INTRODUCTION
MATERIALS AND METHODS
Patient populations
Surgical procedure and postoperative management
 | Fig. 1.Computed tomography images showing the degree of facets removed to remove lateral parts of ossification of the ligamentum flavum (OLF) from the F-group and D-group. In the F-group (A and B), more than half of the facets were sufficiently removed to remove the lateral parts of OLF, but in the D-group (C), residual OLF was observed at the lateral part (arrows). |
Clinical evaluation
Table 1.
Radiographic evaluation
 | Fig. 2.Measurements of radiographic parameters. Thoracic kyphosis angle (TKA) indicating the angle between the upper endplate of T4 and the lower endplate of T12 (A). Segmental kyphosis angle (SKA) indicating the angle between the upper and lower endplates at the disc level where surgery was performed (B). Lumbar lordosis angle (LLA) indicating the angle between the upper endplate of L1 and the upper endplate of S1 (C). Pelvic incidence (PI) indicating the angle between the line perpendicular to the sacral plate and the line connecting the midpoint of the sacral plate to the bicoxofemoral axis, sacral slope (SS) indicating the angle between the sacral plate and the horizontal plane, and pelvic tile (PT) indicating the angle between the lines connecting the midpoint of the sacral plate to the bicoxofemoral axis and the vertical plane (D). |
Statistical analysis
RESULTS
Table 2.
Values are presented as mean±standard deviation or number (%). Comparisons between continuous values were performed using Mann-Whitney U tests (or t-tests), while comparisons between non-continuous values were performed using Wilcoxon signed-rank tests and chi-square tests (or Fisher’s exact tests). D-group : decompression only surgery for thoracic ossification of the ligamentum flavum, F-group : decompression plus instrumented fusion surgery for thoracic ossification of the ligamentum flavum. BMI : body mass index, DM : diabetes mellitus, PI : pelvic incidence
Table 3.
