Abstract
Background/Aims
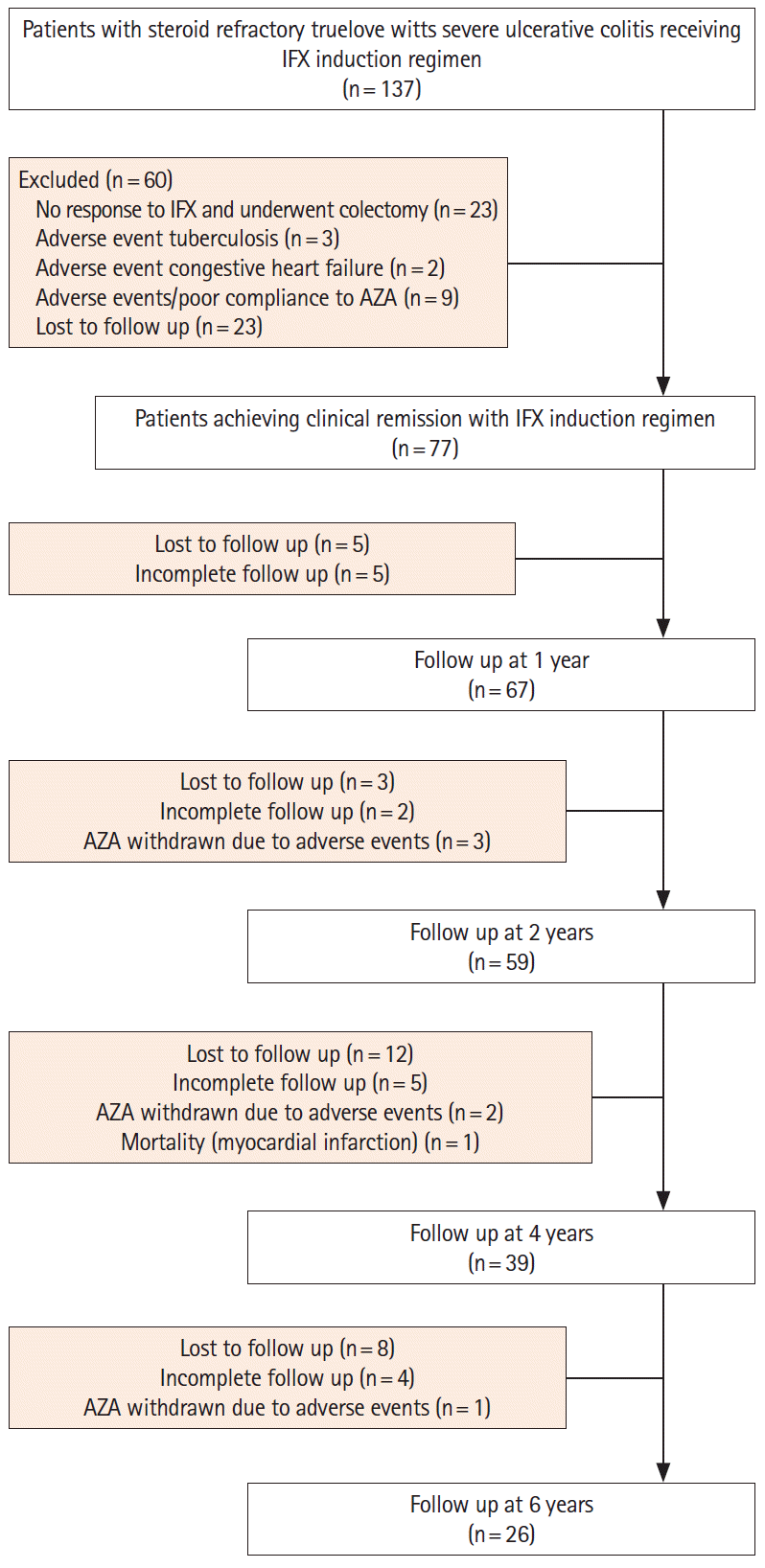
Methods
Results
Notes
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
Sood A is an editorial board member of the journal but was not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflicts of interest relevant to this article were reported.
Author Contribution
Conceptualization: Mahajan R, Midha V, Sood A. Data curation: Kedia S, Kaur K, Sahu P, Singh D, Ahuja V. Formal analysis: Mahajan R, Singh A, Kaur K, Bansal N, Dharni K. Investigation: Mahajan R, Singh A, Kedia S, Kaur K, Singh D, Sood A. Methodology: Mahajan R, Singh A, Kedia S, Kaur K, Midha V, Sahu P, Mehta V, Kaushal S, Ahuja V, Sood A. Project administration: Mahajan R, Kedia S, Sood A. Resources: Midha V, Singh D, Sood A. Software: Singh A, Bansal N, Dharni K. Supervision: Mahajan R, Kedia S, Midha V, Ahuja V, Sood A. Visualization: Mahajan R, Singh A, Kedia S. Writing - original draft: Mahajan R, Singh A, Kedia S, Kaur K, Midha V, Sahu P, Mehta V, Ahuja V, Sood A. Writing - review & editing: Mahajan R, Singh A, Kedia S, Kaur K, Midha V, Sahu P, Mehta V, Bansal N, Dharni K, Kaushal S, Ahuja V, Sood A. Approval of final manuscript: all authors.
