1. GBD 2019 Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021; 20:795–820.
2. O’Donnell MJ, Xavier D, Liu L, Zhang H, Chin SL, Rao-Melacini P, et al. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case-control study. Lancet. 2010; 376:112–123.
3. International Diabetes Federation. IDF Diabetes Atlas. Diabetes around the world in 2021 [Internet]. Brussels: International Diabetes Federation;2021. [cited 2021 Nov 12]. Available from:
https://diabetesatlas.org.
4. Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M, et al. 2021 ESC guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J. 2021; 42:3227–3337.
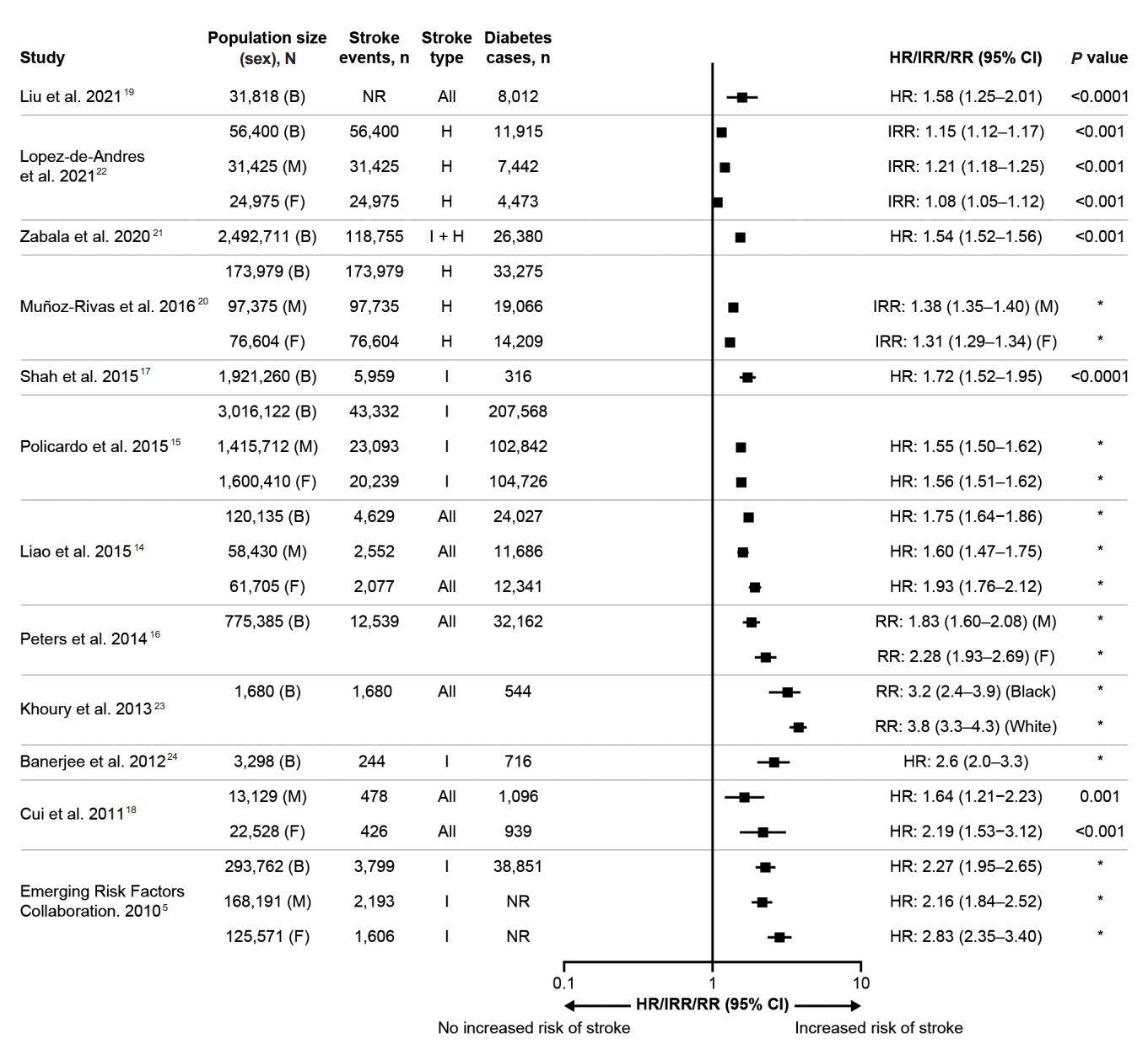
5. Emerging Risk Factors Collaboration. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet. 2010; 375:2215–2222.
6. Sun Y, Toh MP. Impact of diabetes mellitus (DM) on the health-care utilization and clinical outcomes of patients with stroke in Singapore. Value Health. 2009; 12 Suppl 3:S101–S105.
