Abstract
Purpose
Materials and Methods
Results
Notes
Ethical Statement
The protocol was reviewed and approved by the institutional review board of each participating center, and all patients provided written informed consent before treatment initiation; Gachon University Gil Medical Center: GCIRB2015-74, Gyeongsang National University Hospital: GNUH 2015-02-012, Dongsan Medical Center: DSMC 2015-03-015, Korea University Anam Hospital: ED15-024(AN15024-001), Kosin University Gospel Hospital: KUGH 2017-09-008, National Cancer Center: NCC 2015-0091, Dong-A University Medical Center: DMC 15-037, Pusan National University Hospital: H-1701-011-051, Samsung Medical Center: SMC 2014-07-181, Seoul National University Hospital: H-1504-119-667, Asan Medical Center: S2015-0302-0001, Soonchunhyang University Hospital: SCHUH 2015-05-007, Soonchunhyang University Bucheon Hospital: SCHBC 2015-07-008, Wonju Severance Christian Hospital: CR315002-002, Ajou University Hospital: AJIRB-MED-OBS-14-254, Yeungnam University Medical Center: YUMC 2015-03-016, Korea Cancer Center Hospital: K-1409-001-001, Ulsan University Hospital: UUH 2015-11-007, Inje University Sanggye Paik Hospital: SGPAIK 2015-06-002, Inje University Ilsan Paik Hospital: ISPAIK 2015-07-002, Inje University Busan Paik Hospital: BS 2015-0110, Jeonbuk