REFERENCES
Fig. 2.
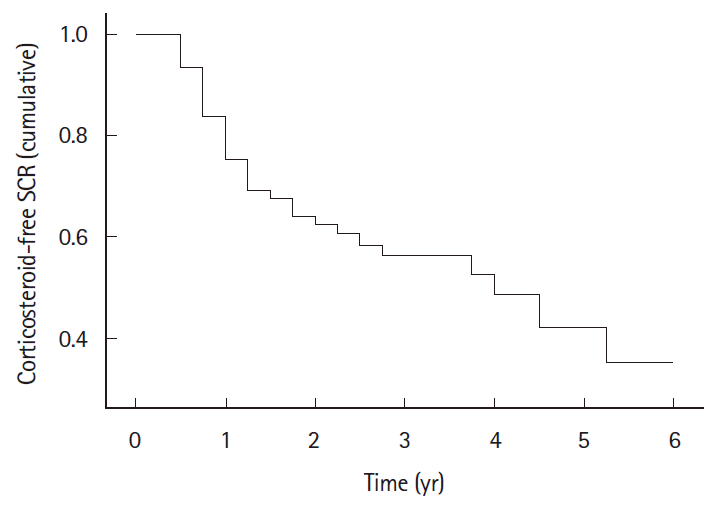
Table 1.
Table 2.
| Year | No. of patients with single relapse | No. of patients with > 1 relapse | No. of relapses |
|---|---|---|---|
| 1 | 14a | 8a | 31 |
| 2 | 4b + 4a | 2b + 1a | 14 |
| 3 | 2b + 1a | 2b + 1a | 9 |
| 4 | 3b | 2a | 7 |
| 5 | 2a | 1b | 4 |
| 6 | 1b | - | 2 |
| Total | 67 | ||
| Treatment | Corticosteroids: 64 Infliximab: 2 (1 patient each in year 1 and 2) Colectomy: 1 (year 3) |
Table 3.
Table 4.
| Maintenance therapy with 5-ASA+AZA | INR (US$) | Maintenance therapy with IFX | INR (US$) |
|---|---|---|---|
| Number of days AZA +5-ASA consumed | 365 | Number of IFX doses needed per patient if used for maintenance of remission | 6 |
| Daily cost of AZA (100 mg/day) +5-ASA (4.8 g/day) | 140 (1.91) | Annual cost of IFX maintenance therapy for the entire cohort (C) | 27,720,000 (3,79,726) |
| Annual cost of maintenance therapy with 5-ASA and AZA for the entire cohort (A) | 39,34,700 (53,900) | Cost of hospitalization and losses incurred due to absence from worka (D) | 415,800 (5,695) |
| Total number of relapses observed at 1 year | 31 | Expected number of relapses on IFX therapy over the entire cohortb | 19 |
| Cost incurred on treatment of relapses with corticosteroids | 7,500 (103) (n = 30, corticosteroids tapered over 16 weeks) | - | - |
| Cost incurred on treatment of relapses with IFX induction regimen | 180,000 (2,465) (n = 1, IFX administered at a dose of 5 mg/kg at 0, 2, 6 weeks) | - | - |
| Cost incurred on colectomy | - | Cost incurred on treatment of relapse with colectomy in the entire cohortc (E) | 1,425,000 (19,520) |
| Total cost incurred on management of relapses in the entire cohort (B) | 187,500 (2,568) | ||
| Total cost incurred for the entire cohort (A+B) | 4,122,200 (56,468) | Total cost incurred for the entire cohort (C+D+E) | 29,560,800 (404,942) |
| Average cost per person per year | 53,535 (733) | Average cost per person per year | 383,906 (5,259) |
All values are approximate 1US$ (INR 73). Annual cost difference per patient: INR 330,371 (US$ 4,526). Cohort size (n)=77; Cost of IFX originator: INR 20,000/100 mg; Cost of AZA 100 mg: INR 20; Cost of oral 5-ASA 4.8 g: INR 120; Cost of colectomy in a tertiary care center: INR 75,000; Costs calculated for average body weight of 60 kg.
a For a single day care admission approximate cost of hospitalization: INR 500 for a tertiary care hospital (data derived from World Health Organization report on Estimates of Unit Costs for Patient Services for India; available at https://www.who.int/choice/country/ind/cost/en/, accessed on October 18, 2020) and approximate loss of income due to absence from work: INR 370 (data derived from per capita income in India for 2018-2019; available at http://mospi.gov.in, accessed on October 18, 2020).
b Calculated considering average relapse rates of 25% and 30% at 1 year and 2 years respectively. [8,17,23,24]




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download