Abstract
Pelvic organ prolapse (POP) is a significant public health concern in women and a common cause of gynecological surgery in elderly women. The prevalence of POP has increased with an increase in the aging population. POP is usually diagnosed based on pelvic examination. However, an imaging study may be necessary for more accurate diagnosis. Translabial ultrasound (TLUS) was used to assess diverse types of POP, particularly posterior-compartment POP. It is beneficial to distinguish between true and false rectocele, and detect the rectocele as clinically apparent. TLUS can also establish whether the underlying cause is a problem of the rectovaginal septum, perineal hypermobility, or isolated enterocele. TLUS also plays a role in differentiating POP from conditions that mimic POP. It is a simple, inexpensive, and non-harmful diagnostic modality that is appropriate for most gynecologic clinics.
Pelvic organ prolapse (POP) is a significant public health concern in women and is a common indication for gynecological surgery [1–6]. POP is defined as “the descent of one or more aspects of the vagina and uterus: the anterior vaginal wall, posterior vaginal wall, the uterus (cervix), or vaginal vault”. POP causes a huge social and economic burden and adversely affects quality of life. The lifetime risk of POP surgery in women is estimated at 20% [1,7–9]. POP can cause various symptoms such as vaginal bulging, pelvic pressure, sexual dysfunction, lower urinary tract dysfunction, and defecatory dysfunction [2,10–13]. To treat these symptoms, doctors use surgical or non-surgical methods according to the patient’s preference or condition [2,12–16]. In the United States, the prevalence of symptomatic POP is expected to exceed 46% to become 4.9 million women by 2050 [1,14]. In addition, older women show a higher rate of POP, with a 30% risk factor; 180 per 100,000 women aged 50 years or older in South Korea suffer from POP [2,16].
POP was diagnosed based on pelvic examination [2,17]. The International Continence Society recommends using a pelvic organ prolapse quantification (POP-Q) system during pelvic examinations [2,10]. Quantitative measurement by the POP-Q system aids in understanding the affected site and the degree of prolapse. However, it does not identify the underlying supporting defects or differentiate the organs involved (e.g., rectocele vs. enterocele) [2,11]. Imaging studies can help to solve this problem. Since the beginning of the 21st century, magnetic resonance imaging (MRI) and translabial ultrasound (TLUS) dynamics with several adjustments have been used as adjunctive tools for diagnosing POP [2,12,13]. MRI is expensive and has a low availability. TLUS is an easy, cheap, and non-harmful diagnostic method that is appropriate in most gynecologic clinics with pre-existing ultrasonography machines, and involves no additional equipment purchase [2,18].
In this review, our goal was to discuss the clinical advantages of using TLUS to diagnose POP along with a literature review.
The authors of this review conducted an online literature search for studies conducted among humans from inception to March 2022. Two databases were included in this investigation: Google Scholar and PubMed. There were no language restrictions. The following words and subject terms were used in this search: “enterocele, pelvic organ prolapse, rectocele, and ultrasonography”.
The authors included all studies found, regardless of the nature of the study. The titles and abstracts were reviewed individually. If information was insufficient in the abstracts, the authors then retrieved and assessed the relevant full texts to determine their eligibility.
Recently, the use of TLUS in urogynecology, particularly for assessing the female pelvic floor, has progressed significantly, accompanied by the development of ultrasonographic technology with enormous potential to improve women’s health [18–22]. TLUS is an objective and reproducible technique to evaluate the female pelvic floor and significantly improves the comprehension of the relationship between female pelvic floor dysfunction and delivery [18–22].
The two-dimensional TLUS can assess the midline structure of the pelvic floor as presened in Fig. 1C [18,23]. The three-dimensional (3D) technique is essential for visualizing the plane of the pelvic floor muscles, including the levator ani muscle (LAM). The LAM is “the main support muscle of the pelvic organs that is fundamental for normal pelvic function”. Because LAM is set in the axial plane, it cannot be observed without 3D ultrasound [18,20,24,25].