National University Hospital: CUH 2015-02-034, Chonnam National University Hwasun Hospital: TMP-2015-023, Chung-Ang University Hospital: C2015048(1506), Chungnam National University Hospital: CNUH 2015-10-060, Chungbuk National University Hospital: CBNUH 2015-03-006, Hallym University Sacred Heart Hospital: HUH 2015-I043.
Author Contributions
Conceived and designed the analysis: Yi JH, Jeong SH, Kim SJ, Yoon DH, Koh Y, Kim JS.
Collected the data: Lee WS, Yang DH, Do YR, Kim MK, Yoo KH, Choi YS, Yun WJ, Park Y, Jo JC, Park BB, Kang HJ, Yun WJ, Kwon JH, Hong DS, Lee HS, Lee GW.
Contributed data or analysis tools: Eom HS, Kwak JY, Shin HJ, Yi SY, Oh SY, Kim HJ, Sohn BS, Won JH.
Performed the analysis: Yi JH, Suh C.
Wrote the paper: Yi JH, Kim WS.
References
Fig. 1
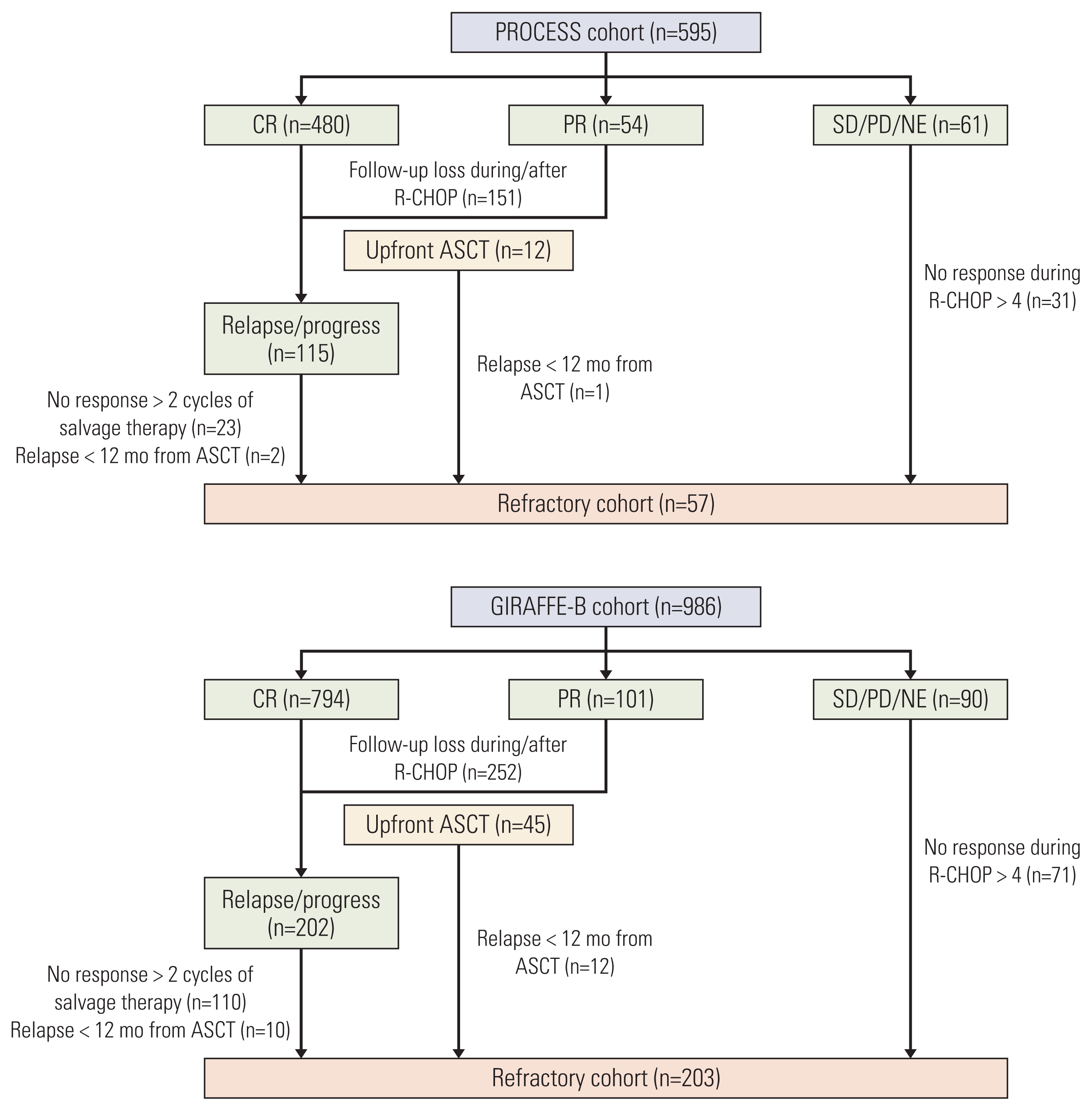
Fig. 2
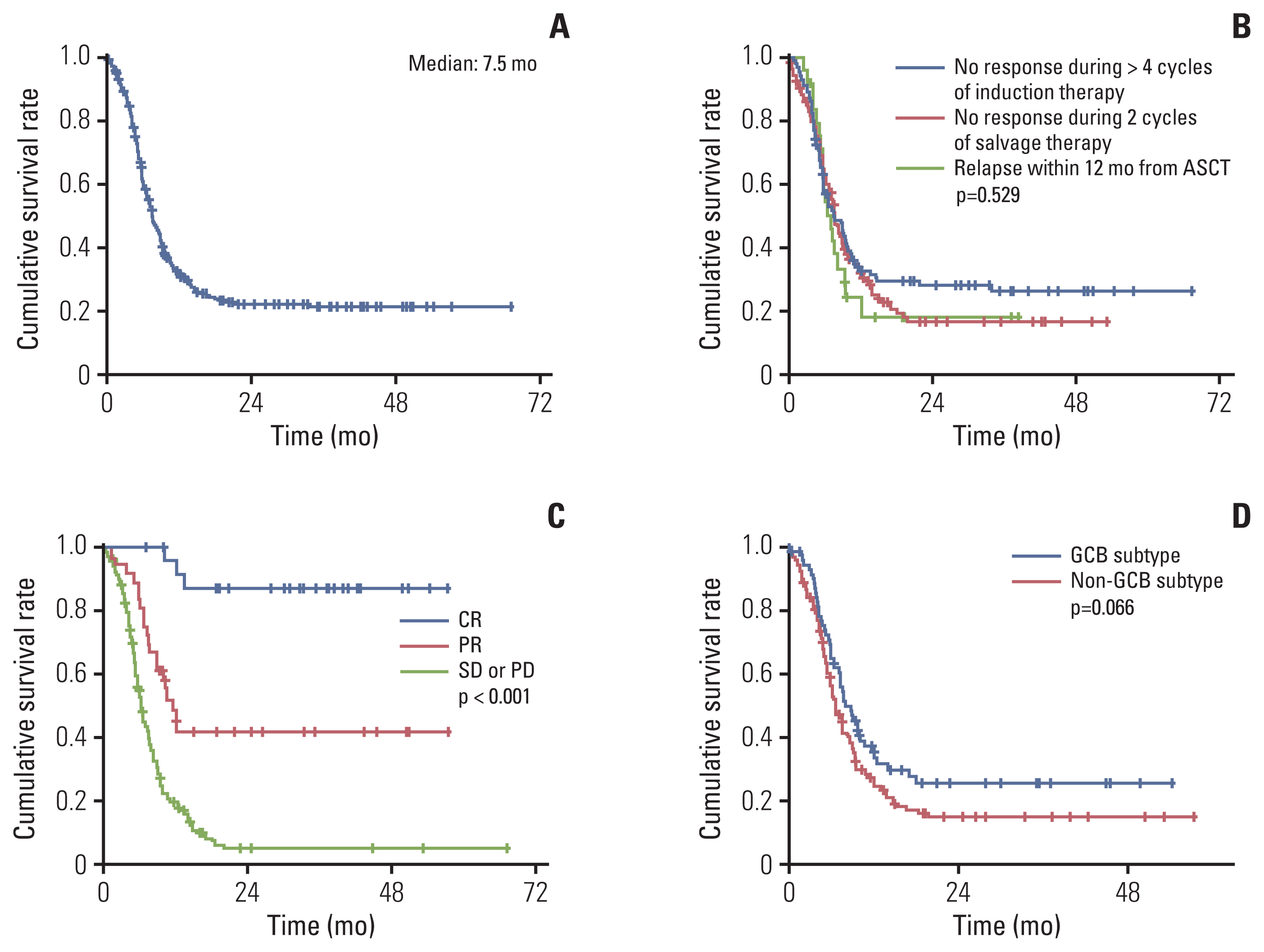
Table 1
Table 2
Table 3
Values are presented as number (%). Platinum-based regimens include ICE (ifosfamide, carboplatin, etoposide, and dexamethasone), DHAP (cytarabine, cisplatin, dexamethasone), GDP (gemcitabine, cisplatin, and dexamethasone) and ESHAP/Ox (etoposide, methyl-prednisone, cytarabine, cisplatin or oxaliplatin); Methotrexate-based regimens include IMVP16 (ifosfamide, methotrexate, etoposide, and prednisone), IVAM (ifosfamide, etoposide, cytarabine, and methotrexate), and high-dose methotrexate; bendamustine-based regimens include benda-mustine with or without rituximab; miscellaneous regimens included brentuximab-vedotin (n=1), CODOX-M/IVAC (n=1), HyperCVAD (n=2), ibrutinib (n=1), and tafasitamab (n=2). CR, complete response; PR, partial response.
Table 4
| No. (%) | Overall response rate (%) | Overall survival (95% CI, mo) | Overall survival rate (%) | |
|---|---|---|---|---|
| SCHOLAR-1 study [9] | 636 (100) | 26 | 6.3 (5.9–7.0) | 1-Year: 28, 2-Year: 20 |
| Primary refractory | 178 (28.0) | 20 | 7.1 | |
| Refractory to salvage treatment | 318 (50.0) | 26 | 6.1 | |
| Relapse ≤ 12 mo post-ASCT | 140 (22.0) | 34 | 6.2 | |
| REAL-TREND study [22] | 350 (100) | 30 | 5.9 (5.5–7.1) | 2-Year: 16 |
| Primary refractory | 181 (51.7) | 39 | 5.9 (5.5–7.1) | |
| Refractory to salvage treatment | 148 (42.3) | 18 | 5.9 (3.2-not reached) | |
| Relapse ≤ 12 mo post-ASCT | 21 (6.0) | 24 | ||
| The current study | 260 (100) | 26.4 | 7.5 (6.4–8.6) | 2-Year: 22.1 |
| Primary refractory | 102 (39.2) | 27.5 | 7.5 (5.2–9.8) | |
| Refractory to salvage treatment | 133 (51.2) | 19.5 | 7.6 (6.5–8.7) | |
| Relapse ≤ 12 mo post-ASCT | 25 (9.6) | 28.0 | 7.0 (5.6–8.4) |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download