7. Eriksson M, Carlberg B, Eliasson M. The disparity in long-term survival after a first stroke in patients with and without diabetes persists: the Northern Sweden MONICA study. Cerebrovasc Dis. 2012; 34:153–160.
8. Zhang L, Li X, Wolfe CDA, O’Connell MDL, Wang Y. Diabetes as an independent risk factor for stroke recurrence in ischemic stroke patients: an updated meta-analysis. Neuroepidemiology. 2021; 55:427–435.
9. Lo JW, Crawford JD, Samaras K, Desmond DW, Köhler S, Staals J, et al. Association of prediabetes and type 2 diabetes with cognitive function after stroke: a STROKOG collaboration study. Stroke. 2020; 51:1640–1646.
10. Echouffo-Tcheugui JB, Xu H, Matsouaka RA, Xian Y, Schwamm LH, Smith EE, et al. Diabetes and long-term outcomes of ischaemic stroke: findings from get with the guidelines-stroke. Eur Heart J. 2018; 39:2376–2386.
11. Li HW, Yang MC, Chung KP. Predictors for readmission of acute ischemic stroke in Taiwan. J Formos Med Assoc. 2011; 110:627–633.
12. Kleindorfer DO, Towfighi A, Chaturvedi S, Cockroft KM, Gutierrez J, Lombardi-Hill D, et al. 2021 guideline for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline from the American Heart Association/American Stroke Association. Stroke. 2021; 52:e364–e467.
13. Lau LH, Lew J, Borschmann K, Thijs V, Ekinci EI. Prevalence of diabetes and its effects on stroke outcomes: a meta-analysis and literature review. J Diabetes Investig. 2019; 10:780–792.
14. Liao CC, Shih CC, Yeh CC, Chang YC, Hu CJ, Lin JG, et al. Impact of diabetes on stroke risk and outcomes: two nationwide retrospective cohort studies. Medicine (Baltimore). 2015; 94:e2282.
15. Policardo L, Seghieri G, Francesconi P, Anichini R, Franconi F, Seghieri C, et al. Gender difference in diabetes-associated risk of first-ever and recurrent ischemic stroke. J Diabetes Complications. 2015; 29:713–717.
16. Peters SA, Huxley RR, Woodward M. Diabetes as a risk factor for stroke in women compared with men: a systematic review and meta-analysis of 64 cohorts, including 775,385 individuals and 12,539 strokes. Lancet. 2014; 383:1973–1980.
17. Shah AD, Langenberg C, Rapsomaniki E, Denaxas S, Pujades-Rodriguez M, Gale CP, et al. Type 2 diabetes and incidence of cardiovascular diseases: a cohort study in 1·9 million people. Lancet Diabetes Endocrinol. 2015; 3:105–113.
18. Cui R, Iso H, Yamagishi K, Saito I, Kokubo Y, Inoue M, et al. Diabetes mellitus and risk of stroke and its subtypes among Japanese: the Japan public health center study. Stroke. 2011; 42:2611–2614.
19. Liu Y, Li J, Dou Y, Ma H. Impacts of type 2 diabetes mellitus and hypertension on the incidence of cardiovascular diseases and stroke in China real-world setting: a retrospective cohort study. BMJ Open. 2021; 11:e053698.
20. Muñoz-Rivas N, Méndez-Bailón M, Hernández-Barrera V, de Miguel-Yanes JM, Jimenez-Garcia R, Esteban-Hernández J, et al. Type 2 diabetes and hemorrhagic stroke: a population-based study in Spain from 2003 to 2012. J Stroke Cerebrovasc Dis. 2016; 25:1431–1443.
21. Zabala A, Darsalia V, Holzmann MJ, Franzén S, Svensson AM, Eliasson B, et al. Risk of first stroke in people with type 2 diabetes and its relation to glycaemic control: a nationwide observational study. Diabetes Obes Metab. 2020; 22:182–190.
22. Lopez-de-Andres A, Jimenez-Garcia R, Hernández-Barrera V, Jiménez-Trujillo I, de Miguel-Yanes JM, Carabantes-Alarcon D, et al. Sex-related disparities in the incidence and outcomes of hemorrhagic stroke among type 2 diabetes patients: a propensity score matching analysis using the Spanish National Hospital Discharge Database for the period 2016-18. Cardiovasc Diabetol. 2021; 20:138.
