Introduction
Method of the narrative review
Table 1.
| Author/year | Surgery/approach | Block | Local anesthetics | Sample size | Primary outcome | Other results |
|---|---|---|---|---|---|---|
| PIP | ||||||
| Fujii et al. 2019 [28] | CABG, valve/sternotomy | Deep PIP | 0.3% or 0.5% ropivacaine, 20 ml | C (n=8) | High patient recruitment (95%) | No statistical analysis |
| B (n=9) | ||||||
| Bloc et al. 2021 [5] | CABG/sternotomy | Superficial PIP | 0.25% ropivacaine, 15 × 4 ml | C (n=17) | Reduced intraoperative OC | Reduced postoperative proinflammatory |
| B (n=18) | cytokines | |||||
| Aydin et al. 2020 [29] | CABG, valve/sternotomy | Deep PIP | 0.25% bupivacaine, 20 ml | C (n=24) | Reduced 24 h post-operation OC | Reduced pain score and PONV incidence |
| B (n=24) | ||||||
| Hamed et al. 2022 [24] | CABG, valve/sternotomy | Superficial PIP | 0.25% bupivacaine, 20 ml | C (n=35) | Reduced 24-h OC | Longer first analgesic request time, reduced wound pain score |
| B (n=35) | ||||||
| Khera et al. 2021 [25] | CABG, valve/sternotomy | Superficial PIP | 0.25% bupivacaine, 20 ml | C (n=40) | No difference in the 48-h OC | No difference in the incidence of postoperative delirium |
| B (n=40) | ||||||
| Zhang et al. 2021 [30] | Open cardiac surgery (unspecified)/sternotomy | Deep PIP | 0.4% ropivacaine, 20 ml | C (n=30) | Less perioperative OC | Improved sleep quality, reduced time to extubation and ICU stay |
| B (n=30) | ||||||
| Abdelbaser and Mageed 2020 [31] | Pediatric/sternotomy | Deep PIP | 0.25% bupivacaine, 0.2–0.4 ml/kg | C (n=36) | Decreased 24-h perioperative OC | Lower intraoperative HR and MAP, longer time to first rescue analgesia, shorter time to extubate and ICU stay |
| B (n=37) | ||||||
| Zhang et al. 2020 [6] | Pediatric/sternotomy | Deep PIP | 0.2% ropivacaine, 0.75 ml/kg | C (n=50) | Lower MOPS | Less OC, reduced time to extubate and ICU and hospital stay |
| B (n=50) | ||||||
| Zhang et al. 2022 [32] | Pediatric/sternotomy | Superficial PIP | 0.2% ropivacaine, 1.5 mg/kg | C (n=51) | Lower MOPS at 24 h post-operation | Lower perioperative OC, reduced time to extubate, time to initial flatus, and length of ICU and hospital stay |
| B (n=50) | ||||||
| ESP | ||||||
| Athar et al. 2021 [8] | CABG, valve/sternotomy | ESP | 0.25% levobupivacaine, 20 ml | C (n=15) | Reduced OC in 24 h post-operation | Prolonged time to first rescue analgesia, shorter duration of mechanical ventilation |
| B (n=15) | ||||||
| Krishna et al. 2019 [40] | CABG, valve, ASD/unspecified | ESP | 0.375% ropivacaine, 3 mg/kg | C (n=53) | Reduced postoperative pain | Higher mean duration of analgesia |
| B (n=53) | ||||||
| Wasfy et al. 2021 [42] | CABG/sternotomy | Continuous ESP | 0.25% bupivacaine, 15 ml | C (n=20) | Lower pain score to 48 h after extubation | Reduced total perioperative OC, higher peak inspiratory flow, shorter duration of ventilation and ICU stay |
| 0.125% bupivacaine, 8 ml/h for 48 h | B (n=20) | |||||
