Abstract
Cesarean scar pregnancy (CSP) is a rare complication that occurs in less than 1% of ectopic pregnancies, and uterine didelphys is one of the rarest uterine forms. We report a successful laparoscopic excision and repair of CSP in a woman with uterine didelphys and a double vagina. A 34-year-old gravida one, para one woman with a history of low transverse cesarean section presented to our hospital with a suspected CSP. She was confirmed to have uterine didelphys with a double vagina during an infertility examination 7 years earlier. Magnetic resonance imaging showed a 2.5-cm gestational sac-like cystic lesion in the lower segment of the right uterus at the cesarean scar. We decided to perform a laparoscopic approach after informing the patient of the surgical procedure. The lower segment of the previous cesarean site was excised with monopolar diathermy to minimize bleeding. We identified the gestational sac in the lower segment of the right uterus, which was evacuated using spoon forceps. The myometrium and serosa of the uterus were sutured layer-by-layer using synthetic absorbable sutures. No remnant gestational tissue was visible on follow-up ultrasonography one month after the surgery. This laparoscopic approach to CSP in a woman with uterine didelphys is an effective and safe method of treatment. In women with uterine anomalies, it is important to confirm the exact location of the gestational sac by preoperative imaging for successful surgery.
Cesarean scar pregnancy (CSP) is a rare complication of pregnancy, occurring in less than 1% of ectopic pregnancies [1]. A CSP is defined as implantation of the blastocyst at the myometrium through dehiscence of a previous cesarean scar [2]. There are two types of CSP using imaging modalities. Type 1 (endogenic type) develops in the myometrium and progresses toward the uterine cavity, and type 2 (exogenic type) deeply invades in the scar defect and progresses toward the uterine serosa with possible uterine rupture [3]. CSPs have increased in prevalence with increasing cesarean section deliveries and advances in ultrasonography [4]. However, CSPs are not easy to diagnose, with a misdiagnosis rate of 70% to 80% during the first examination [5]. Upon ultrasonography, CSPs can be misdiagnosed as threatened and incomplete abortions and cervical pregnancies, depending on the gestational sac’s location and shape. Moreover, the diagnosis of CSP may be more difficult if there is a uterine malformation, such as uterine didelphys. Uterine didelphys is one of the rarest uterine forms, accounting for 8.3% of all Müllerian anomalies [6]. Uterine didelphys arises from failure of the Müllerian ducts to fuse resulting in two separate endometrial cavities, two cervices, and often a longitudinal vaginal septum. We report a successful laparoscopic treatment of a very rare case of CSP in a woman with uterine didelphys.
Ethical statements: This study was approved by the Institutional Review Board (IRB) of Keimyung University Dongsan Hospital (IRB No: 2022-02-062) and written informed consent from the patient was waived by the IRB due to retrospective nature of this case study.
A 34-year-old woman, gravida two, para one, with a history of low transverse cesarean section, was initially referred to Keimyung University Dongsan Hospital for a suspected CSP at 7 weeks gestation. Her serum β-human chorionic gonadotropin (β-HCG) concentration was 30,626 mIU/mL at her first visit to our hospital. During a fertility examination performed at another hospital 7 years earlier, she was diagnosed with uterine didelphys, two cervices, and a longitudinal vaginal septum. This pregnancy was confirmed as naturally conceived without infertility procedures. Upon pelvic examination, the left vagina was narrow and inaccessible. The pelvic examination was performed entirely through the right vagina and included vaginal ultrasonography. Based on vaginal ultrasonography, a CSP with active cardiac activity was suspected in the right uterus, and magnetic resonance imaging (MRI) was performed for an accurate diagnosis. A fetal pole (crown-rump length, 20 mm) in a gestational sac was located at the right lower uterine segment with a 41-mm sized intramural myoma at the uterine anterior wall (Fig. 1). There was no evidence of hemorrhage in the endometrial cavity or free fluid in the cul-de-sac. The patient was counseled about the medical treatment of CSP, which had a high probability of failure based on the high level of β-HCG. Because of the risk of uterine bleeding during the pregnancy, we decided to remove only the gestational sac through laparoscopic surgery without removing the uterine myoma, with the patient’s consent.
