Abstract
The accurate measurement of ABO antibodies is essential for successful ABO-incompatible solid organ transplantation. Titration using two-fold dilution is considered a standard method and is applied in most laboratories. However, this titration method has inherent limitations, including differences in methods between laboratories, a lack of standardization, its semiquantitative nature, and the difficulty of considering the results to be representative of the in vivo activity of ABO antibodies. Various measurement methods other than titration have been developed, and new methods continue to be introduced. Physicians and laboratory specialists who are involved in ABO-incompatible solid organ transplantation need to fully understand these methods for optimal patient management.
Unlike other common blood groups, ABO antigens are expressed not only on red blood cells, but also in many other tissues. In particular, ABO antigens are present in vascular endothelial cells; therefore, ABO-incompatible solid organ transplantation was contraindicated for a long time. However, with the development of various desensitization and immunosuppression modalities, ABO-incompatible solid organ transplantation is no longer an absolute contraindication [1]. According to data from the Center for Korean Network for Organ Sharing, 26.4% of living donor kidney and liver transplantations performed in Korea in 2019 were ABO-incompatible [2].
For ABO-incompatible solid organ transplantation, the level of ABO antibodies should be reduced below a specific threshold through desensitization using rituximab administration and an antibody removal method such as plasmapheresis before surgery. Additional treatment is determined by monitoring whether ABO antibodies increase after surgery [3]. The specific protocols vary across laboratories, but accurate ABO antibody measurements play a decisive role in establishing the treatment plan regardless of the protocol followed. However, ABO antibody tests are not standardized and have significant variations between laboratories [4,5]. This review covers the various methods of ABO antibody measurements for ABO-incompatible solid organ transplantation.
ABO antibodies naturally occur and differ from the known adaptive immune response. Unlike immune antibodies, which develop as immunoglobulin M (IgM) before switching to immunoglobulin G (IgG) when exposed to antigens, ABO antibodies often persist as IgM. In particular, most type A and type B ABO antibodies are IgM [6]. IgG antibodies are found relatively frequently in type O, and the IgG2 subclass is the main component [7]. Institutions have different protocols regarding which antibodies are monitored for ABO-incompatible transplantation. As mentioned above, the main component of ABO antibodies is IgM, and the IgG antibodies that are present often belong to the IgG2 subclass, which has a low complement activation capacity. A recent study reported that ABO-incompatible transplantation could be successfully conducted by monitoring IgM isoagglutinin titers [8]. However, another study reported the clinical significance of anti-A/B IgG titers in ABO-incompatible transplantation [9]. Further studies are needed to clarify this issue.
At birth, there is only a small amount of ABO antibodies, which derive from the placental transfer of maternal IgG antibodies, but ABO antibodies begin to develop at 3–6 months after birth and reach adult levels at the age of 5–10 years. This provides a rationale for ABO-incompatible heart transplantation in infants [10].
It is understood that the cause of naturally occurring ABO antibodies without exposure to ABO antigens is due to cross-reaction to similar antigens present in the environment, especially bacteria. Hence, the ABO antibody titer is well-known to be elevated when exposed to bacterial antigens [11].
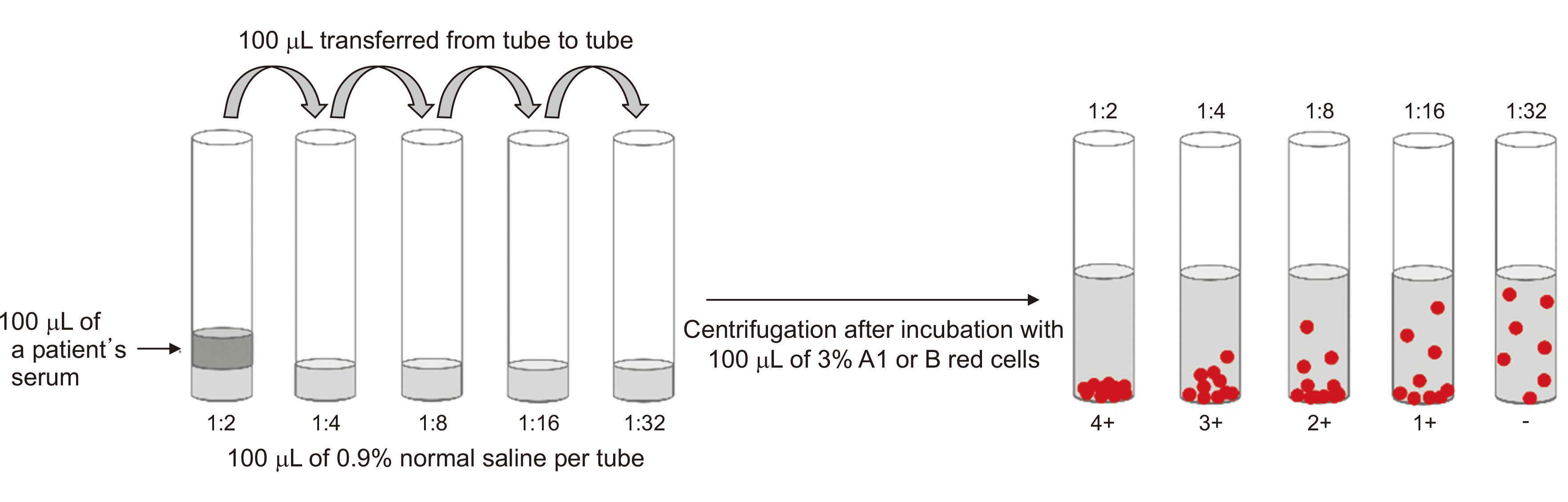