To evaluate POP using TLUS, the patient must be in the semi-fowler position with the hips bent and abducted. Using a transabdominal probe, a 3.5–5 MHz convex array transducer can be implemented on the labium major after evacuation before the exam (Fig. 1A). Even after evacuation, some patients still have full bladders because of incomplete evacuation of the cystocele. Doctors should apply a large amount of jelly on both the transducer and probe cover so as not to interfere with the air of the vaginal sulcus. All TLUS measurements should be performed in the relaxing mode and during the maximal Valsalva maneuver. Rectocele or enterocele presence was defined as “the descent of either the rectal ampulla or the small bowel, sigmoid colon, or omentum into the cul-de-sac” (Fig. 1B). The peritoneal cul-de-sac loaded with echogenic small bowel and peristalsis was clear in cases of enterocele during the maximal Valsalva maneuver [22]. Even in patients with procidentia, there can be no rectocele or enterocele, with only posterior vaginal wall relief. Dietz et al. [23] reported that the descending of the cervix, urethra, bladder, and rectum during the Valsalva maneuver is simple to record in the midsagittal plane with a signal to a landmark determined by the inferoposterior margin of the symphysis pubis, with no need for the 3D technology [17,23]. However, in other cases, 3D ultrasound is likely to be helpful in identifying fascial defects such as transverse or lateral ruptures of the rectovaginal septum [17,25]. In addition, 3D datasets allow data storage for further analysis. 3D TLUS increases intra- and inter-observer reproducibility in assessing POP [18].
Dietz et al. [26] in 2005 demonstrated 3D TLUS as a reliable and reproducible technique for evaluating LAM biometry, and recognized the biometric parameters of the LAM and levator cavity. They also reported a considerable correlation between the levator cavity area and pelvic organ falling, which affirmed the previous hypothesis that LAM anatomy and integrity play an independent role in determining pelvic organ prolapse. LAM abnormalities are common in urogynecological patients, especially women of childbearing age who have at least one baby, and are correlated with falling of the anterior and apical parts [27]. It is also possible to have “tomographic imaging” with 3D TLUS by tomographic ultrasound imaging (TUI), which reproduces pelvic floor “slices” [18].
3D/4D TLUS is a fast-evolving area with considerable potential to improve women’s health and increase comprehension of the pathophysiology and etiology of female pelvic floor dysfunction [2,18]. Several studies have determined an average-to-almost excellent reproducibility (inter-observer reliability) for measuring levator hiatal dimensions with 3D/4D TLUS under relaxed and moving conditions [28,29]. van Veelen et al. [30] revealed that in women with or without a baby, the use of 3D/4D ultrasound imaging to measure the levator hiatus and levator-urethra gap is very reliable. They stated that the mechanism used to measure levator hiatal measures is simple, while the levator-urethra gap measurement using TUI is more complex [26,28–31]. Another study by van Delft et al. [32,33] compared 3D and 4D TLUS with endovaginal ultrasound and digital palpation to assess pelvic floor muscle contractility. They reported a high correlation among the three methods.
In terms of the role of TLUS as an adjunct to the POP-Q examination to provide an accurate diagnosis of POP, the standard POP-Q examination quantifies all parameters. Six points are placed on the vaginal wall. Then, the space between the hymen and these points is measured using the maximal Valsalva maneuver. The following points should be determined: “two posterior points (Ap and Bp), two anterior points (Aa and Ba), two apical points (C and D), genital hiatus, total vaginal length, and perineal body”. POP-Q 0 to the fourth stage is identified based on the area between the most dependent part of the pelvic organ and the hymenal ring as presented in Figs 2, 3 [2,34]. Although this is a widely used standardized method, it is subjective and cannot precisely identify which organ is in the prolapsed vaginal sac. In addition, several reports have confirmed false POP due to conditions mimicking POP [35–37].
In cases of real POP, particularly in posterior prolapse, it is difficult to determine the existence of rectocele or enterocele with posterior vaginal wall repose only by physical examination, even with POP-Q staging. In uterovaginal prolapse, most doctors anticipate that the rectum or, infrequently, the small bowel is in the sack of the posterior vaginal wall if the POP is completely prolapsed. However, on TLUS, they usually face only a reposed posterior vaginal wall composed of a pouch that does not involve an enterocele or rectocele [2]. Treating plans must be adjusted according to the anatomical state and the presence of rectocele or enterocele [17]. TLUS simplifies the differential diagnosis of a real rectocele or simple posterior vaginal repose. Some researchers have reported that for asymptomatic rectoceles, the combination of posterior reform and surgical reform of apical prolapse minimizes the risk of poor surgical outcomes. However, there is still a debate regarding this surgical plan [38,39]. The necessity for regular posterior reform should be re-evaluated after the differential diagnosis of a real rectocele or simple posterior vaginal repose to decrease the rate of avoidable posterior repair. In cases of POP with an enterocele, doctors should surgically repair the enterocele by excision or ligation of the enterocele pouch. Fixing the rectovaginal septum without enterocele reform may lead to the POP recurring [2].
