Abstract
Background
Hyponatremia is a common complication after transsphenoidal surgery (TSS) for pituitary adenomas. This study retrospectively reviewed various clinical factors of pituitary adenoma patients who underwent TSS and aimed to identify possible risk factors of postoperative hyponatremia.
Methods
Total 1,343 patients who underwent TSS for their pituitary adenomas in a single institution were enrolled to this study. We identified and analyzed 93 patients (6.9%) with postoperative hyponatremia and compared them with other 1,250 patients in a control group. Patients' age, sex, tumor extent based on the modified Hardy classification, tumor size and hormonal type of the tumors were analyzed in comparison between two groups. The results of pre- and post-operative combined pituitary function test and their comparison were analyzed to elucidate a possible association between postoperative hyponatremia and hypopituitarism.
Results
The onset of postoperative hyponatremia was 7.8 days after surgery (range: postoperative day 3–13). The initial level of serum sodium (Na) level at the start of treatment was 123.5 mmol/L (range: 114–131 mmol/L) and the duration of treatment was 2.7 days (range: 1–9 days). Female predominance was evident in the hyponatremia group (77.4% vs. 61.4%, p=0.02). Preoperative hypopituitarism (58.5% vs. 71.5%, p=0.007) and postoperative hypocortisolism (33.7% vs. 23.4%, p=0.029) were related to postoperative hyponatremia. Other pre- and post-operative clinical factors were not associated with postoperative hyponatremia.
Conclusion
Postoperative hyponatremia is a common complication after TSS and is potentially fatal. Female patients were more likely to develop delayed hyponatremia. We demonstrated possible associations of delayed postoperative hyponatremia with preoperative and postoperative pituitary functions, but the mechanism behind it should be further investigated.
Postoperative hyponatremia is relatively common among postoperative complications that occur in patients who underwent transsphenoidal surgery (TSS) for pituitary adenomas, with incidence varying from 1.8% to 35% [123456]. Common clinical presentations include headaches, nausea and vomiting, but more severe symptoms like seizures, mental change can develop if left untreated. It is known that significant postoperative hyponatremia usually develops after the latter part of first postoperative week, however, its pathophysiology has been under debate [5789]. Hormonal imbalance caused by disruption of the normal pituitary gland during surgery is thought to be the mechanism behind it, but the exact mechanism or risk factors of postoperative hyponatremia is still unclear despite several previous studies [134610111213]. As the consequences of untreated hyponatremia are often fatal, this study aims to ascertain risk factors associated with postoperative hyponatremia in patients who underwent TSS for their pituitary adenomas.
Total 1,343 patients with pituitary adenomas operated with TSS in our institution were included in this study. Patients' medical record and imaging studies were retrospectively reviewed. This study was conducted in accordance with the Declaration of Helsinki and approved by Institutional Review Board in our institute (4-2018-0039), waving the patient's written consent as a retrospective study. Delayed hyponatremia was defined as sodium level less than 135 mmol/L that occurred on or after postoperative day 3. We identified 93 patients who presented with rapid drop of their sodium level or significant clinically relevant symptoms, and thus required administration of hypertonic saline (3% NaCl). These 93 patients were defined as hyponatremia group and their clinical features were compared with those of control group.
All patients were evaluated preoperatively with dynamic sellar MRI using either 1.5-Tesla system (Signa, General Electric, Minneapolis, MN, USA) or 3.0-Tesla system (Achieva, Philips, Best, The Netherlands). Tumor size was determined as a maximum diameter of the tumor on a coronal view of preoperative MRI. With the preoperative MRI all the patients were classified into modified Hardy's classification of pituitary adenoma [141516]. Immediate postoperative 3-Tesla MRI was performed 48 hours after the surgery to determine the radicality of tumor removal. Specific methods of endocrinological assessment were fully described in our previous report [13]. Combined pituitary function test (CPFT) was performed to evaluate anterior pituitary function before surgery, 6–12 months after surgery and every 2 years thereafter.