| Gado et al. 2022 [50] | Pediatric/sternotomy | ESP | 0.25% bupivacaine, 0.4 ml/kg | C (n=48) | Lower intraoperative OC | Delayed first rescue analgesia |
| B (n=50) | ||||||
| Kaushal et al. 2020 [7] | Pediatric/sternotomy | ESP | 0.2% ropivacaine, 1.5 mg/kg | C (n=40) | Reduced pain score to 10 h post-operation | Less postoperative OC, longer time to first rescue dose, lower postoperative sedation scores and ICU stay |
| B (n=40) | ||||||
| Karacaer et al. 2022 [49] | Pediatric/sternotomy | ESP | 0.25% bupivacaine, 0.5 ml/kg | C (n=20) | Lower OC in first 24 h post-operation | No significant difference in pain score |
| B (n=20) | ||||||
| Macaire et al. 2020 [51] | Pediatric/sternotomy | Continuous ESP | 0.1% or 0.2% ropivacaine, 0.5 ml/kg/side | C (n=23) | Decreased OC during first 48 h post-operation | Reduced pain scores, no differences in times to extubation and drain removal or length of hospital stay |
| B (n=27) | ||||||
| Retrolaminar | ||||||
| Abdelbaser et al. 2022 [55] | Pediatric/sternotomy | Retrolaminar | 0.25% bupivacaine, 0.4 ml/kg | C (n=28) | Reduced perioperative OC | Lower pain score, shorter time to extubation |
| B (n=29) | ||||||
| IPP/PSP/SAP | ||||||
| Kaushal et al. 2019 [59] | Pediatric/thoracotomy | SAP, IPP-PSP, INB | 0.2% ropivacaine, 3 mg/kg | SAP (n=36) | Mean MOPS at 6, 8, 10, 12 h were lower with the SAP and IPP-PSP block | Reduced postoperative OC for the SAP, IPP-PSP block |
| IPP (n=36) | ||||||
| INB (n=36) | ||||||
| Kamal et al. 2022 [60] | Pediatric/sternotomy | IPP-PSP | 0.25% bupivacaine, 0.5 ml/kg | C (n=20) | Lower pain score at 6 h post-operation | Longer interval to first rescue analgesia, lower PAED score, shorter ICU stay |
| B (n=20) | ||||||
| Kumar et al. 2018 [61] | CABG, valve/sternotomy | IPP-PSP | 0.25% bupivacaine, 60 ml | C (n=20) | Shorter duration of ventilator support | Lower pain score, higher peak inspiratory flow rates, less rescue analgesia administered |
| B (n=20) | ||||||
| Gautam et al. 2020 [64] | MIDCAB/left anterior thoracotomy | Continuous deep SAP | 0.2% ropivacaine, 20 ml (1 μg/ml fentanyl), 8 ml/h infusion | C (n=20) | Reduced pain scores | Reduced postoperative 48-h OC |
| B (n=24) | ||||||
| Combined | ||||||
| Gawęda et al. 2020 [63] | Valve/mini-thoracotomy approach | ESP, ESP + IPP-PSP | 0.375% ropivacaine, 0.2 ml/kg | ESP (n=15) | ESP + IPP-PSP block reduced OC and pain score | Increased patient satisfaction in ESP + IPP-PSP group |
| ESP + IPP-PSP (n=15) |
CABG: coronary artery bypass graft, C: control group, B: block group, OC: opioid consumption, HR: heart rate, MAP: mean arterial pressure, ASD: atrial septal defect, MIDCAB: minimally invasive direct coronary artery bypass, INB: intercostal nerve block, PIP: parasternal intercostal plane, IPP: interpectoral plane, PSP: pectoserratus plane, SAP: serratus anterior plane, ESP: erector spinae plane, PONV: postoperative nausea and vomiting, ICU: intensive care unit, MOPS: modified objective pain score, NRS: numeric rating scale, PAED: pediatric anesthesia emergence delirium.
Nomenclature
Table 2.