Until now, TLUS was mainly used to detect “the ureterocele, cystocele, rectocele, and enterocele,” which could remain undiagnosed, or misdiagnosed, using the POP-Q method. In 2005, Dietz and Steensma [17] stated that TLUS could be used to measure POP linked with the clinical staging of the POP-Q method. The researchers indicated that TLUS could identify POP using a reference line comprising the symphysis pubis inferior margin [2]. Greater descent of the uterus, cul-de-sac, bladder, and rectal ampulla during the Valsalva maneuver was computed [40]. Dietz et al. [23] reported an excellent relationship between ultrasound results and clinical staging of anterior and apical prolapses.
In contrast, Nam et al. [2] reported a weak connection between POP-Q outcomes and TLUS findings, mainly in the posterior part of POP. The POP-Q method showed prolapse of the posterior part and repose of the posterior vaginal wall with no enterocele or rectocele, which was evident on TLUS [2]. Some researchers have stated that using TLUS is an accurate and simple measurement method of the anorectal junction position, either in the rest mode or when it is active during straining [41]. It is difficult to distinguish between true and false rectoceles and detect whether a clinically apparent rectocele is caused by a rectovaginal septum, perineal hypermobility, or an isolated enterocele [17]. The moderate sagittal plane on TLUS can identify real rectocele cases caused by a rectovaginal septum defect [16].
Many conditions characterized by multiple vaginal masses may mimic POP. Braga et al. [42] reported the case of a woman with the main complaint of vaginal bulging and overactive bladder signs with POP-Q stage II anterior vaginal prolapse. Histopathological examination revealed an anterior vaginal leiomyoma. POP can be misdiagnosed in patients with vaginal wall cysts, vulvovaginal hemangioma in Klippel-Trénaunay syndrome, a perineal mass, or long cervical polyps [35,36].
Consequently, imaging techniques have been used to detect pelvic floor deflects and their association with neighboring organs, as an accompaniment to the POP-Q method. Even with the critical information provided by defecation proctography and MRI [2,43], they are seldom used in daily clinical practice, because they are costly, may not be accepted by patients, and carry risks of radiation exposure or allergic reactions to the contrast media. TLUS is more widely utilized because it is a commonly used, user-friendly imaging modality that is cheaper, easier to perform, and quicker, taking approximately 5 minutes. It provides the same information as the other two techniques [2,18]. The urogynecologist can implement surgical procedures for POP patients without performing defecation proctography and MRI. TLUS can enhance the accuracy of POP diagnosis when combined with a POP-Q exam with minimal extra effort and cost.
TLUS is an easy, inexpensive, and non-harmful diagnostic method that is appropriate for most gynecologic clinics.
TLUS is valuable in POP diagnosis, as it plays a role in recognizing POP cases and separating them from cases mimicking POP. There is a debate about the relationship between the POP-Q exam and TLUS. Particularly for posterior compartment POP. TLUS should be performed for patients with POP-Q exam results indicating a posterior compartment POP, to determine the existence of enterocele or rectocele. Further studies on the TLUS technique and its use in urogynaecology are needed to improve women’s health.
Notes
Sa Ra Lee has been an Editorial Board of Obstetrics & Gynecology Science; however, she was not involved in the peer reviewer selection, evaluation, or decision process of this article. Otherwise, no other potential conflicts of interest relevant to this article were reported.
References
1. Shek KL, Dietz HP. Assessment of pelvic organ prolapse: a review. Ultrasound Obstet Gynecol. 2016; 48:681–92.
2. Nam G, Lee SR, Kim SH, Chae HD. Importance of translabial ultrasound for the diagnosis of pelvic organ prolapse and its correlation with the POP-Q examination: analysis of 363 cases. J Clin Med. 2021; 10:4267.