Values are presented as mean±standard deviation or number. Comparisons between continuous values were performed using Mann-Whitney U tests (or t-tests). D-group : decompression only surgery for thoracic ossification of the ligamentum flavum, F-group : decompression plus instrumented fusion surgery for thoracic ossification of the ligamentum flavum. JOA : Japanese Orthopedic Association, VAS-B : Visual analogue scale of the back, VAS-L : Visual analogue scale of the leg, K-ODI : Korean version of the Oswestry disability index, SVA : sagittal vertical axis, PT : pelvic tilt, SS : sacral slope, TKA : thoracic kyphosis angle, SKA : segmental kyphosis angle at the operated level, LLA : lumbar lordosis angle (a negative value implying lordosis)
Table 4.
| Postoperative 1 year | Postoperative 2 years | p-value* | p-value† | |
|---|---|---|---|---|
| d_JOA | 0.631 | 0.898 | ||
| D-group | 1.5±1.0 | 1.5±0.9 | ||
| F-group | 1.6±1.2 | 1.6±1.2 | ||
| d_VAS-B | 0.814 | 0.939 | ||
| D-group | -2.2±1.5 | -2.1±2.0 | ||
| F-group | -2.2±2.1 | -2.4±1.6 | ||
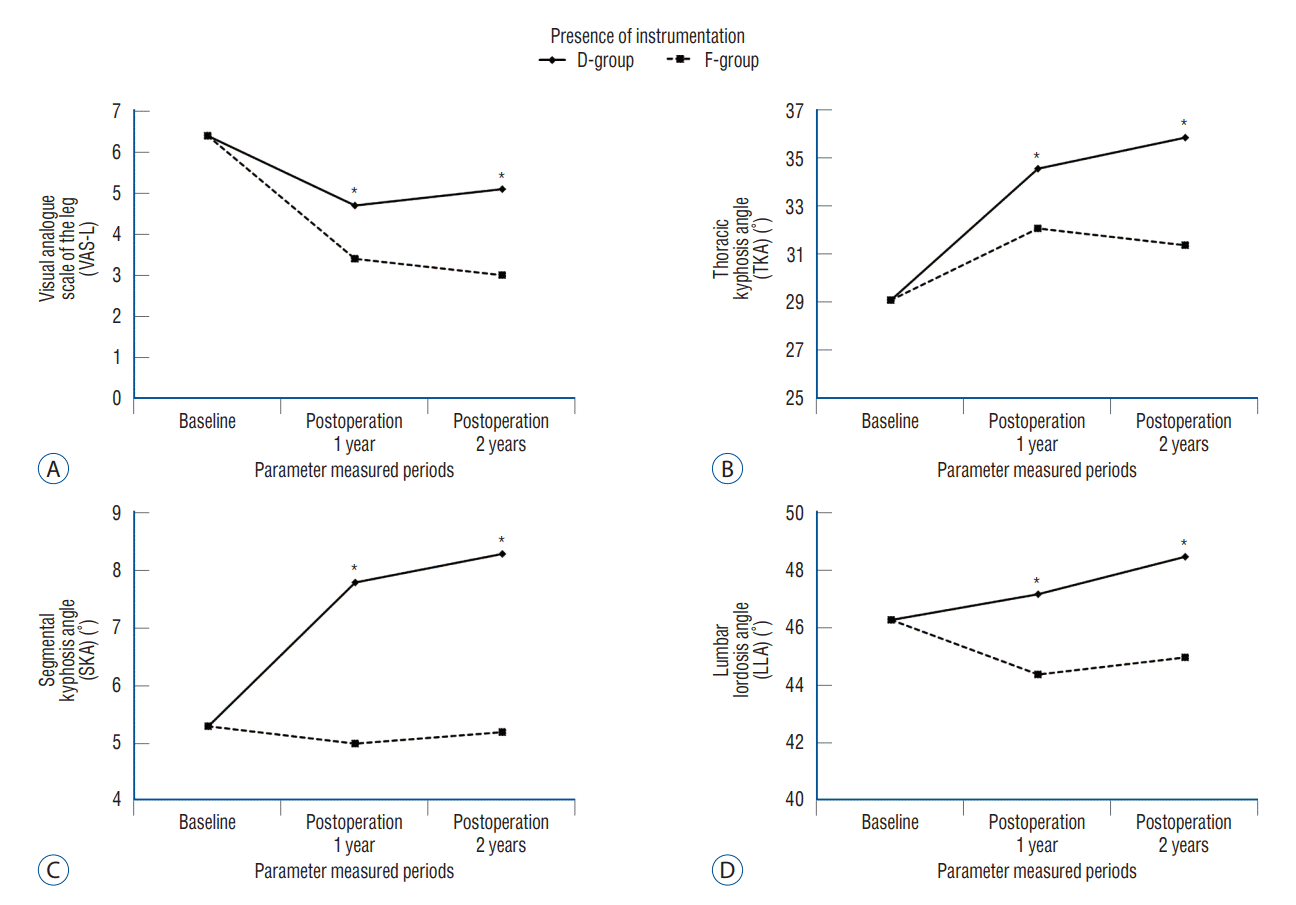
| d_VAS-L | 0.008 | 0.908 | ||
| D-group | -1.7±1.7 | -1.3±2.2 | ||
| F-group | -3.0±2.6 | -3.4±2.5 | ||
| d_ODI | 0.701 | 0.804 | ||
| D-group | -9.7±8.6 | -9.5±7.9 | ||
| F-group | -10.9±7.6 | -9.9±6.2 | ||
| d_SVA (mm) | 0.072 | 0.516 | ||
| D-group | 1.7±3.1 | 0.6±2.3 | ||
| F-group | -0.5±3.2 | -0.2±3.5 | ||
| d_PT (°) | 0.806 | 0.152 | ||
| D-group | 3.5±13.5 | -0.6±2.8 | ||
| F-group | 2.9±6.6 | 1.2±2.4 | ||
| d_SS (°) | 0.137 | 0.193 | ||
| D-group | -0.8±4.4 | 0.4±2.9 | ||
| F-group | -2.9±6.4 | -1.0±2.5 | ||
| d_TKA (°) | 0.013 | 0.596 | ||
| D-group | 5.5±6.1 | 6.8±6.1 | ||
| F-group | 2.1±4.9 | 2.3±4.7 | ||
| d_SKA (°) | <0.001 | 0.602 | ||
| D-group | 2.5±2.1 | 3.0±2.8 | ||
| F-group | -0.3±1.4 | -0.1±1.4 | ||
| d_LLA (°) | 0.037 | 0.509 | ||
| D-group | 0.9±5.9 | 2.2±5.3 | ||
| F-group | -1.9±4.8 | -1.3±5.6 |
Values are presented as mean±standard deviation or number. D-group : decompression only surgery for thoracic ossification of the ligamentum flavum, F-group : decompression plus instrumented fusion surgery for thoracic ossification of the ligamentum flavum.