Fascial plane blocks for cardiac surgery
Parasternal intercostal plane block
Superficial PIP block
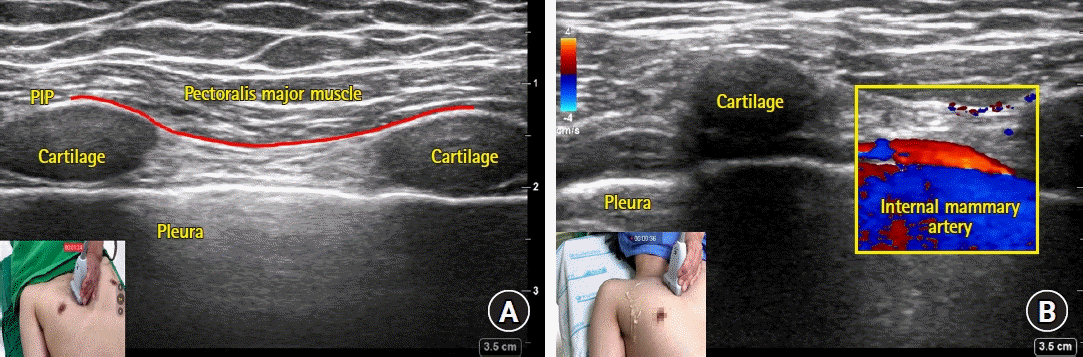 | Fig. 1.Sonoanatomy (A) and color doppler image of internal mammary artery (B) captured during parasagittal approach of superficial parasternal intercostal plane (PIP) block. (A) The fascial plane between the pectoralis major and internal intercostal muscles (PIP) is indicated as a red solid line. Cartilages were captured as echolucent structures. (B) The internal mammary artery is captured as a red-colored tubular structure on the long axis view. |
Deep PIP block
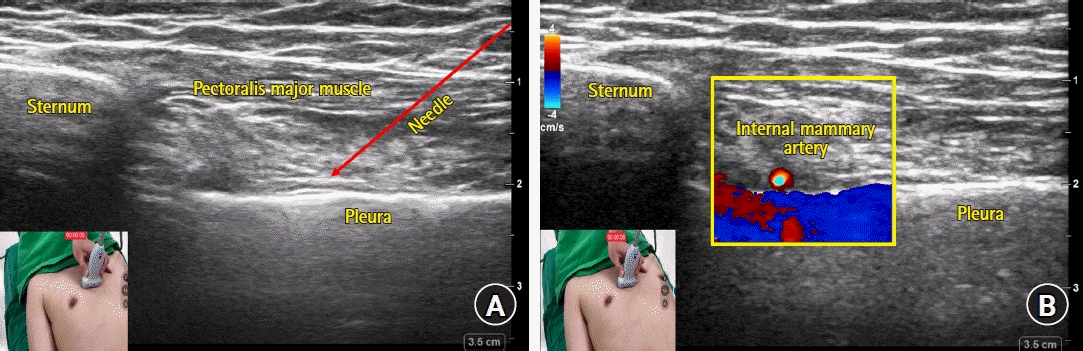 | Fig. 2.Sonoanatomy (A) and color doppler image of internal mammary artery (B) captured during transverse approach of deep parasternal intercostal plane (PIP) block. (A) The needle tip placement target is the fascial plane between the internal intercostal and transversus thoracis muscles. The transversus thoracis muscle is often difficult to clearly distinguish on an ultrasound image. The needle path of the transverse approach is indicated as a red solid line with an arrowhead. (B) The internal mammary artery is captured as a bright-colored round structure on the short axis image. It should be visualized and used as a landmark in order to avoid inadvertent punctures. |
Use of PIP blocks in pediatric patients
Erector spinae plane block