3. Brown JS, Waetjen LE, Subak LL, Thom DH, Van den Eeden S, Vittinghoff E. Pelvic organ prolapse surgery in the United States, 1997. Am J Obstet Gynecol. 2002; 186:712–6.
4. Oh S, Namkung HR, Yoon HY, Lee SY, Jeon MJ. Factors associated with unsuccessful pessary fitting and reasons for discontinuation in Korean women with pelvic organ prolapse. Obstet Gynecol Sci. 2022; 65:94–9.
5. Kim BH, Lee SB, Na ED, Kim HC. Correlation between obesity and pelvic organ prolapse in Korean women. Obstet Gynecol Sci. 2020; 63:719–25.
6. Oh S, Namkung HR, Yoon HY, Lee SY, Jeon MJ. Factors associated with unsuccessful pessary fitting and reasons for discontinuation in Korean women with pelvic organ prolapse. Obstet Gynecol Sci. 2022; 65:94–9.
7. Kim JH, Lee SY, Chae HD, Shin YK, Lee SR, Kim SH. Outcomes of vaginal hysterectomy combined with anterior and posterior colporrhaphy for pelvic organ prolapse: a single center retrospective study. Obstet Gynecol Sci. 2022; 65:74–83.
8. Smith FJ, Holman CD, Moorin RE, Tsokos N. Lifetime risk of undergoing surgery for pelvic organ prolapse. Obstet Gynecol. 2010; 116:1096–100.
9. Løwenstein E, Ottesen B, Gimbel H. Incidence and lifetime risk of pelvic organ prolapse surgery in Denmark from 1977 to 2009. Int Urogynecol J. 2015; 26:49–55.
10. Digesu GA, Chaliha C, Salvatore S, Hutchings A, Khullar V. The relationship of vaginal prolapse severity to symptoms and quality of life. BJOG. 2005; 112:971–6.
11. Nygaard I, Bradley C, Brandt D. Pelvic organ prolapse in older women: prevalence and risk factors. Obstet Gynecol. 2004; 104:489–97.
12. Smith FJ, Holman CD, Moorin RE, Tsokos N. Lifetime risk of undergoing surgery for pelvic organ prolapse. Obstet Gynecol. 2010; 116:1096–100.
13. Oliphant SS, Jones KA, Wang L, Bunker CH, Lowder JL. Trends over time with commonly performed obstetric and gynecologic inpatient procedures. Obstet Gynecol. 2010; 116:926–31.
14. Wu JM, Hundley AF, Fulton RG, Myers ER. Forecasting the prevalence of pelvic floor disorders in U.S. women: 2010 to 2050. Obstet Gynecol. 2009; 114:1278–83.
15. Kim SR, Suh DH, Kim W, Jeon MJ. Current techniques used to perform surgery for anterior and posterior vaginal wall prolapse in South Korea. Obstet Gynecol Sci. 2022; 65:273–8.
16. Yuk JS, Lee JH, Hur JY, Shin JH. The prevalence and treatment pattern of clinically diagnosed pelvic organ prolapse: a Korean National Health Insurance database-based cross-sectional study 2009–2015. Sci Rep. 2018; 8:1334.
17. Dietz HP, Steensma AB. Posterior compartment prolapse on two-dimensional and three-dimensional pelvic floor ultrasound: the distinction between true rectocele, perineal hypermobility and enterocele. Ultrasound Obstet Gynecol. 2005; 26:73–7.
18. Salsi G, Cataneo I, Dodaro G, Rizzo N, Pilu G, Sanz Gascón M, et al. Three-dimensional/four-dimensional transperineal ultrasound: clinical utility and future prospects. Int J Womens Health. 2017; 9:643–56.
19. Dietz HP. Pelvic floor trauma following vaginal delivery. Curr Opin Obstet Gynecol. 2006; 18:528–37.
20. DeLancey JO, Morgan DM, Fenner DE, Kearney R, Guire K, Miller JM, et al. Comparison of levator ani muscle defects and function in women with and without pelvic organ prolapse. Obstet Gynecol. 2007; 109:295–302.
