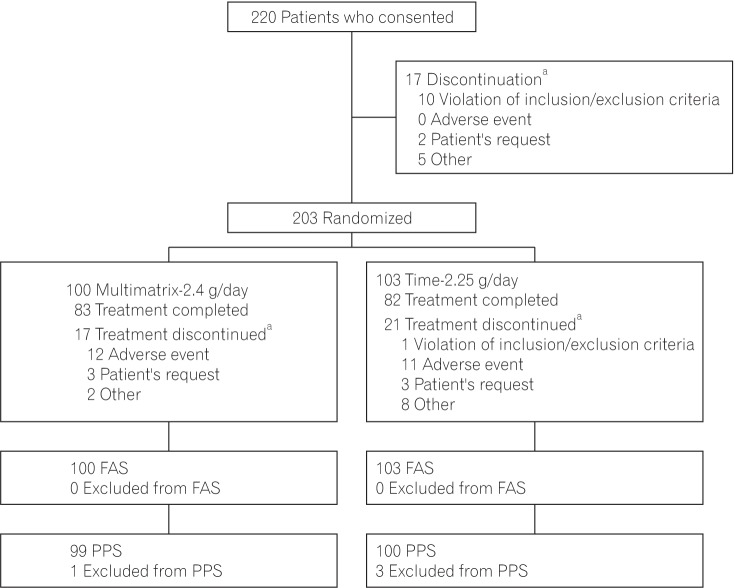
Fig. 2
The proportion of patients without rectal bleeding during the treatment period in the per protocol set. The difference between the multimatrix mesalazine 2.4 g/day group and the controlled-release mesalazine 2.25 g/day group was 6.8% (two-sided 95% CI, –3.9% to 17.6%).
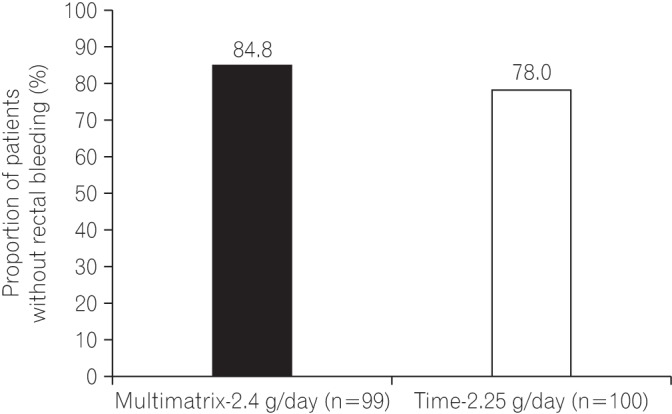
Fig. 3
The Kaplan-Meier plot of time to rectal bleeding (rectal bleeding score ≥1) in the per protocol set.
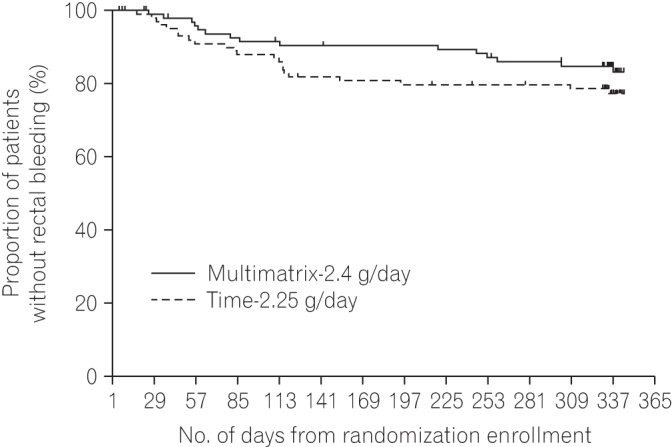
Fig. 4
The proportion of patients who relapsed (UC-Disease Activity Index score ≥3 and rectal bleeding score ≥1) at week 48 or at the discontinuation of treatment in the per protocol set. The difference between the multimatrix mesalazine 2.4 g/day group and the controlled-release mesalazine 2.25 g/day group was −4.8% (two-sided 95% CI, −15.6% to 5.9%) at week 48 and −4.9% (two-sided 95% CI, −14.9% to 5.2%) at the end of treatment period.