All tumors were approached by conventional microscopic or endoscopic transsphenoidal approach. After dural opening, identification of normal anatomy and evaluation of tumor and the surrounding pseudocapsule was done. If the tumor was hard or surrounded by a thick pseudocapsule, dissection was performed along the outer margin of the pseudocapsule. If the tumor was fragile and soft enough, it was removed in a piecemeal fashion. Pseudocapsule was removed in all possible circumstances to prevent recurrence. Hormonal type of pituitary adenoma was confirmed with microscopic examination of specimen with immunohistochemistry of relevant pituitary hormones by a proficient pathologist.
Student's t-test was performed to evaluate the difference of average age and tumor size between the hyponatremia group and control. Chi-squared test was performed to evaluate the difference of distribution regarding sex, Hardy classification, hormonal type, preoperative hypopituitarism, tumor removal radicality, tumor removal fashion and hormonal outcome. In all statistical analyses, p-value of <0.05 was considered significant. Statistical analyses were performed using SPSS statistical software (version 15.0; SPSS Inc., Chicago, IL, USA).
Out of total 1,343 pituitary adenoma patients, we identified 93 patients (6.9%) with postoperative hyponatremia that required hypertonic saline infusion. Our routine daily laboratory evaluation includes water intake/output, serum electrolytes (sodium, potassium, chloride), urine specific gravity. Once hyponatremia was detected, 3% NaCl infusion was started immediately when the sodium level was below 130 mmol/L or patient showed significant relevant symptoms. Four hours after initiation of hypertonic saline, serum electrolyte levels were followed up and infusion rate was adjusted, which were repeated until we stabilized hyponatremia. For correction rate of hyponatremia, we stuck to the consensus guidelines [1718]. The initiation of hypertonic saline infusion, which can be considered as onset of hyponatremia, was 7.8 days after surgery (range: postoperative day 3–13) (Fig. 1). Initial mean serum sodium level right before starting hypertonic saline infusion was 124.3±4.8 mmol/L, ranging from 111 to 132 mmol/L. Patients required average 2.7 days (range: 1–9) of hypertonic saline infusion before resolution of symptoms and sustained recovery of serum sodium level.
Preoperative characteristics of 93 patients with postoperative hyponatremia are summarized in Table 1. The group included 21 males and 72 females with a mean age of 44.7 (range: 19–75) at the time of surgery. The proportion of female patients were significantly higher in the hyponatremia group compared to control (77.4% vs. 61.4%, p=0.02). The mean preoperative size of tumor was 19.7 mm (range: 5–55). We witnessed no significant difference in age (44.7 vs. 43.1, p=0.245) and tumor size (19.7 vs. 19.3, p=0.692). Patients were classified into modified Hardy's classification, resulting in 31 patients (33.3%) of type I, 15 patients (16.1%) of type II, 31 patients (33.3%) of type III and 16 patients (17.2%) of type IV. Regarding hormonal subtypes, nonfunctioning adenoma was the most common, numbering up to 41 patients (44.1%), followed by growth hormone-secreting (33 patients, 35.5%), prolactin-secreting (11 patients, 11.8%) and adrenocorticotropin-secreting subtype (8 patients, 8.6%). There was no significant difference of distribution regarding modified Hardy's classification, hormonal subtypes of adenomas. Among 1,299 patients who underwent preoperative CPFT, total 917 patients presented with preoperative hypopituitarism in at least one of their pituitary hormone axes. The incidence of preoperative hypopituitarism was lower in a hyponatremia group than in a control group (58.5% vs. 71.5%, p=0.007). However, the incidence of adrenocorticotropic hormone (ACTH) axis hypofunction was similar in both groups.
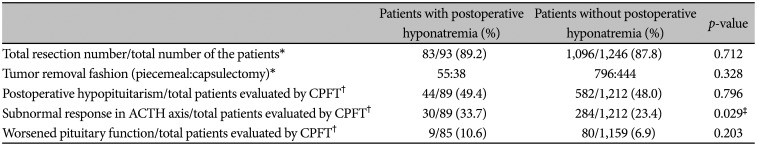
Postoperative outcomes are summarized in Table 2. Total resection was achieved in 83 patients (89.2%) of hyponatremia group. Tumors were removed in piecemeal fashion in 55 patients (59.1%) and in capsulectomy fashion in 38 patients (40.9%). There was no significant difference in total resection rate (89.2% vs. 87.8%, p=0.712) or capsulectomy rate (59.1% vs. 64.2%, p=0.328) between the two groups.