21. Shek KL, Dietz HP. The effect of childbirth on hiatal dimensions. Obstet Gynecol. 2009; 113:1272–8.
22. Dietz HP. Pelvic floor ultrasound: a review. Am J Obstet Gynecol. 2010; 202:321–34.
23. Dietz HP, Haylen BT, Broome J. Ultrasound in the quantification of female pelvic organ prolapse. Ultrasound Obstet Gynecol. 2001; 18:511–4.
24. DeLancey JO. The hidden epidemic of pelvic floor dysfunction: achievable goals for improved prevention and treatment. Am J Obstet Gynecol. 2005; 192:1488–95.
25. Dietz HP. Ultrasound imaging of the pelvic floor. Part II: three-dimensional or volume imaging. Ultrasound Obstet Gynecol. 2004; 23:615–25.
26. Dietz HP, Shek C, Clarke B. Biometry of the pubovisceral muscle and levator hiatus by three-dimensional pelvic floor ultrasound. Ultrasound Obstet Gynecol. 2005; 25:580–5.
27. Dietz HP, Steensma AB. The prevalence of major abnormalities of the levator ani in urogynaecological patients. BJOG. 2006; 113:225–30.
28. Siafarikas F, Staer-Jensen J, Braekken IH, Bø K, Engh ME. Learning process for performing and analyzing 3D/4D transperineal ultrasound imaging and interobserver reliability study. Ultrasound Obstet Gynecol. 2013; 41:312–7.
29. Majida M, Braekken IH, Umek W, Bø K, Saltyte Benth J, Ellstrøm Engh M. Interobserver repeatability of three- and four-dimensional transperineal ultrasound assessment of pelvic floor muscle anatomy and function. Ultrasound Obstet Gynecol. 2009; 33:567–73.
30. van Veelen GA, Schweitzer KJ, van der Vaart CH. Reliability of pelvic floor measurements on three- and four-dimensional ultrasound during and after first pregnancy: implications for training. Ultrasound Obstet Gynecol. 2013; 42:590–5.
31. Chen R, Song Y, Jiang L, Hong X, Ye P. The assessment of voluntary pelvic floor muscle contraction by three-dimensional transperineal ultrasonography. Arch Gynecol Obstet. 2011; 284:931–6.
32. van Delft K, Thakar R, Sultan AH. Pelvic floor muscle contractility: digital assessment vs transperineal ultrasound. Ultrasound Obstet Gynecol. 2015; 45:217–22.
33. van Delft KW, Sultan AH, Thakar R, Shobeiri SA, Kluivers KB. Agreement between palpation and transperineal and endovaginal ultrasound in the diagnosis of levator ani avulsion. Int Urogynecol J. 2015; 26:33–9.
34. Gao Y, Zhao Z, Yang Y, Zhang M, Wu J, Miao Y. Diagnostic value of pelvic floor ultrasonography for diagnosis of pelvic organ prolapse: a systematic review. Int Urogynecol J. 2020; 31:15–33.
35. Nam G, Lee SR, Eum HR, Kim SH, Chae HD, Kim GJ. A huge hemorrhagic epidermoid cyst of the perineum with hypoechoic semisolid ultrasonographic feature mimicking scar endometriosis. Medicina (Kaunas). 2021; 57:276.
36. Nam G, Lee SR, Choi S. Clitoromegaly, vulvovaginal hemangioma mimicking pelvic organ prolapse, and heavy menstrual bleeding: gynecologic manifestations of klippel-trénaunay syndrome. Medicina (Kaunas). 2021; 57:366.
37. Lee SR, Lim YM, Jeon JH, Park MH. Diagnosis of urethral diverticulum mimicking pelvic organ prolapse with translabial ultrasonography. Am J Obstet Gynecol. 2017; 217:482.
38. Chang OH, Davidson ERW, Thomas TN, Paraiso MFR, Ferrando CA. Does concurrent posterior repair for an asymptomatic rectocele reduce the risk of surgical failure in patients undergoing sacrocolpopexy? Int Urogynecol J. 2020; 31:2075–80.
39. Arunachalam D, Hale DS, Heit MH. Posterior compartment surgery provides no differential benefit for defecatory symptoms before or after concomitant mesh-augmented apical suspension. Female Pelvic Med Reconstr Surg. 2018; 24:183–7.
40. Dietz HP. Ultrasound imaging of the pelvic floor. Part I: two-dimensional aspects. Ultrasound Obstet Gynecol. 2004; 23:80–92.