Blood group antibodies are conventionally measured as titers using the two-fold dilution method. Titration is a method wherein the amount of antibodies is indirectly expressed as the reciprocal of the endpoint titer obtained by serially diluting the serum. The titer endpoint that shows agglutination is interpreted as the isoagglutinin titer (Fig. 1). Two representative methods for the titration of anti-A and anti-B antibodies are the tube method and the column agglutination technique (CAT).
The accurate measurement of anti-D antibody titers in RhD-negative mothers has been critical for the diagnosis and management of hemolytic disease of the fetus and newborn (HDFN). For HDFN, titration has been widely used for a long time. As ABO-incompatible solid organ transplantation became possible, the necessity and demand for measurements of anti-A and anti-B antibodies increased. Titration using two-fold dilution is widely performed for the measurement of ABO antibodies in many clinical laboratories because it has a long history, practitioners have accumulated related experience, and it does not deviate from the basic framework of other transfusion-related tests. However, titration has several inherent limitations. It has not been standardized, and there are significant interobserver and interlaboratory variations [12,13]. Titration techniques are different among laboratories in terms of whether they use the tube or CAT method, as well as according to whether they measure IgM or IgG [7,14]. The semiquantitative nature of titration is another disadvantage for clinical usage. The selection of reagent cells is another aspect that can influence the standardization of titration. Dosage effects for some blood groups, including Rh, are a well-known phenomenon, although relatively little is known regarding ABO antigens. Further research is needed for the selection of appropriate reagent cells for ABO antibody titration standardization. Lastly, like other techniques that have been developed to date, titration gives information about the antigen-antibody binding reaction, but this is insufficient to tell us what might happen in vivo.
According to the College of American Pathologists (CAP), which is a representative external quality assessment organization, external quality assessment is used for the titration of ABO antibodies and anti-D antibodies [15]. These processes vary depending on the reagent cell, the diluent, the presence of anti-human globulin or dithiothreitol (DTT), the incubation time, and the reaction temperature. In order to reduce variations and standardize these methods, the CAP proposed a uniform procedure in 2008 [16]. It aims to ensure that the results are consistent regardless of the laboratory through the use of specific suggested protocols for reagent cells, diluents, reaction conditions, and reagents for titration testing using the tube method or CAT. However, it was found that these protocols did not actually help to reduce the variation among laboratories [12]. Thus, the mode±2 titers is set as an acceptable range in external quality assessments of the CAP [15]. Thus, laboratories that report results with a difference of up to four titers for the same specimen may all receive acceptable results from external quality assessments. Thus, it is challenging to compare titer results among laboratories. Each laboratory should set its own cutoff for titers in the ABO-incompatible transplantation protocol. The recent development of antibody titration conducted by automated blood type analyzers is expected to enhance the uniformity of test results in the future.
Some reports have described ABO antibody measurement using flow cytometry [17-20]. This method has the advantage of classifying the immunoglobulin type and subclass depending on which antibody is used as the secondary antibody. Moreover, there is no need for DTT treatment to remove IgM, and multiple types of antibodies can be analyzed simultaneously using various combinations of antibodies. However, this method is also not standardized, and although the fluorescent intensity value is expressed, it is not quantitative data. In addition, special equipment and skilled personnel are required for the test, and there are limitations in using it for routine tests due to cost and time.
