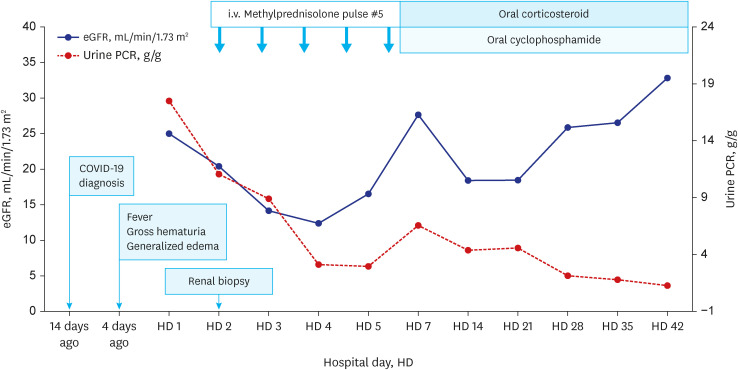
An 11-year-old boy presented to our hospital with gross hematuria, generalized edema, and fever of 3 days duration. He had a history of COVID-19 infection two weeks prior with a mild fever. He was not vaccinated against COVID-19 and had no other exhibiting symptoms of infection, such as upper respiratory tract infection or gastrointestinal symptoms. He had normal urinalysis results at a school medical check-up six months prior and had no underlying disease or family history. At the time of admission, the patient was febrile (body temperature, 39°C) and hypertensive at 139/89 mmHg (> 95th percentile). The patient’s laboratory test results upon admission were as follows: white blood cell count, 14,730/uL; erythrocyte sedimentation rate, 93 mm/h; C-reactive protein level, 6.9 mg/dL; blood urea nitrogen level, 39.4 mg/dL; serum creatinine level, 2.44 mg/dL; estimated glomerular filtration rate (eGFR, calculated by Schwartz bedside equation), 16.6 mL/min/1.73 m
2; sodium, 130 mmol/L; potassium, 5.4 mmol/L; total protein content, 6.2 g/dL; albumin level, 3.6 g/dL; calcium, 8.3 mg/dL; phosphorus, 5.7 mg/dL; uric acid level, 10.6 mg/dL; spot urine protein/creatinine ratio, 17.5 g/g; creatinine and 24-hour urine protein excretion, 7,124.7 mg/day. Complement levels were within the normal ranges and autoantibodies including antineutrophil cytoplasmic antibody and anti-glomerular basement membrane (GBM) antibody were all absent. Ultrasonography of the kidney revealed increased parenchymal echogenicity and swelling. His urine output was within normal range without oliguria (600–1,200 mL/day). On hospital day 2, considering the patient’s clinical presentation (
Fig. 1) of rapid and progressive acute kidney injury (AKI) accompanied by gross hematuria, proteinuria, and hypertension, methylprednisolone (MP) pulse therapy was initiated, and a renal biopsy was performed the next day. Renal biopsy specimens (
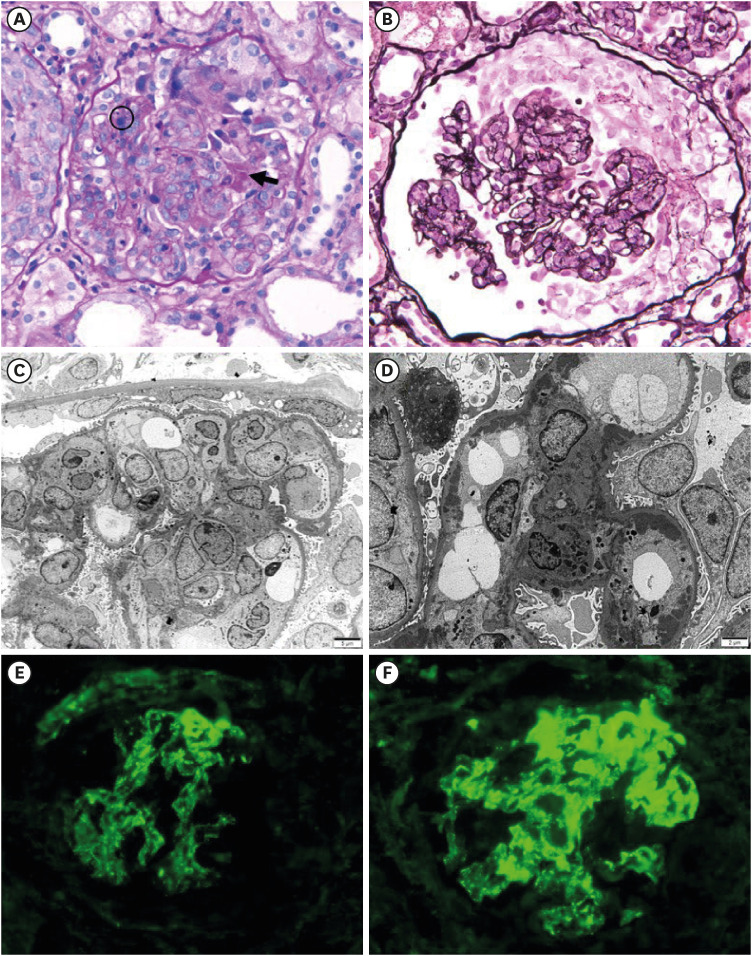
Fig. 2) revealed crescentic immune-complex glomerulonephritis. Nineteen out of 36 glomeruli (53%) showed crescentic formation (17 cellular, 2 fibrocellular) and there was no tubulointerstitial change on light microscopy. Immunofluorescence revealed diffuse mesangial immunoglobulin (Ig)G, IgA, C3, Kappa and Lambda deposit. Electron microscopy demonstrated profuse subendothelial and mesangial electron-dense deposits, numerous leukocytes in the capillary lumen (endocapillary proliferation), extensive foot process effacement with microvillous transformation, and intraluminal leukocytes, ruling out IgA nephropathy. Consequently, we diagnosed the patient with post-COVID-19 infection-associated de novo crescentic immune-mediated glomerulonephritis. After five treatments of MP pulse, oral prednisolone and cyclophosphamide treatment for 12 weeks were started. On hospital day 10, we started antihypertensive treatment with calcium channel blocker due to persistent hypertension. After antihypertensive treatment, his systolic blood pressure was maintained around 120 mmHg. Azathioprine was added as the maintenance therapy after the induction therapy of cyclophosphamide treatment for 12 weeks. After these medical treatments for 5 weeks, his renal function began to recover gradually. Currently, 20 weeks later, his level of serum creatinine was 0.87 mg/dL and an eGFR of 69.5 mL/min/1.73 m
2.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download