23. Khoury JC, Kleindorfer D, Alwell K, Moomaw CJ, Woo D, Adeoye O, et al. Diabetes mellitus: a risk factor for ischemic stroke in a large biracial population. Stroke. 2013; 44:1500–1504.
24. Banerjee C, Moon YP, Paik MC, Rundek T, Mora-McLaughlin C, Vieira JR, et al. Duration of diabetes and risk of ischemic stroke: the Northern Manhattan Study. Stroke. 2012; 43:1212–1217.
25. American Diabetes Association Professional Practice Committee. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2022. Diabetes Care. 2021; 45(Suppl 1):S17–S38.
26. Lee M, Saver JL, Hong KS, Song S, Chang KH, Ovbiagele B. Effect of pre-diabetes on future risk of stroke: meta-analysis. BMJ. 2012; 344:e3564.
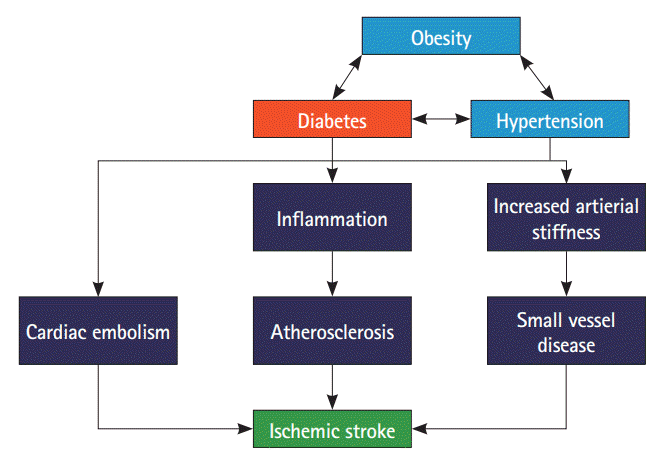
27. Petrie JR, Guzik TJ, Touyz RM. Diabetes, hypertension, and cardiovascular disease: clinical insights and vascular mechanisms. Can J Cardiol. 2018; 34:575–584.
28. Chen R, Ovbiagele B, Feng W. Diabetes and stroke: epidemiology, pathophysiology, pharmaceuticals and outcomes. Am J Med Sci. 2016; 351:380–386.
29. Clarkson P, Celermajer DS, Donald AE, Sampson M, Sorensen KE, Adams M, et al. Impaired vascular reactivity in insulin-dependent diabetes mellitus is related to disease duration and low density lipoprotein cholesterol levels. J Am Coll Cardiol. 1996; 28:573–579.
30. Kamel H, Healey JS. Cardioembolic stroke. Circ Res. 2017; 120:514–526.
31. Kim JS, Nah HW, Park SM, Kim SK, Cho KH, Lee J, et al. Risk factors and stroke mechanisms in atherosclerotic stroke: intracranial compared with extracranial and anterior compared with posterior circulation disease. Stroke. 2012; 43:3313–3318.
32. Pasi M, Cordonnier C. Clinical relevance of cerebral small vessel diseases. Stroke. 2020; 51:47–53.
33. Yaghi S, Prabhakaran S, Khatri P, Liebeskind DS. Intracranial atherosclerotic disease. Stroke. 2019; 50:1286–1293.
34. Poznyak A, Grechko AV, Poggio P, Myasoedova VA, Alfieri V, Orekhov AN. The diabetes mellitus–atherosclerosis connection: the role of lipid and glucose metabolism and chronic inflammation. Int J Mol Sci. 2020; 21:1835.
35. King DE, Mainous AG 3rd, Buchanan TA, Pearson WS. C-reactive protein and glycemic control in adults with diabetes. Diabetes Care. 2003; 26:1535–1539.
36. van Sloten TT, Sedaghat S, Carnethon MR, Launer LJ, Stehouwer CDA. Cerebral microvascular complications of type 2 diabetes: stroke, cognitive dysfunction, and depression. Lancet Diabetes Endocrinol. 2020; 8:325–336.
37. Creager MA, Lüscher TF, Cosentino F, Beckman JA. Diabetes and vascular disease: pathophysiology, clinical consequences, and medical therapy: part I. Circulation. 2003; 108:1527–1532.
38. Goette A, Lendeckel U. Atrial cardiomyopathy: pathophysiology and clinical consequences. Cells. 2021; 10:2605.
39. Seyed Ahmadi S, Svensson AM, Pivodic A, Rosengren A, Lind M. Risk of atrial fibrillation in persons with type 2 diabetes and the excess risk in relation to glycaemic control and renal function: a Swedish cohort study. Cardiovasc Diabetol. 2020; 19:9.