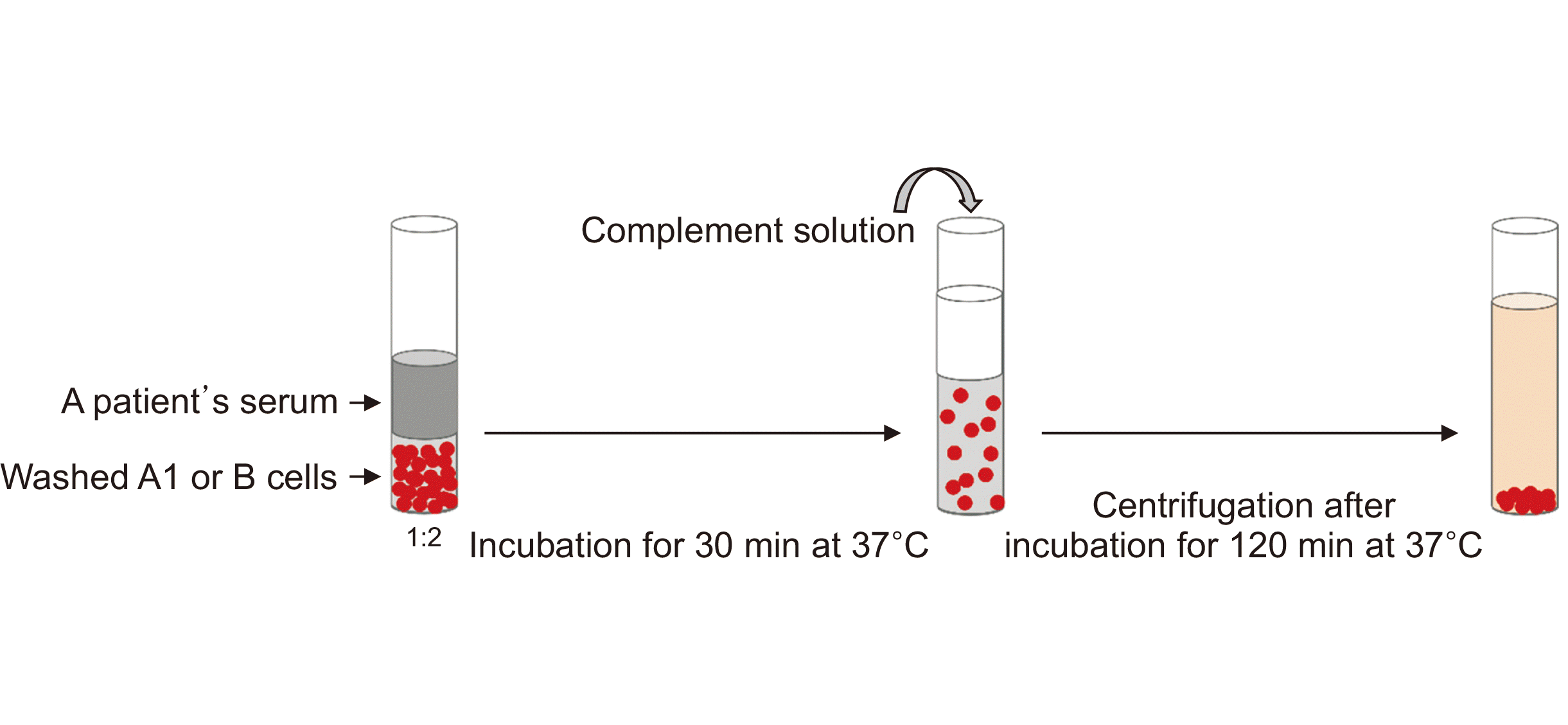
The authors recently developed and published an ABO antibody measurement method that applies the complement-dependent cytotoxicity method using a principle similar to that of the human leukocyte antigen antibody test [21] (Fig. 2). This method was developed based on the fact that the in vivo pathological process that causes problems with ABO antibodies during ABO-incompatible transplantation begins with complement activation [5,22]. The advantage of this method is that quantitative data can be obtained by establishing 0% and 100% hemolysis control, and in vivo activity can be estimated regardless of immunoglobulin type or subclass. However, since this is still a newly developed method, it has not yet been validated on a large scale. Furthermore, since this test involves several manual procedures, it is labor-intensive and time-consuming. The enzyme-linked immunosorbent assay method was also recently introduced for the measurement of ABO antibodies, but it has not yet been widely distributed [22].