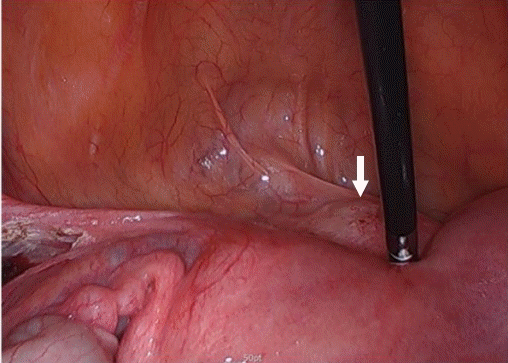
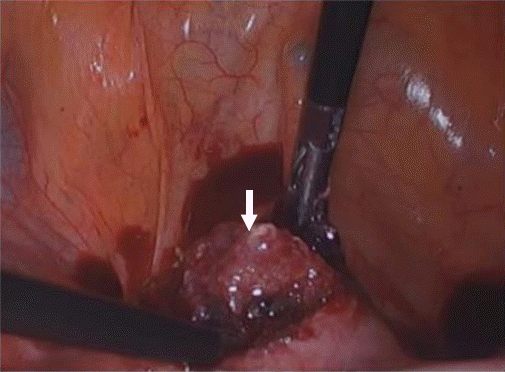
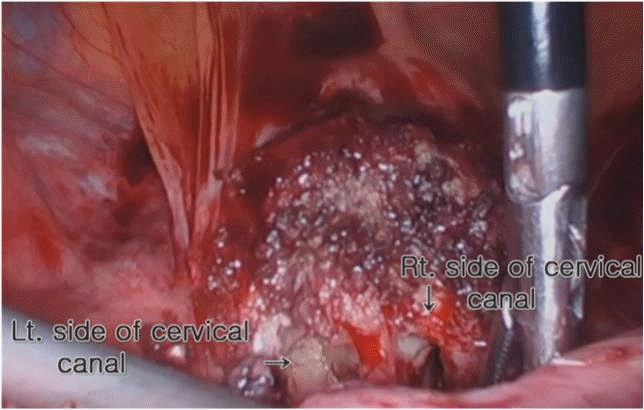
The patient underwent CSP laparoscopic excision with repair based on a stepwise approach. A uterine manipulator was used to facilitate the surgery by easily moving the uterus, which was entered through the right cervix. During the laparoscopy, it was noted that the bladder had adhered to the uterine anterior wall due to the previous cesarean section, and the focus of the ectopic pregnancy was presumed to be a lump protruding outwards from the uterine lower segment (Fig. 2). As the first step, both uterine arteries were clamped to reduce bleeding risk during the surgery. A small transverse incision was made in the lower segment of the uterus, just above the adherent bladder to differentiate it from the bladder flap after the uterus was positioned as far as possible into the abdominal cavity using the uterine manipulator. Careful dissection was performed using monopolar diathermy within the areolar tissue at the vesicouterine plane. As the second step, the focus of the ectopic pregnancy was verified at the uterine lower segment and excised without residual lesion. The products of conception on the lower anterior myometrium were removed using spoon forceps (Fig. 3). No massive bleeding occurred from the implantation site while removing the gestational sac. After complete excision, clear bifurcated endocervical canals could be identified without residual lesion (Fig. 4). As a final step, the uterus was repaired layer-by-layer. The defective myometrium was sutured using VICRYL sutures (Ethicon, Somerville, NJ, USA) to prevent occlusion of the bifurcated endocervical canals. The uterine serosa was sutured with 2-0 barbed monofilament sutures.
The patient’s postoperative condition was uneventful, and she was discharged home on day 2 after surgery. The histological analysis of the extracted tissue confirmed that it was gestational tissue. Ultrasonography was performed and β-HCG level was assessed 2 weeks later during her follow-up at the outpatient clinic. The ultrasonography showed that the CSP had completely resolved and endometrial thickness was approximately 15 mm without myometrial defect; the β-HCG level also decreased to 88 mIU/mL. She is currently being followed up for 28 months with no surgery-related problems noted to date.
We presented a 34-year-old patient who had been diagnosed with uterine didelphys during a fertility test and had previously undergone a cesarean section. She was asymptomatic at 7 weeks gestation with suspected CSP detected by imaging. Although we successfully finished the laparoscopic surgery by excising and repairing the myometrial defect, we performed the surgery under anatomical uncertainty. Moreover, as in our patient’s case, CSP in patients with uterine malformations, such as uterine didelphys, can make treatment decisions more difficult. Due to limited reports and varying clinical circumstances involving CSPs, the treatment decision is made by clinical judgment and a clinician’s preference among various treatment options. Options range from expectant management, medical management (systemic or gestational sac injections), and surgical approaches. Regardless, early diagnosis and treatment can provide the best outcome because delayed treatment due to misdiagnosis is associated with severe maternal morbidity and mortality due to bleeding risks [7].
