Introduction
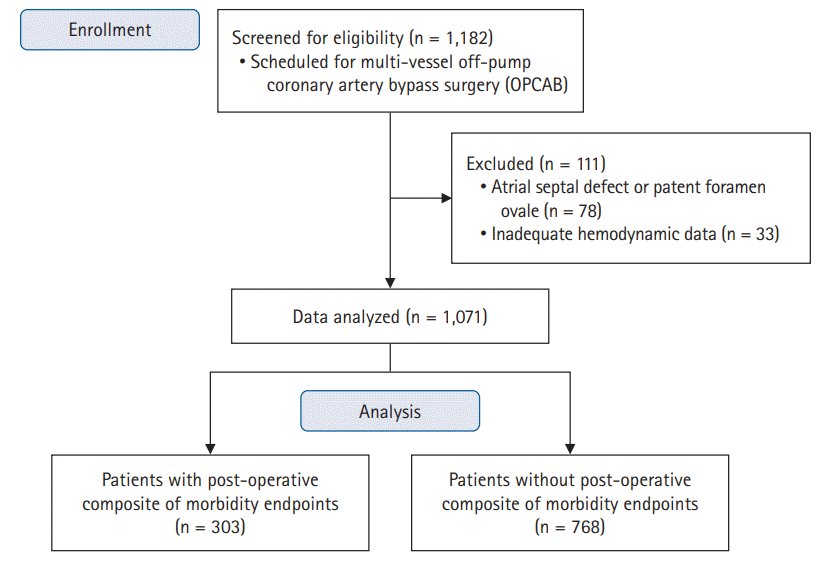
Materials and Methods
Intraoperative management
Study protocol
Study endpoints
Statistical analysis
Results
Table 1.
Values are presented as median (Q1, Q3) or number of patients (%). SvO2: mixed venous oxygen saturation, LCX: left circumflex artery, SvO2 ≥ 60% group: patients whose nadir SvO2 during LCX grafting was more than or equal to 60%, SvO2 < 60% group: patients whose nadir SvO2 during LCX grafting was less than 60%, BMI: body mass index, HTN: hypertension, DM: diabetes mellitus, COPD: chronic obstructive pulmonary disease, CKD: chronic kidney disease, CVA: cerebrovascular accident, MI: myocardial infarction, CHF: chronic heart failure, E/e’: early mitral inflow velocity / mitral annular early diastolic velocity, LVEDD: left ventricular end-diastolic diameter.
Table 2.
| Variable | Baseline | LAD grafting | LCX grafting | RCA grafting | Sternal closure | Pgroup × time |
|---|---|---|---|---|---|---|
| HR (beats/min) | 0.997 | |||||
| SvO2 ≥ 60% group | 59 (53, 65) | 66 (60, 72)* | 70 (63, 76)* | 71 (63, 77)* | 77 (71, 82)* | |
| SvO2 < 60% group | 59 (53, 66) | 68 (61, 74)* | 70 (63, 78)* | 71 (65, 79)* | 77 (71, 83)* | |
| MAP (mmHg) | 0.196 | |||||
| SvO2 ≥ 60% group | 75 (69, 81) | 77 (72, 82)* | 78 (72, 84)* | 77 (72, 82)* | 77 (70, 84) | |
| SvO2 < 60% group | 76 (69, 87) | 76 (71, 81) | 75 (68, 82) | 76 (71, 82) | 77 (72, 83) | |
| CVP (mmHg) | 0.223 | |||||
| SvO2 ≥ 60% group | 9 (8, 11) | 12 (10, 14)* | 17 (13, 19)* | 16 (14, 18)* | 10 (8, 11) | |
| SvO2 < 60% group | 9 (7, 11) | 11 (10, 13)* | 16 (12, 19)* | 16 (13, 18)* | 9 (8, 11) | |
| mPAP (mmHg) | 0.148 | |||||
| SvO2 ≥ 60% group | 17 (15, 20) | 21 (19, 23)* | 24 (21, 27)* | 25 (22, 27)* | 18 (16, 20) | |
| SvO2 < 60% group | 17 (15, 20) | 20 (18, 22)* | 22 (19, 26)* | 23 (20, 27)* | 18 (16, 21) | |
| Cardiac index (L/min/m2) | 0.031 | |||||
| SvO2 ≥ 60% group | 2.3 (1.9, 2.6) | 2.0 (1.7, 2.3)* | 1.8 (1.6, 2.1)* | 1.8 (1.6, 2.0)* | 2.2 (2.0, 2.6) | |
| SvO2 < 60% group | 2.1 (1.7, 2.5)† | 1.7 (1.5, 2.1)*† | 1.6 (1.4, 1.8)*† | 1.5 (1.3, 1.7)*† | 1.9 (1.6, 2.2)*† | |
| SvO2 (%) | < 0.001 | |||||
| SvO2 ≥ 60% group | 80 (77, 84) | 77 (73, 81)* | 73 (68, 78)* | 74 (69, 78)* | 78 (74, 82)* | |
| SvO2 < 60% group | 75 (71, 80)† | 67 (62, 70)*† | 57 (54, 58)*† | 61 (58, 66)*† | 70 (65, 75)*† | |
| Incidence of SvO2 < 60% (n) | N/A | |||||
| SvO2 ≥ 60% group | 0 (0%) | 3 (0.3%) | 0 (0%) | 1 (0%) | 0 (0%) | |
| SvO2 < 60% group | 0 (0%) | 8 (8.6%)† | 93 (100%)† | 22 (23.7%)† | 0 (0%) |
Values are presented as median (Q1, Q3) or number of patients (%). SvO2: mixed venous oxygen saturation, LCX: left circumflex artery, LAD: left anterior descending artery, RCA: right coronary artery, SvO2 ≥ 60% group: patients whose nadir SvO2 during LCX grafting was more than or equal to 60%, SvO2 < 60% group: patients whose nadir SvO2 during LCX grafting was less than 60%, HR: heart rate, MAP: mean systemic arterial pressure, CVP: central venous pressure, mPAP: mean pulmonary arterial pressure.