41. Beer-Gabel M, Teshler M, Schechtman E, Zbar AP. Dynamic transperineal ultrasound vs. defecography in patients with evacuatory difficulty: a pilot study. Int J Colorectal Dis. 2004; 19:60–7.
42. Braga A, Soave I, Caccia G, Regusci L, Ruggeri G, Pitaku I, et al. What is this vaginal bulge? An atypical case of vaginal paraurethral leiomyoma. A case report and literature systematic review. J Gynecol Obstet Hum Reprod. 2021; 50:101822.
43. Rechi-Sierra K, Sánchez-Ballester F, García-Ibáñez J, Pardo-Duarte P, Flores-Delatorre M, Monzó-Cataluña A, et al. Magnetic resonance imaging to evaluate anterior pelvic prolapse: H line is the key. Neurourol Urodyn. 2021; 40:1042–7.
Fig. 1
(A) Application of a convex transducer to labium minora for translabial ultrasonography. (B) Symphysis pubis, urethra, bladder, uterus, vagina, cul-de-sac, rectum, rectal ampulla, and colon are visible on translabial ultrasound. (C) Two-dimensional view of the midsagittal plane of the pelvic floor. The main structures identified on this plane are, from left to right, symphysis pubis (SP), urethra (U), urinary bladder (UB), vagina (V), rectum (R) and the puborectalis muscle (PR) passing behind the rectum [18].
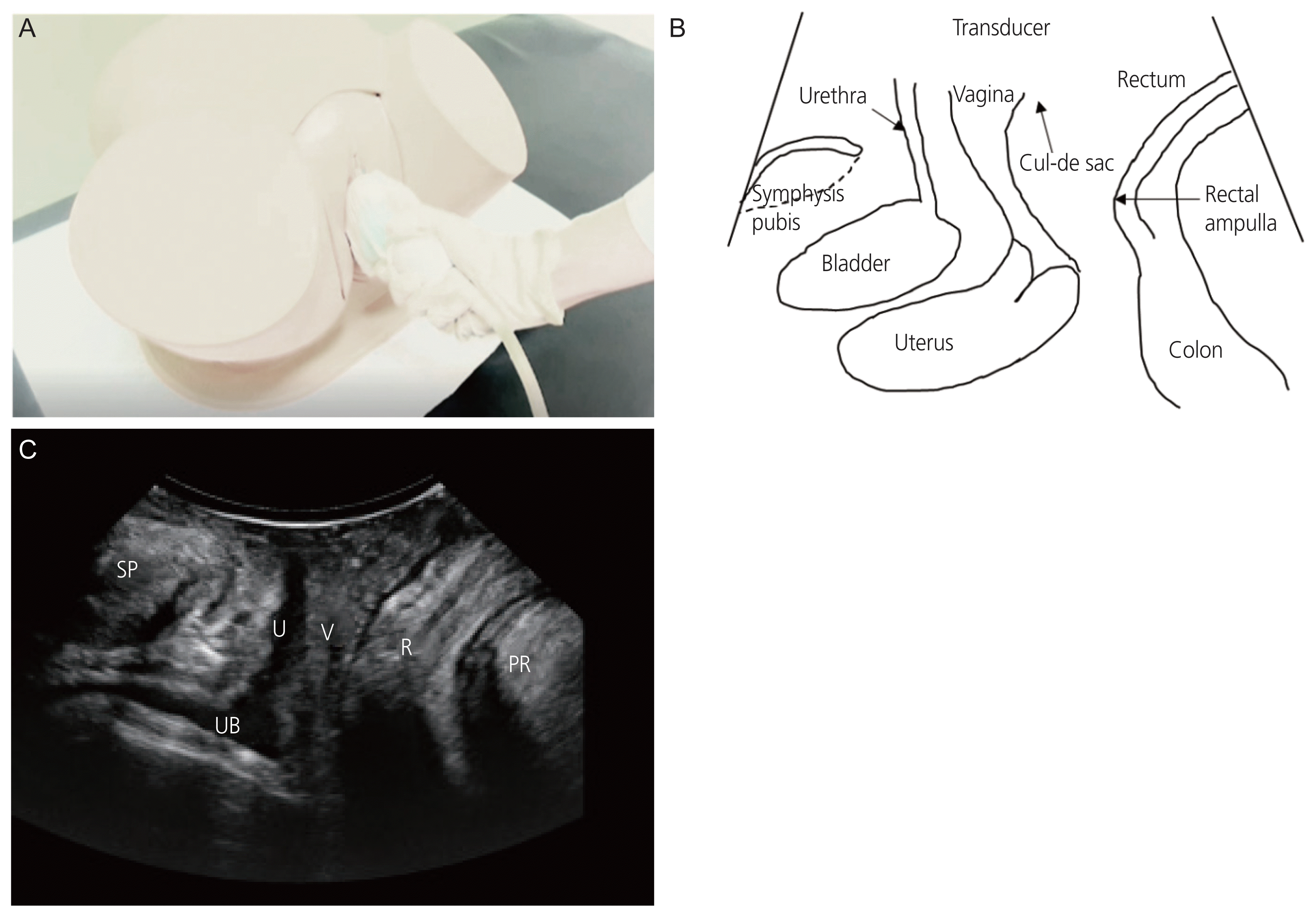
Fig. 2
Avulsion injury detected on a three-dimensional transperineal ultrasound image of women with pelvic organ prolapse. The yellow dashed circle includes the avulsion injury structure [34].