Methotrexate (MTX) is a common treatment for ectopic pregnancy and has traditionally been considered an option for ectopic pregnancies with β-HCG values less than 10,000 mIU/mL. In a review report, systemic MTX treatment for CSPs was found to be effective when the serum β-HCG levels were less than 12,000 mIU/mL and fetal cardiac activity was negative [8]. Unlike tubal pregnancies, exposure of the gestational sac to MTX is limited with CSPs due to the fibrous tissue surrounding the gestational sac. This means that local MTX injection is considered an alternative [7]. However, Peng et al. [9] reported the results of a randomized trial comparing local versus systemic injection of MTX in 104 patients with CSP, and success rates were similar for local and systemic injections (69.2% vs. 67.3%, respectively). A surgical approach was our inevitable option because the fetus had active cardiac activity and the β-HCG level was above 30,000 mIU/mL at the first visit. Surgical treatment with a success rate of over 95% is the most obvious option for gestational sac removal and uterine defect repair while maintaining fertility [10]. The main advantage of a surgical approach is that if the gestational sac is completely removed during surgery, the treatment itself can be terminated and the outpatient follow-up time can be shortened.
A variety of surgical methods can be performed, including laparotomy, laparoscopy, hysteroscopy, and dilation and curettage, depending on various circumstances, such as the patient's condition at the time of diagnosis, location of the gestational sac, and surgical skills of the attending physician. Hysteroscopy for gestational sac access can be considered in patients with normal anatomy, but it would have been difficult in our case with uterine anomalies. We chose the laparoscopic approach in consulting the patient, despite her having uterine didelphys. The first reason was that the patient’s condition was hemodynamically stable. Second, as the gestational sac was presumed to be type 2 CSP (exogenic type), protruding into the abdominal cavity by MRI, it was thought that the gestational sac would be easily accessible through laparoscopy. In related literature, it was reported that laparoscopic surgery in CSP is suitable when the gestational sac is growing toward the abdominal cavity [11,12]. Third, surgeons are more likely to choose the procedure they are most familiar with, especially in difficult circumstances involving patients with CSP and uterine didelphys. Finally, since laparoscopic resection of CSP has been reported at up to 11 weeks gestation, the gestational sac was considered sufficiently accessible [13].
There are several case reports of laparoscopic bilateral ligation of the uterine arteries during surgical management of CSP, and we also removed the gestational sac after bilateral ligation of the uterine arteries [14,15]. Ligating both uterine arteries prior to removal of the gestational sac is a very useful procedure, as it can minimize bleeding risk and enables the surgeon to perform the operation under stable conditions. In randomized clinical trials, permanent uterine artery ligation was an effective method for blood loss reduction in surgeries for excision and myometrial defect repair [16,17]. Although there have been reports that permanent uterine artery ligation is associated with early miscarriage, intrauterine growth restriction, and preterm birth [18,19], if the patient wishes no future pregnancy, it is a surgical procedure that can be fully considered, as in our case.
As cesarean delivery increases, clinicians will have more clinical experience with CSPs, but CSPs are still rare to date and those with uterine malformations are even rarer. To our knowledge, this is the first case report of CSP in a woman with uterine didelphys in the medical literature. Therefore, it is important for clinicians to identify this rare condition, such as with uterine didelphys, and to make an accurate diagnosis for prompt treatment. The optimal management of CSP in a patient with uterine didelphys remains unclear. The choice of treatment modality can be influenced by various clinical conditions, including the type of CSP, gestational week, hemodynamic stability, level of β-HCG, possibility of future pregnancy, clinician expertise, and surgical proficiency. Surgical treatment has a high success rate. However, the surgeon must be skilled, especially in laparoscopic surgery, and the surgical risk itself must be discussed with the patient.
In summary, this case is the successful treatment of a woman with CSP and uterine didelphys through a laparoscopic approach. We demonstrated that a laparoscopic stepwise approach can ensure a safe and efficient surgical treatment of CSP. Although the surgical approach selected in our report is one of various treatment methods for CSP, it can be a useful reference for similar cases in the future.