40. Bohne LJ, Johnson D, Rose RA, Wilton SB, Gillis AM. The association between diabetes mellitus and atrial fibrillation: clinical and mechanistic insights. Front Physiol. 2019; 10:135.
41. Kruyt ND, Biessels GJ, Devries JH, Roos YB. Hyperglycemia in acute ischemic stroke: pathophysiology and clinical management. Nat Rev Neurol. 2010; 6:145–155.
42. Maida CD, Daidone M, Pacinella G, Norrito RL, Pinto A, Tuttolomondo A. Diabetes and ischemic stroke: an old and new relationship an overview of the close interaction between these diseases. Int J Mol Sci. 2022; 23:2397.
43. Capes SE, Hunt D, Malmberg K, Pathak P, Gerstein HC. Stress hyperglycemia and prognosis of stroke in nondiabetic and diabetic patients: a systematic overview. Stroke. 2001; 32:2426–2432.
44. Baird TA, Parsons MW, Phan T, Butcher KS, Desmond PM, Tress BM, et al. Persistent poststroke hyperglycemia is independently associated with infarct expansion and worse clinical outcome. Stroke. 2003; 34:2208–2214.
45. Johnston KC, Bruno A, Pauls Q, Hall CE, Barrett KM, Barsan W, et al. Intensive vs standard treatment of hyperglycemia and functional outcome in patients with acute ischemic stroke: the SHINE randomized clinical trial. JAMA. 2019; 322:326–335.
46. Fuentes B, Castillo J, San José B, Leira R, Serena J, Vivancos J, et al. The prognostic value of capillary glucose levels in acute stroke: the GLycemia in Acute Stroke (GLIAS) study. Stroke. 2009; 40:562–568.
47. Olaiya MT, Cadilhac DA, Kim J, Thrift AG, Courten B, Andrew NE, et al. Quality of care and one-year outcomes in patients with diabetes hospitalised for stroke or TIA: a linked registry study. J Stroke Cerebrovasc Dis. 2021; 30:106083.
48. MacDougal EL, Herman WH, Wing JJ, Morgenstern LB, Lisabeth LD. Diabetes and ischaemic stroke outcome. Diabet Med. 2018; 35:1249–1257.
49. Kamalesh M, Shen J, Eckert GJ. Long term postischemic stroke mortality in diabetes: a veteran cohort analysis. Stroke. 2008; 39:2727–2731.
50. Alter M, Lai SM, Friday G, Singh V, Kumar VM, Sobel E. Stroke recurrence in diabetics. Does control of blood glucose reduce risk? Stroke. 1997; 28:1153–1157.
51. Kalaria RN, Akinyemi R, Ihara M. Stroke injury, cognitive impairment and vascular dementia. Biochim Biophys Acta. 2016; 1862:915–925.
52. Shukla V, Shakya AK, Perez-Pinzon MA, Dave KR. Cerebral ischemic damage in diabetes: an inflammatory perspective. J Neuroinflammation. 2017; 14:21.
53. Gæde P, Oellgaard J, Kruuse C, Rossing P, Parving HH, Pedersen O. Beneficial impact of intensified multifactorial intervention on risk of stroke: outcome of 21 years of follow-up in the randomised Steno-2 Study. Diabetologia. 2019; 62:1575–1580.
54. Gyldenkerne C, Kahlert J, Olesen KKW, Thrane PG, Sørensen HT, Thomsen RW, et al. Twenty-year temporal trends in risk of ischemic stroke in incident type 2 diabetes: a Danish population-based cohort study. Diabetes Care. 2022; 45:2144–2151.
55. Benn M, Emanuelsson F, Tybjærg-Hansen A, Nordestgaard BG. Impact of high glucose levels and glucose lowering on risk of ischaemic stroke: a Mendelian randomisation study and meta-analysis. Diabetologia. 2021; 64:1492–1503.
56. Sattar N, Lee MMY, Kristensen SL, Branch KRH, Del Prato S, Khurmi NS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol. 2021; 9:653–662.
57. Gæde P, Oellgaard J, Carstensen B, Rossing P, Lund-Andersen H, Parving HH, et al. Years of life gained by multifactorial intervention in patients with type 2 diabetes mellitus and microalbuminuria: 21 years follow-up on the Steno-2 randomised trial. Diabetologia. 2016; 59:2298–2307.
58. Røder ME. Major adverse cardiovascular event reduction with GLP-1 and SGLT2 agents: evidence and clinical potential. Ther Adv Chronic Dis. 2018; 9:33–50.