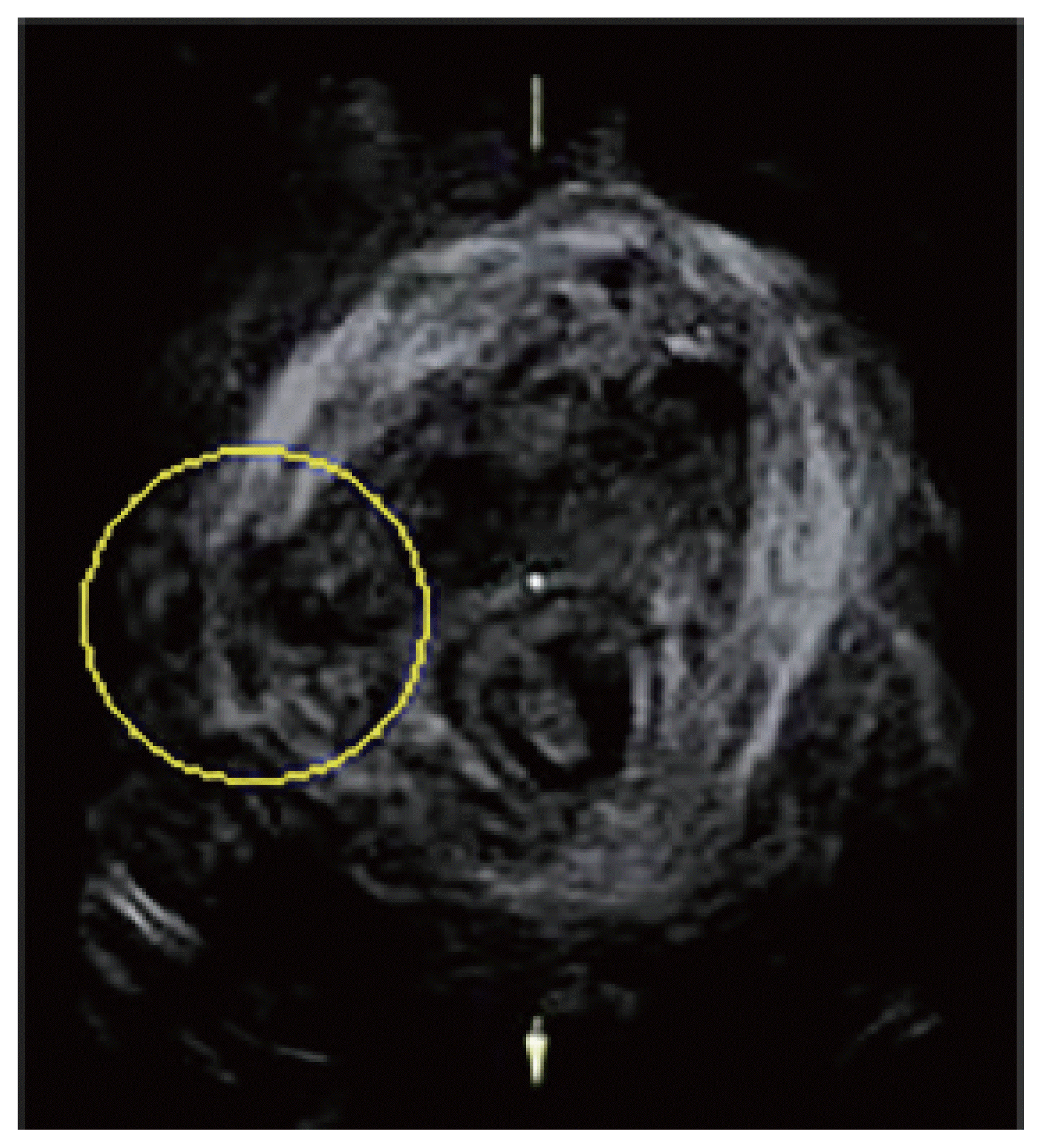
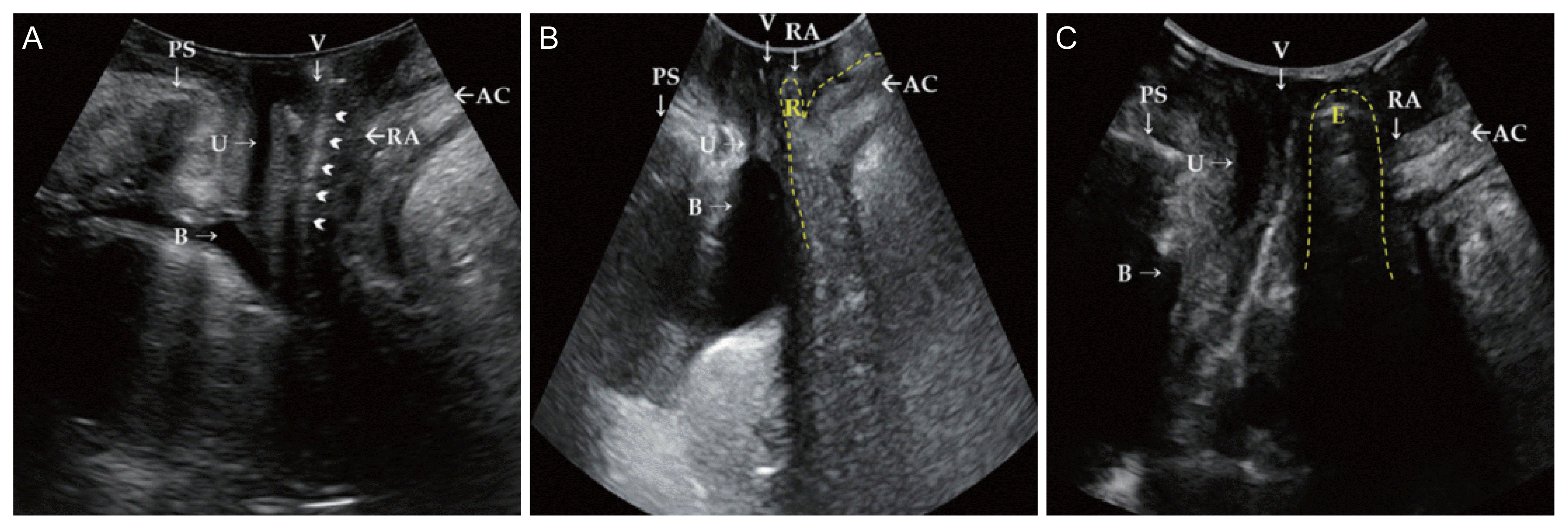
Fig. 3
Translabial ultrasonography (A) in the resting phase. Rectovaginal septum (arrowheads). Translabial ultrasonography in patients with (B) rectocele and (C) enterocele in maximal Valsalva phase. Rectocele filled with stool and air, resulting in hyper echogenicity, and the defect of the rectovaginal septum is observed in the maximal Valsalva maneuver (B). The contents of an enterocele generally appear iso- to hyperechogenic compared with a rectocele, and bowel peristalsis is usually observed in the enterocele sac (C) [2]. PS, pubis symphysis; V, vagina; AC, anal canal; U, urethra; RA, rectal ampulla; B, bladder; R, rectocele; E, enterocele.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download