References
1. Seow KM, Huang LW, Lin YH, Lin MY, Tsai YL, Hwang JL. Cesarean scar pregnancy: issues in management. Ultrasound Obstet Gynecol. 2004; 23:247–53.
2. Moschos E, Wells CE, Twickler DM. Biometric sonographic findings of abnormally adherent trophoblastic implantations on cesarean delivery scars. J Ultrasound Med. 2014; 33:475–81.
3. Timor-Tritsch IE, Monteagudo A, Santos R, Tsymbal T, Pineda G, Arslan AA. The diagnosis, treatment, and follow-up of cesarean scar pregnancy. Am J Obstet Gynecol. 2012; 207:44.
4. Kirk E. Ultrasound in the diagnosis of ectopic pregnancy. Clin Obstet Gynecol. 2012; 55:395–401.
5. Jurkovic D, Knez J, Appiah A, Farahani L, Mavrelos D, Ross JA. Surgical treatment of Cesarean scar ectopic pregnancy: efficacy and safety of ultrasound-guided suction curettage. Ultrasound Obstet Gynecol. 2016; 47:511–7.
6. Rezai S, Bisram P, Lora Alcantara I, Upadhyay R, Lara C, Elmadjian M. Didelphys uterus: a case report and review of the literature. Case Rep Obstet Gynecol. 2015; 2015:865821.
7. Riaz RM, Williams TR, Craig BM, Myers DT. Cesarean scar ectopic pregnancy: imaging features, current treatment options, and clinical outcomes. Abdom Imaging. 2015; 40:2589–99.
8. Bodur S, Özdamar Ö, Kılıç S, Gün I. The efficacy of the systemic methotrexate treatment in caesarean scar ectopic pregnancy: a quantitative review of English literature. J Obstet Gynaecol. 2015; 35:290–6.
9. Peng P, Gui T, Liu X, Chen W, Liu Z. Comparative efficacy and safety of local and systemic methotrexate injection in cesarean scar pregnancy. Ther Clin Risk Manag. 2015; 11:137–42.
10. Glenn TL, Bembry J, Findley AD, Yaklic JL, Bhagavath B, Gagneux P, et al. Cesarean scar ectopic pregnancy: current management strategies. Obstet Gynecol Surv. 2018; 73:293–302.
11. Tulandi T, Cohen A. Emerging manifestations of cesarean scar defect in reproductive-aged women. J Minim Invasive Gynecol. 2016; 23:893–902.
12. Zhai JF, Xu M, Zhang B, Gao JW, Chen N. Treatments of caesarean scar pregnancy and the corresponding results in ten years. Eur Rev Med Pharmacol Sci. 2015; 19:2523–7.
13. Wang YL, Su TH, Chen HS. Operative laparoscopy for unruptured ectopic pregnancy in a caesarean scar. BJOG. 2006; 113:1035–8.
14. Wang L, Sun L, Wang L, Chen H, Ouyang X, Qiu H. Laparoscopic temporary bilateral uterine artery occlusion with silicone tubing to prevent hemorrhage during vacuum aspiration of cesarean scar pregnancies. J Obstet Gynaecol Res. 2015; 41:1762–8.
15. Fuchs N, Manoucheri E, Verbaan M, Einarsson JI. Laparoscopic management of extrauterine pregnancy in caesarean section scar: description of a surgical technique and review of the literature. BJOG. 2015; 122:137–40.
16. Vercellino G, Erdemoglu E, Joe A, Hopfenmueller W, Holthaus B, Köhler C, et al. Laparoscopic temporary clipping of uterine artery during laparoscopic myomectomy. Arch Gynecol Obstet. 2012; 286:1181–6.
17. Fletcher H, Frederick J, Hardie M, Simeon D. A randomized comparison of vasopressin and tourniquet as hemostatic agents during myomectomy. Obstet Gynecol. 1996; 87:1014–8.
18. Chen YJ, Wang PH, Yuan CC, Yen YK, Yang MJ, Ng HT, et al. Pregnancy following treatment of symptomatic myomas with laparoscopic bipolar coagulation of uterine vessels. Hum Reprod. 2003; 18:1077–81.
19. Holub Z, Mara M, Kuzel D, Jabor A, Maskova J, Eim J. Pregnancy outcomes after uterine artery occlusion: prospective multicentric study. Fertil Steril. 2008; 90:1886–91.




 PDF
PDF Citation
Citation Print
Print




 XML Download
XML Download