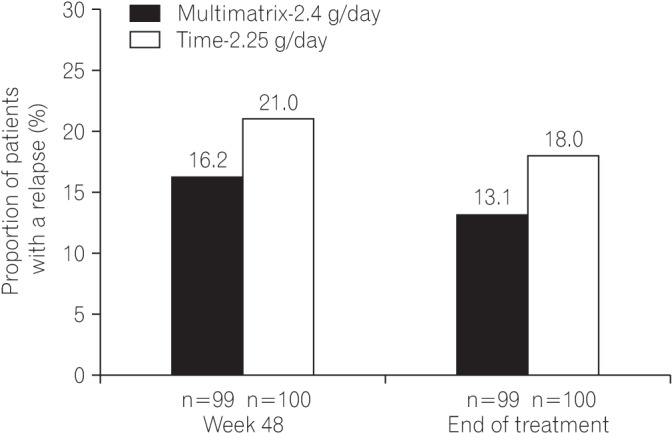
Table 1
UC-Disease Activity Index
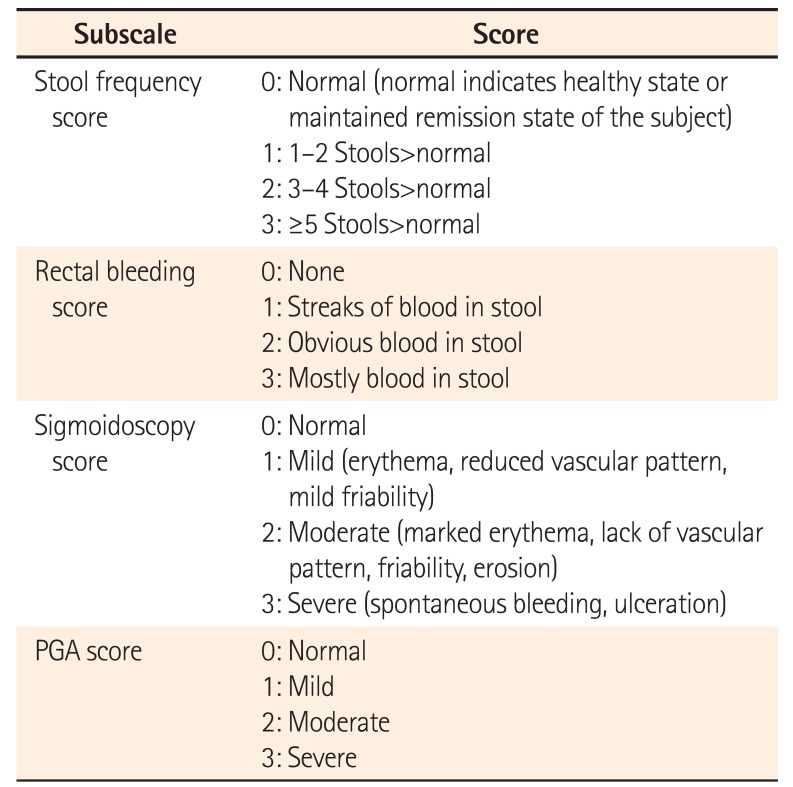
Table 2
Patient Demographics (Full Analysis Set)
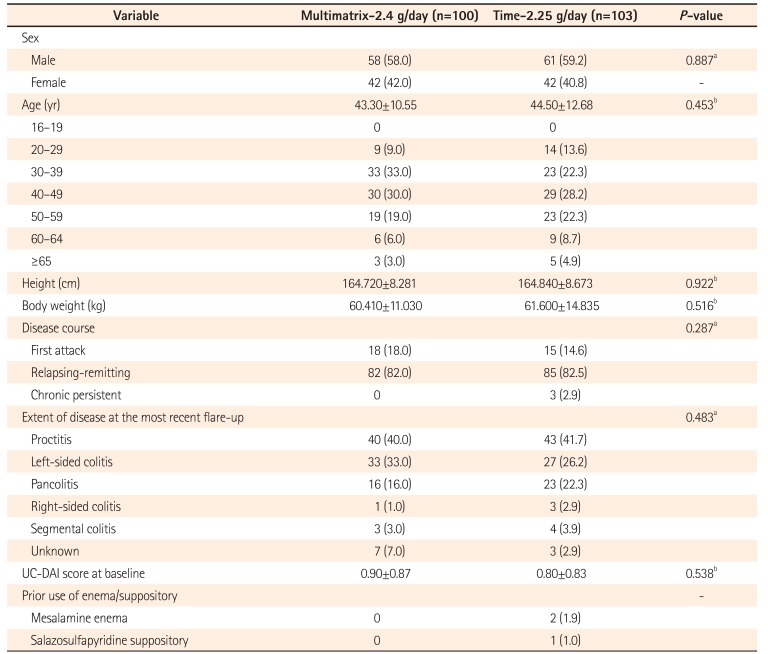