Table 3.
| Variable | Univariable analysis | Multivariable analysis | ||
|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | |
| Age | 1.05 (1.03, 1.06)* | < 0.001 | 1.03 (1.01, 1.05)* | 0.008 |
| CKD | 3.28 (2.23, 4.81)* | < 0.001 | 2.15 (1.32, 3.51)* | 0.002 |
| Pre-operative anemia | 1.96 (1.50, 2.56)* | < 0.001 | 1.45 (1.00, 2.12) | 0.053 |
| E/e’ | 1.07 (1.05, 1.10)* | < 0.001 | 1.05 (1.01, 1.08)* | 0.005 |
| EuroSCORE II | 1.26 (1.16, 1.37)* | < 0.001 | 1.12 (1.01, 1.24)* | 0.040 |
| SvO2 < 60% during LAD grafting | 4.67 (1.36, 16.10)* | 0.015 | 2.61 (0.64, 10.56) | 0.180 |
| SvO2 < 60% during LCX grafting | 3.33 (2.16, 5.13)* | < 0.001 | 2.72 (1.60, 4.61)* | < 0.001 |
| SvO2 < 60% during RCA grafting | 2.55 (1.11, 5.86)* | 0.028 | 0.46 (0.15, 1.38) | 0.166 |
Table 4.
| Variable | SvO2 ≥ 60% (n = 978) | SvO2 < 60% (n = 93) | P value |
|---|---|---|---|
| ICU days | 3 (2, 3) | 3 (2, 4)* | 0.015 |
| Hospital days | 13 (10, 15) | 14 (11, 20)* | 0.003 |
| 30-day or in-hospital mortality | 13 (1.3) | 6 (6.5)* | < 0.001 |
| Post-operative MI | 4 (0.4) | 0 (0) | 0.537 |
| Prolonged mechanical ventilation > 24 h | 71 (7.3) | 10 (10.8) | 0.223 |
| Post-operative CVA | 21 (2.1) | 2 (2.2) | 0.998 |
| AKI | 192 (19.6) | 41 (44.1)* | < 0.001 |
Values are presented as median (Q1, Q3) or number of patients (%). SvO2: mixed venous oxygen saturation, LCX: left circumflex artery, SvO2 ≥ 60% group: patients whose nadir SvO2 during LCX grafting was more than or equal to 60%, SvO2 < 60% group: patients whose nadir SvO2 during LCX grafting was less than 60%, ICU: intensive care unit, MI: myocardial infarction, CVA: cerebrovascular accident, AKI: acute kidney injury.
Table 5.
| Variable | Univariable analysis | Multivariable analysis | ||
|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | |
| Age | 1.03 (1.00, 1.05)* | 0.041 | 1.01 (0.99, 1.04) | 0.250 |
| HTN | 0.98 (0.61, 1.57) | 0.935 | ||
| DM | 1.20 (0.78, 1.84) | 0.414 | ||
| COPD | 0.24 (0.32, 1.74) | 0.236 | ||
| CKD | 1.86 (1.06, 3.27)* | 0.031 | 1.25 (0.68, 2.30) | 0.206 |
| Old CVA | 0.85 (0.44, 1.63) | 0.614 | ||
| MI within one month | 1.20 (0.75, 1.90) | 0.451 | ||
| CHF | 1.37 (0.73, 2.54) | 0.325 | ||
| Pre-operative anemia | 2.35 (1.51, 3.64)* | < 0.001 | 2.09 (1.33, 3.29)* | 0.001 |
| Ejection fraction | 1.00 (0.98, 1.02) | 0.953 | ||
| E/e’ | 1.02 (0.98, 1.05) | 0.338 | ||
| LVEDD | 1.01 (0.98, 1.04) | 0.645 | ||
SvO2: mixed venous oxygen saturation, LCX: left circumflex artery, OR: odds ratio, HTN: hypertension, DM: diabetes mellitus, COPD: chronic obstructive pulmonary disease, CKD: chronic kidney disease, CVA: cerebrovascular accident, MI: myocardial infarction, CHF: chronic heart failure, E/e’: early mitral inflow velocity / mitral annular early diastolic velocity, LVEDD: left ventricular end-diastolic diameter.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download