59. Buse JB, Wexler DJ, Tsapas A, Rossing P, Mingrone G, Mathieu C, et al. 2019 update to: management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia. 2020; 63:221–228.
60. American Diabetes Association Professional Practice Committee. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2022. Diabetes Care. 2022; 45(Suppl 1):S125–S143.
61. Kernan WN, Viscoli CM, Furie KL, Young LH, Inzucchi SE, Gorman M, et al. Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med. 2016; 374:1321–1331.
62. Patoulias DI, Boulmpou A, Teperikidis E, Katsimardou A, Siskos F, Doumas M, et al. Cardiovascular efficacy and safety of dipeptidyl peptidase-4 inhibitors: a meta-analysis of cardiovascular outcome trials. World J Cardiol. 2021; 13:585–592.
63. Rados DV, Falcetta MRR, Pinto LC, Leitão CB, Gross JL. All-cause mortality and cardiovascular safety of basal insulin treatment in patients with type 2 diabetes mellitus: a systematic review with meta-analysis and trial sequential analysis. Diabetes Res Clin Pract. 2021; 173:108688.
64. Dormandy JA, Charbonnel B, Eckland DJ, Erdmann E, MassiBenedetti M, Moules IK, et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet. 2005; 366:1279–1289.
65. Wilcox R, Bousser MG, Betteridge DJ, Schernthaner G, Pirags V, Kupfer S, et al. Effects of pioglitazone in patients with type 2 diabetes with or without previous stroke: results from PROactive (PROspective pioglitAzone Clinical Trial In macroVascular Events 04). Stroke. 2007; 38:865–873.
66. Verma S, Poulter NR, Bhatt DL, Bain SC, Buse JB, Leiter LA, et al. Effects of liraglutide on cardiovascular outcomes in patients with type 2 diabetes mellitus with or without history of myocardial infarction or stroke. Circulation. 2018; 138:2884–2894.
67. Leiter LA, Bain SC, Hramiak I, Jódar E, Madsbad S, Gondolf T, et al. Cardiovascular risk reduction with once-weekly semaglutide in subjects with type 2 diabetes: a post hoc analysis of gender, age, and baseline CV risk profile in the SUSTAIN 6 trial. Cardiovasc Diabetol. 2019; 18:73.
68. Nauck MA, Meier JJ, Cavender MA, Abd El Aziz M, Drucker DJ. Cardiovascular actions and clinical outcomes with glucagonlike peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Circulation. 2017; 136:849–870.
69. Nauck MA, Quast DR. Cardiovascular safety and benefits of semaglutide in patients with type 2 diabetes: findings from SUSTAIN 6 and PIONEER 6. Front Endocrinol (Lausanne). 2021; 12:645566.
70. McGuire DK, Shih WJ, Cosentino F, Charbonnel B, Cherney DZI, Dagogo-Jack S, et al. Association of SGLT2 inhibitors with cardiovascular and kidney outcomes in patients with type 2 diabetes: a meta-analysis. JAMA Cardiol. 2021; 6:148–158.
71. Goldenberg RM, Cheng AYY, Fitzpatrick T, Gilbert JD, Verma S, Hopyan JJ. Benefits of GLP-1 (glucagon-like peptide 1) receptor agonists for stroke reduction in type 2 diabetes: a call to action for neurologists. Stroke. 2022; 53:1813–1822.
72. Pan Y, Wang Y, Li H, Gaisano HY, Wang Y, He Y. Association of diabetes and prognosis of minor stroke and its subtypes: a prospective observational study. PLoS One. 2016; 11:e0153178.
73. Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Køber LV, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med. 2015; 373:2247–2257.
74. Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JF, Nauck MA, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016; 375:311–322.
75. Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016; 375:1834–1844.
76. Holman RR, Bethel MA, Mentz RJ, Thompson VP, Lokhnygina Y, Buse JB, et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2017; 377:1228–1239.
77. Hernandez AF, Green JB, Janmohamed S, D’Agostino RB Sr, Granger CB, Jones NP, et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (harmony outcomes): a double-blind, randomised placebo-controlled trial. Lancet. 2018; 392:1519–1529.
78. Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet. 2019; 394:121–130.
79. Husain M, Birkenfeld AL, Donsmark M, Dungan K, Eliaschewitz FG, Franco DR, et al. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2019; 381:841–851.
80. Gerstein HC, Sattar N, Rosenstock J, Ramasundarahettige C, Pratley R, Lopes RD, et al. Cardiovascular and renal outcomes with efpeglenatide in type 2 diabetes. N Engl J Med. 2021; 385:896–907.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download