Table 3
Change in UC-DAI Score at End of Treatment (Per Protocol Set)
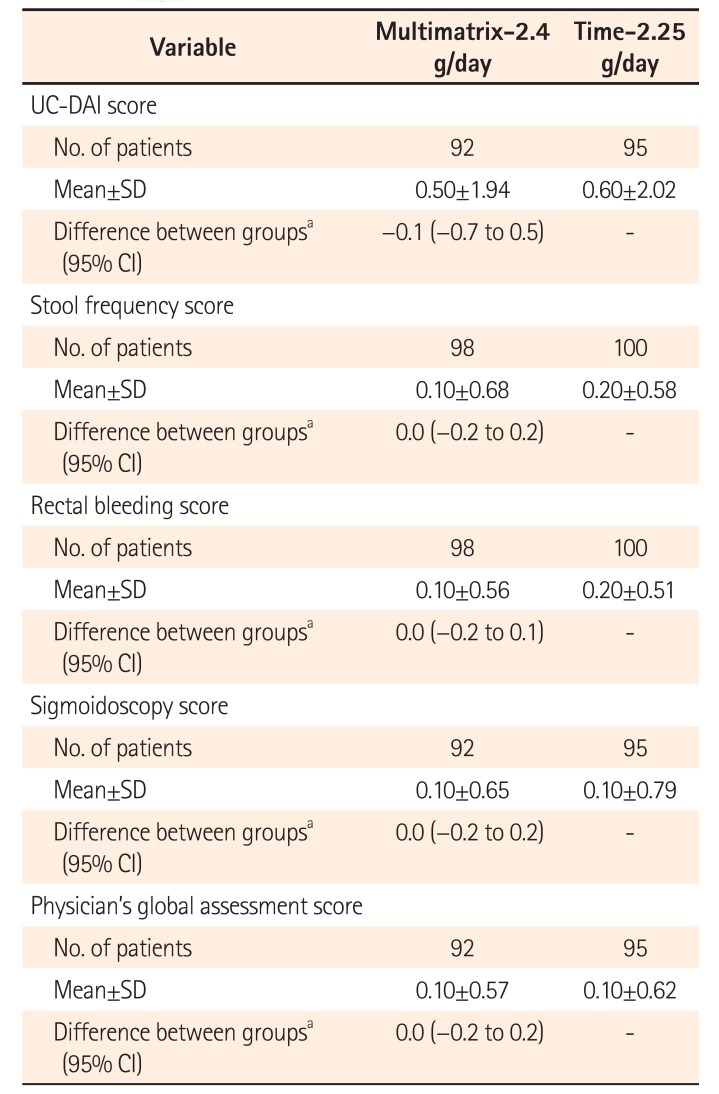
Table 4
Proportion of Patients without Rectal Bleeding by Subgroup (Per Protocol Set)