The accommodation phenomenon, in which rejection does not occur even if the ABO antibody concentration increases, can occur 2–4 weeks after ABO-incompatible transplantation. Various theories have been suggested to explain this phenomenon: (1) the inhibition of complement and apoptosis; (2) no T-cell-dependent B-cell response; (3) blood group chimerism or shift; and (4) B-cell tolerance [5]. Thus, the clinical significance of measuring the ABO antibody level after accommodation has not yet been accurately established, and the Korean health insurance system covers ABO antibody measurements only up to 2 weeks after transplantation. The British Society for Haematology guidelines also state that there is no issue with transfusion of blood components of the patient’s blood type 4 weeks after transplant [23].
However, there was a case in which the ABO antibody titer increased rapidly due to sepsis, resulting in graft rejection several months after transplantation [24]. In this regard, it has been hypothesized that primary sensitization occurs due to ABO-associated antigens. The properties of antibody-mediated rejection (AMR) immediately after surgery and delayed AMR, which usually occurs 2 to 4 weeks after transplantation, are different, and in the latter type of AMR, there is a greater increase of the IgM titer than of the IgG titer [25]. Further studies are needed to elucidate the underlying mechanism.
The accurate measurement of ABO antibodies is essential for successful ABO-incompatible transplantation. The conventional titration method is considered standard and is applied in most laboratories. Various measurement methods have been developed, and new methods continue to be introduced. Each method has advantages and disadvantages. Therefore, physicians and laboratory specialists who are involved in ABO-incompatible transplantation need to fully understand these methods for optimal patient management.
ACKNOWLEDGMENTS
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Funding/Support
This study was supported by research grant from the Korean Society for Transplantation (2022-00-02001-002).
Author Contributions
Conceptualization: DHK. Visualization: YC. Writing–original draft: YC. Writing–review & editing: DHK.
REFERENCES
1. Alexandre GP, De Bruyere M, Squifflet JP, Moriau M, Latinne D, Pirson Y. 1985; Human ABO-incompatible living donor renal homografts. Neth J Med. 28:231–4. PMID: 3892322.
2. Korean Network for Organ Sharing (KONOS). 2019 Annual data report [Internet]. KONOS;Seoul: Available from: http://konos.go.kr. cited 2022 May 1.
3. Böhmig GA, Farkas AM, Eskandary F, Wekerle T. 2015; Strategies to overcome the ABO barrier in kidney transplantation. Nat Rev Nephrol. 11:732–47. DOI: 10.1038/nrneph.2015.144. PMID: 26324199.
4. Kang SJ, Lim YA, Baik SY. 2014; Comparison of ABO antibody titers on the basis of the antibody detection method used. Ann Lab Med. 34:300–6. DOI: 10.3343/alm.2014.34.4.300. PMID: 24982835. PMCID: PMC4071187.
5. Lee EY, Kim S, Kim HO, Kwon SW, Kim DW, Han KS. 2011; Survey analysis of ABO antibody titration at four university hospitals in Korea. Korean J Blood Transfus. 22:24–30.
6. Rawson AJ, Abelson NM. 1960; Studies of blood groups antibodies. III. Observations on the physicochemical properties of isohemagglutinins and isohemolysins. J Immunol. 85:636–9. PMID: 13739933.
7. Brouwers HA, Overbeeke MA, Gemke RJ, Maas CJ, van Leeuwen EF, Engelfriet CP. 1987; Sensitive methods for determining subclasses of IgG anti-A and anti-B in sera of blood-group-O women with a blood-group-A or -B child. Br J Haematol. 66:267–70. DOI: 10.1111/j.1365-2141.1987.tb01310.x. PMID: 3300763.
8. Kim H, Choe W, Shin S, Kim YH, Han DJ, Park SK, et al. 2020; ABO-incompatible kidney transplantation can be successfully conducted by monitoring IgM isoagglutinin titers during desensitization. Transfusion. 60:598–606. DOI: 10.1111/trf.15672. PMID: 31957888.
9. Lee J, Kim EJ, Yang SJ, Kim DG, Ihn K, Lee JG, et al. 2020; Impact of the baseline anti-A/B IgG titer on the clinical outcome in ABO-incompatible liver transplantation. Transplantation. 104:S526. DOI: 10.1097/01.tp.0000701344.29242.43.
10. Urschel S, West LJ. 2016; ABO-incompatible heart transplantation. Curr Opin Pediatr. 28:613–9. DOI: 10.1097/MOP.0000000000000398. PMID: 27379804. PMCID: PMC5123853.