44 patients from hyponatremia group and 582 patients from control group (49.4% vs. 48.0%, p=0.796) showed postoperative hypopituitarism in at least one axis of anterior pituitary function. Incidence of pituitary function deterioration after surgery was evaluated with direct comparison of preoperative and postoperative CPFT, and no difference was found between the two groups (10.6% vs. 6.9%, p=0.203). On the contrary, decreased pituitary function in ACTH axis showed strong association with the presence of postoperative hyponatremia (33.7% vs. 23.4%, p=0.029).
Patients with hyponatremia present a variety of symptoms from simple headaches, muscle cramps to more severe symptoms like seizures, mental change, respiratory arrest [19]. Severe symptomatic hyponatremia should be managed immediately as delayed treatment can lead to fatal results. Although hyponatremia is a common postoperative complication of TSS, little is known about its time course and risk factors. The exact mechanism behind postoperative hyponatremia is still unknown; theories suggest excessive antidiuretic hormone (ADH) secretion after surgery induced by vagal stimuli (headache, stress, nausea) or immediate release of ADH into vascular space due to surgical exploration as possible mechanisms. Previous studies suggested old age [13], female sex [6], Cushing's disease [1020], tumor size [11], presence of transient diabetes insipidus as risk factors of postoperative hyponatremia. It is also reported that significant hyponatremia does not occur until the latter part of 1st postoperative week to early 2nd week [5789].
This study retrospectively reviewed data of 1,343 pituitary adenoma patients, of which 93 patients (6.9%) presented with postoperative hyponatremia. The incidence, onset time, and duration of postoperative hyponatremia presented in this study are consistent with previous reports. Various preoperative and postoperative characteristics were analyzed to identify any possible risk factor of hyponatremia. Although old age is a frequently reported risk factor for postoperative hyponatremia, there was no significant difference of average age between the hyponatremia group and the control group in this study. There was significant sex predilection towards female in hyponatremia group compared to control, which was also witnessed by other group [6]. Hormonal type of tumor was not related to postoperative hyponatremia. Factors related to higher risk of normal gland violation, such as bigger tumor size or suprasellar extension, was not associated with postoperative hyponatremia. As hyponatremia frequently associates with hypocortisolism [21], we speculated that the incidence of ACTH dysfunction would be higher in a hyponatremia group than in a control group, we failed to demonstrate a statistically significant difference in the incidence of preoperative ACTH dysfunction between two groups. It was an interesting finding that the incidence of hypopituitarism was lower in a hyponatremia group, which may suggest this complication may more frequently associates with patients with relatively well preserved pituitary functional capacity.
There was no difference in total removal rate or capsulectomy rate between the hyponatremia group and the control group. Postoperative hypopituitarism was not associated with postoperative hyponatremia, but postoperative ACTH dysfunction was strongly related to hyponatremia suggesting ACTH dysfunction may be the leading cause of hyponatremia.
Mechanism of postoperative hyponatremia in pituitary adenoma patients is still unclear. Some previous studies suggested that increased level of ADH after surgery may be a possible cause of postoperative hyponatremia [72223]. Unregulated ADH release from surgically manipulated posterior pituitary gland occurs few days after surgery, resulting in fluid retention and natriuresis [10]. This theory is in good agreement with the delayed onset of hyponatremia [723]. On the contrary, other studies have also reported cases of hyponatremia in the settings of normal or low level of ADH [9242526]. In this case, hyponatremia is explained by actions of atrial natriuretic hormones and brain natriuretic hormone resulting in excessive natriuresis rather than fluid retention, also known as cerebral salt wasting syndrome. As aforementioned, another possible mechanism for delayed hyponatremia is secondary adrenocortical insufficiency [927]. Although the present study showed no correlation between hormonal type of pituitary adenoma and postoperative hyponatremia, some previous studies have reported higher risks of hyponatremia after TSS in Cushing patients compared to prolactinoma by 3 folds, or non-functioning adenomas by 2.8 folds [24]. This study showed that postoperative adrenal insufficiency can be a risk factor for delayed hyponatremia. It should be further investigated whether adequate cortisol replacement could prevent development of postoperative hyponatremia.