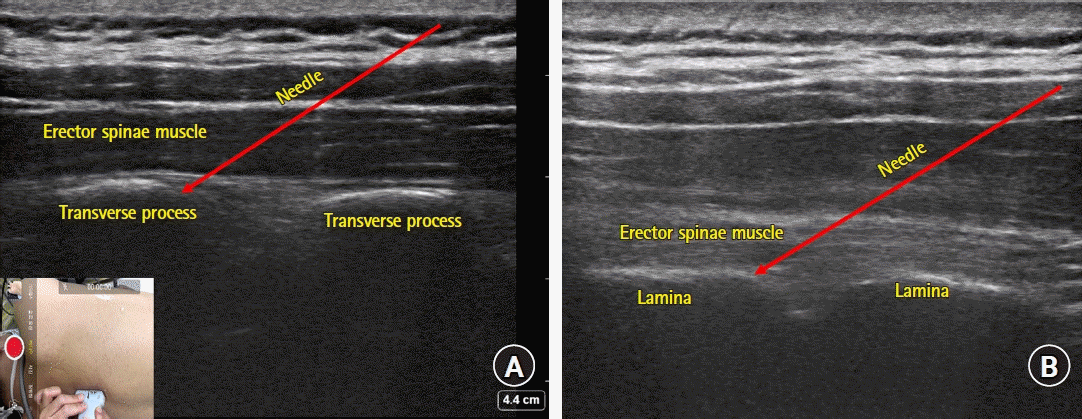 | Fig. 3.Sonoanatomy of erector spinae plane (ESP) (A) and retrolaminar (B) blocks. (A) Note the transverse process of the spine captured as bright squared-off bony structures underneath the erector spinae muscle. The edge of the transverse process is the preferred target for needle placement and slight advancement deeper off the edge may be needed to achieve proper spread of the injectate into the plane between the erector spinae muscle and the transverse process. (B) Note the flat structures (lamina) with small notches and the overlying erector spinae muscle. Using an in-plane technique, the needle can be introduced until the tip contacts with the lamina. Optimal needle positioning can be confirmed by observing the proper spread of injectate throughout the plane between the lamina and the erector spinae muscle. |
Use of ESP block in pediatric patients
Retrolaminar block
Interpectoral plane, pectoserratus plane, and serratus anterior plane blocks
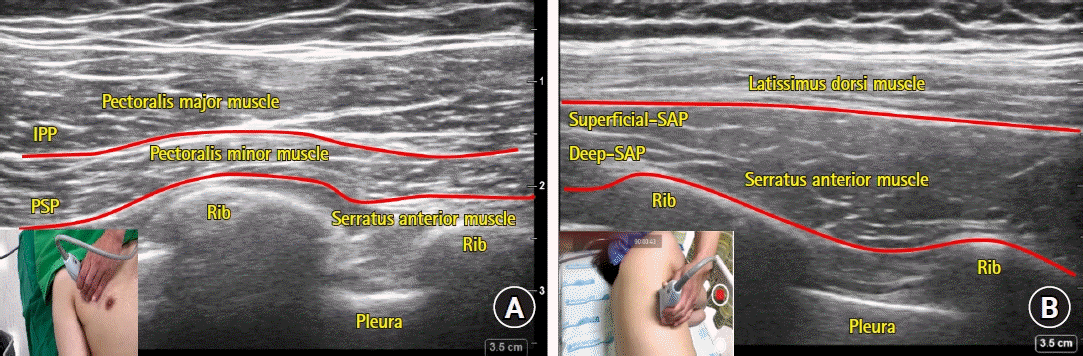 | Fig. 4.Sonoanatomy of interpectoral plane (IPP) and pectoserratus plane (PSP) blocks (A), and deep and superficial serratus anterior plane (SAP) blocks (B). (A) The fascial planes between pectoralis major and minor muscles (IPP) and pectoralis minor and serratus anterior muscles (PSP) are indicated as solid red lines. Note that the probe is placed at the right upper quadrant of the chest wall (over the pectoralis muscles). (B) The planes superficial (superficial SAP) or deep (deep SAP) to the serratus anterior muscle are indicated as solid red lines. Note that the probe is placed at the lateral chest wall. |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download