11. Springer GF, Horton RE. 1969; Blood group isoantibody stimulation in man by feeding blood group-active bacteria. J Clin Invest. 48:1280–91. DOI: 10.1172/JCI106094. PMID: 4893685. PMCID: PMC322351.
12. Bachegowda LS, Cheng YH, Long T, Shaz BH. 2017; Impact of uniform methods on interlaboratory antibody titration variability: antibody titration and uniform methods. Arch Pathol Lab Med. 141:131–8. DOI: 10.5858/arpa.2015-0351-OA. PMID: 27681333.
13. Pandey P, Setya D, Ranjan S, Singh MK, Singh MK. 2021; Comparison of ABO isoagglutinin titres by three different methods in group O blood donors. ISBT Sci Ser. 16:76–84. DOI: 10.1111/voxs.12608.
14. Kang MG, Lee SJ, Oh JS, Lim YA. 2009; Comparison of ABO isoagglutinin titers by different tube hemagglutination techniques. Korean J Blood Transfus. 20:227–34.
15. College of American Pathologists. 2021 ABT-A, participant summary report. College of American Pathologists;2021.
16. AuBuchon JP, de Wildt-Eggen J, Dumont LJ. Biomedical Excellence for Safer Transfusion Collaborative; Transfusion Medicine Resource Committee of the College of American Pathologists. 2008; Reducing the variation in performance of antibody titrations. Arch Pathol Lab Med. 132:1194–201. DOI: 10.5858/2008-132-1194-RTVIPO. PMID: 18605771.
17. Stussi G, Huggel K, Lutz HU, Schanz U, Rieben R, Seebach JD. 2005; Isotype-specific detection of ABO blood group antibodies using a novel flow cytometric method. Br J Haematol. 130:954–63. DOI: 10.1111/j.1365-2141.2005.05705.x. PMID: 16156865.
18. Yung GP, Valli PV, Starke A, Mueller RJ, Fehr T, Cesar-Ozpamir M, et al. 2007; Flow cytometric measurement of ABO antibodies in ABO-incompatible living donor kidney transplantation. Transplantation. 84(12 Suppl):S20–3. DOI: 10.1097/01.tp.0000296646.17845.12. PMID: 18162982.
19. Sundbäck M, Grufman P, Teller J, Westphal F, Korchagina E, Bovin N, et al. 2007; Quantification of blood group A and B antibodies by flow cytometry using beads carrying A or B trisaccharides. Transplantation. 84(12 Suppl):S24–6. DOI: 10.1097/01.tp.0000296043.95984.9a. PMID: 18162983.
20. Won D, Kim BC. 2012; Optimized flow cytometry to measure anti-ABO immunoglobulin G. Lab Med. 43:281–90. DOI: 10.1309/LM1O8F7MLLYIPIMZ.
21. Youk HJ, Ryu HY, Seo SW, Kim JS, Chung Y, Kim H, et al. 2021; ABO antibody measurement using complement-dependent cytotoxicity. Ann Lab Med. 41(S1):15.
22. Satoh A, Kawagishi N, Minegishi M, Takahashi H, Akamatsu Y, Doi H, et al. 2007; Development of a novel ELISA for detection of anti-A and anti-B antibodies in recipients of ABO-incompatible living donor liver grafts. Tohoku J Exp Med. 211:359–67. DOI: 10.1620/tjem.211.359. PMID: 17409676.
23. Green L, Bolton-Maggs P, Beattie C, Cardigan R, Kallis Y, Stanworth SJ, et al. 2018; British Society of Haematology Guidelines on the spectrum of fresh frozen plasma and cryoprecipitate products: their handling and use in various patient groups in the absence of major bleeding. Br J Haematol. 181:54–67. DOI: 10.1111/bjh.15167. PMID: 29527654.
24. de Weerd A, Vonk A, van der Hoek H, van Groningen M, Weimar W, Betjes M, et al. 2014; Late antibody-mediated rejection after ABO-incompatible kidney transplantation during Gram-negative sepsis. BMC Nephrol. 15:31. DOI: 10.1186/1471-2369-15-31. PMID: 24517251. PMCID: PMC3925416.
25. Takahashi K. 2007; Recent findings in ABO-incompatible kidney transplantation: classification and therapeutic strategy for acute antibody-mediated rejection due to ABO-blood-group-related antigens during the critical period preceding the establishment of accommodation. Clin Exp Nephrol. 11:128–41. DOI: 10.1007/s10157-007-0461-z. PMID: 17593512.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download