This study defined hyponatremia group as patients with postoperative hyponatremia with symptoms severe enough to need hypertonic saline infusion. Overall incidence of hyponatremia might have been underrated compared to previous studies. We were not able to provide a reasonable explanation for the female predilection reported in hyponatremia group. Although some of the previous studies have reported the same, none had success in providing a meaningful discussion. This study did not include any analysis regarding the mechanism of hyponatremia such as serum ADH level or urine sodium level. The hyponatremia group we set might be heterogeneous, including different types of hyponatremia. The primary endpoint of this study was to seek for any clinical factor relevant to postoperative hyponatremia. Further study regarding the mechanism behind postoperative hyponatremia needs to be done.
In conclusion, postoperative hyponatremia is a common complication after TSS. Recognition of its risk factors is essential since delayed detection or treatment can lead to critical results. While tumor size, modified Hardy classification and tumor removal radicality were not associated with delayed hyponatremia, we demonstrated delayed postoperative hyponatremia was more prevalent in female and patients with perioperative pituitary dysfunction, rendering them as possible risk factors.
Acknowledgments
The authors thank Juyoon Park, RN, MPH, OCN, Min Kyeong Jang, PhD, RN, KOAPN, Sung Ja Kang, RN, Sujin Ryu, RN, Young Hwa Yim, MSN, RN, KCAPN, Yong Jun Jang, RN for their tremendous effort in performing the endocrinological tests and data acquisition for such a long follow-up duration.
This study was supported by a faculty research grant of Yonsei University College of Medicine (6-2020-0224).
References
1. Olson BR, Gumowski J, Rubino D, Oldfield EH. Pathophysiology of hyponatremia after transsphenoidal pituitary surgery. J Neurosurg. 1997; 87:499–507. PMID: 9322839.
2. Olson BR, Rubino D, Gumowski J, Oldfield EH. Isolated hyponatremia after transsphenoidal pituitary surgery. J Clin Endocrinol Metab. 1995; 80:85–91. PMID: 7829644.
3. Sane T, Rantakari K, Poranen A, Tähtelä R, Välimäki M, Pelkonen R. Hyponatremia after transsphenoidal surgery for pituitary tumors. J Clin Endocrinol Metab. 1994; 79:1395–1398. PMID: 7962334.
4. Sata A, Hizuka N, Kawamata T, Hori T, Takano K. Hyponatremia after transsphenoidal surgery for hypothalamo-pituitary tumors. Neuroendocrinology. 2006; 83:117–122. PMID: 16864995.
5. Taylor SL, Tyrrell JB, Wilson CB. Delayed onset of hyponatremia after transsphenoidal surgery for pituitary adenomas. Neurosurgery. 1995; 37:649–653. discussion 653–4. PMID: 8559292.
6. Zada G, Liu CY, Fishback D, Singer PA, Weiss MH. Recognition and management of delayed hyponatremia following transsphenoidal pituitary surgery. J Neurosurg. 2007; 106:66–71. PMID: 17236489.
7. Cusick JF, Hagen TC, Findling JW. Inappropriate secretion of antidiuretic hormone after transsphenoidal surgery for pituitary tumors. N Engl J Med. 1984; 311:36–38. PMID: 6727963.
8. Murty GE, Lamballe P, Welch AR. Early inappropriate secretion of antidiuretic hormone after trans-sphenoidal pituitary adenomectomy. J Laryngol Otol. 1990; 104:894–895. PMID: 2266317.
9. Whitaker SJ, Meanock CI, Turner GF, et al. Fluid balance and secretion of antidiuretic hormone following transsphenoidal pituitary surgery. A preliminary series. J Neurosurg. 1985; 63:404–412. PMID: 4020468.