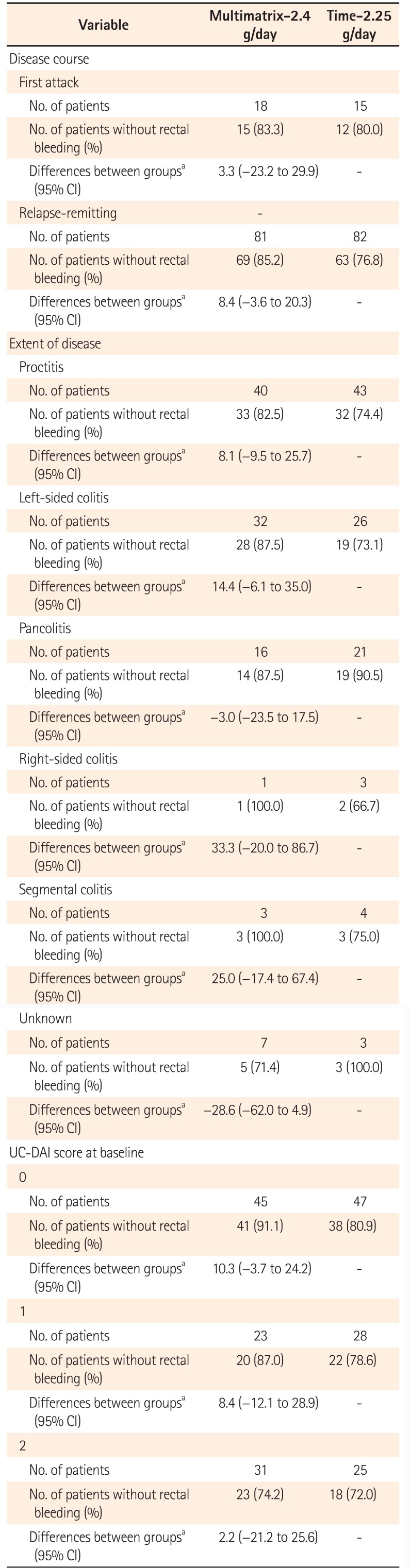
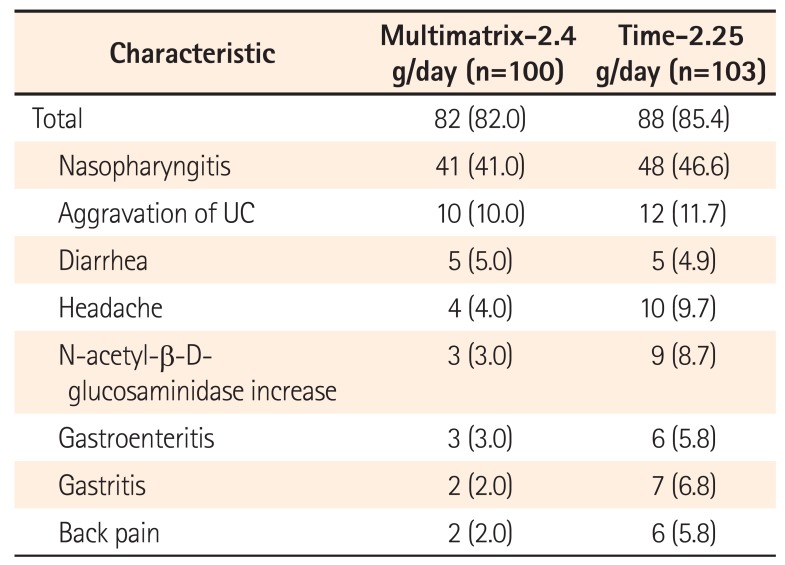
Table 5
Incidence of Adverse Events




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download