10. Hensen J, Henig A, Fahlbusch R, Meyer M, Boehnert M, Buchfelder M. Prevalence, predictors and patterns of postoperative polyuria and hyponatraemia in the immediate course after transsphenoidal surgery for pituitary adenomas. Clin Endocrinol (Oxf). 1999; 50:431–439. PMID: 10468901.
11. Kelly DF, Laws ER Jr, Fossett D. Delayed hyponatremia after transsphenoidal surgery for pituitary adenoma. Report of nine cases. J Neurosurg. 1995; 83:363–367. PMID: 7616287.
12. Kinoshita Y, Tominaga A, Arita K, et al. Post-operative hyponatremia in patients with pituitary adenoma: post-operative management with a uniform treatment protocol. Endocr J. 2011; 58:373–379. PMID: 21467692.
13. Lee JI, Cho WH, Choi BK, Cha SH, Song GS, Choi CH. Delayed hyponatremia following transsphenoidal surgery for pituitary adenoma. Neurol Med Chir (Tokyo). 2008; 48:489–492. discussion 492–4. PMID: 19029775.
14. Kim EH, Oh MC, Kim SH. Application of low-field intraoperative magnetic resonance imaging in transsphenoidal surgery for pituitary adenomas: technical points to improve the visibility of the tumor resection margin. Acta Neurochir (Wien). 2013; 155:485–493. PMID: 23318686.
15. Kim J, Yoon SJ, Moon JH, et al. Clinical significance of radical surgery in the treatment of silent corticotroph adenoma. J Korean Neurosurg Soc. 2019; 62:114–122. PMID: 30630298.
16. Oh MC, Kim EH, Kim SH. Coexistence of intracranial aneurysm in 800 patients with surgically confirmed pituitary adenoma. J Neurosurg. 2012; 116:942–947. PMID: 22304451.
17. Spasovski G, Vanholder R, Allolio B, et al. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Nephrol Dial Transplant. 2014; 29 Suppl 2:i1–i39.
18. Verbalis JG, Goldsmith SR, Greenberg A, et al. Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. Am J Med. 2013; 126(10 Suppl 1):S1–S42.
19. Adrogué HJ, Madias NE. Hyponatremia. N Engl J Med. 2000; 342:1581–1589. PMID: 10824078.
20. Adams JR, Blevins LS Jr, Allen GS, Verity DK, Devin JK. Disorders of water metabolism following transsphenoidal pituitary surgery: a single institution's experience. Pituitary. 2006; 9:93–99. PMID: 16832585.
21. Raff H. Glucocorticoid inhibition of neurohypophysial vasopressin secretion. Am J Physiol. 1987; 252(4 Pt 2):R635–R644. PMID: 3032001.
22. Lester MC, Nelson PB. Neurological aspects of vasopressin release and the syndrome of inappropriate secretion of antidiuretic hormone. Neurosurgery. 1981; 8:735–740. PMID: 7279164.
23. Ultmann MC, Hoffman GE, Nelson PB, Robinson AG. Transient hyponatremia after damage to the neurohypophyseal tracts. Neuroendocrinology. 1992; 56:803–811. PMID: 1369588.
24. Diringer M, Ladenson PW, Borel C, Hart GK, Kirsch JR, Hanley DF. Sodium and water regulation in a patient with cerebral salt wasting. Arch Neurol. 1989; 46:928–930. PMID: 2757534.
25. Kern PA, Robbins RJ, Bichet D, Berl T, Verbalis JG. Syndrome of inappropriate antidiuresis in the absence of arginine vasopressin. J Clin Endocrinol Metab. 1986; 62:148–152. PMID: 3940262.
26. Wijdicks EF, Ropper AH, Hunnicutt EJ, Richardson GS, Nathanson JA. Atrial natriuretic factor and salt wasting after aneurysmal subarachnoid hemorrhage. Stroke. 1991; 22:1519–1524. PMID: 1835809.
27. Kröll M, Juhler M, Lindholm J. Hyponatraemia in acute brain disease. J Intern Med. 1992; 232:291–297. PMID: 1328460.
Fig. 1
Characteristics of hyponatremia patients. A: Initial serum sodium level right at the onset of postoperative hyponatremia is 124.3±4.8 mmol/L. B: Onset of delayed hyponatremia is 7.8 days after surgery. C: Hypertonic saline infusion is administrated for 2.7 days.
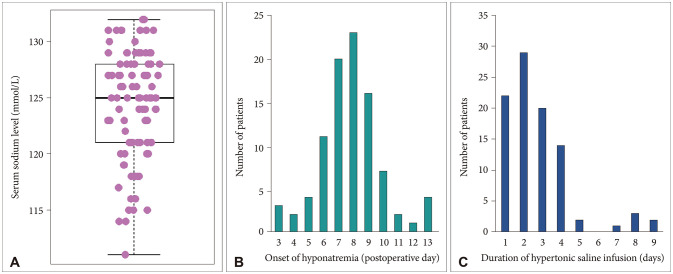
Table 1
Preoperative characteristics of patients
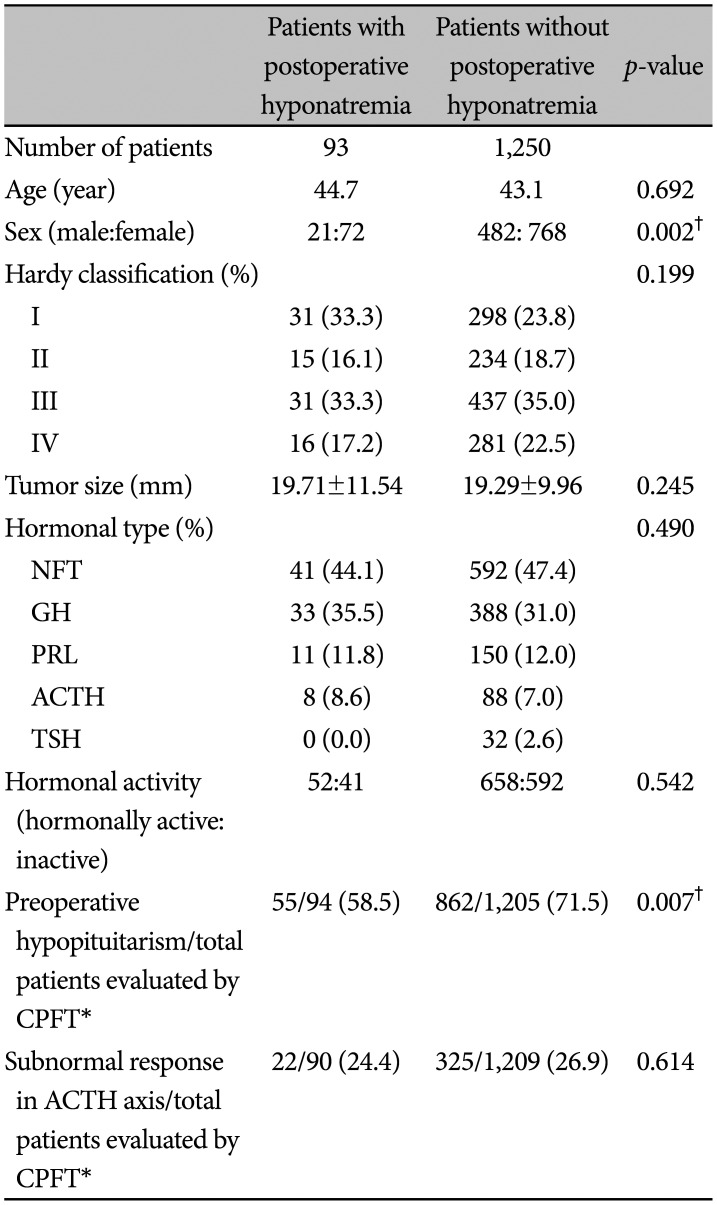
Table 2
Postoperative outcomes of the patients




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download