d_ : change from baseline, JOA : Japanese Orthopedic Association, VAS-B : Visual analogue scale of the back, VAS-L : Visual analogue scale of the leg, ODI : Oswestry disability index, SVA : sagittal vertical axis, PT : pelvic tilt, SS : sacral slope, TKA : thoracic kyphosis angle, SKA : segmental kyphosis angle at the operated level, LLA : lumbar lordosis angle (a negative value implying lordosis)
Surgical complications and OLF recurrence
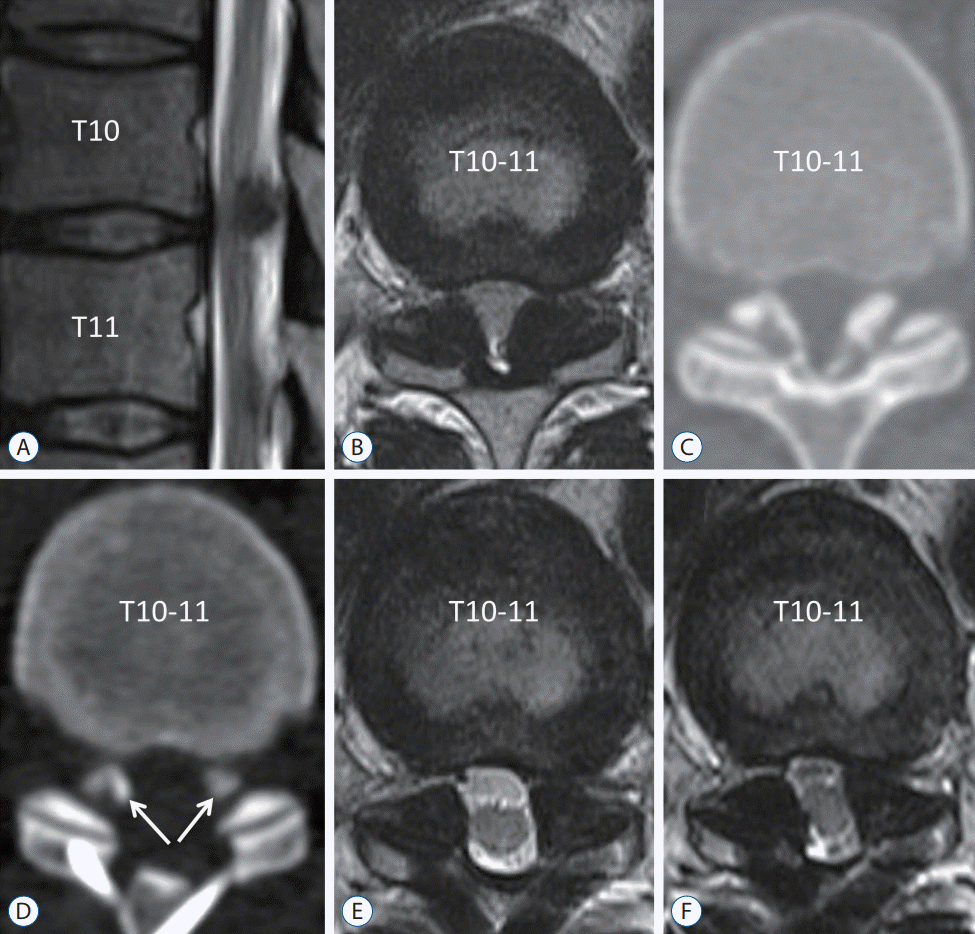 | Fig. 4.Recurrence case of ossification of the ligamentum flavum (OLF). A 43-year-old man underwent decompression surgery because of weakness in both legs (manual motor grade IV/V), decreased sensation, and voiding difficulty. Magnetic resonance (MR) imaging and computed tomography scan showed thoracic myelopathy due to OLF (A-C). The OLF was removed and laminoplasty was performed without instrumented fusion (D and E). Residual OLF was observed in the lateral part (D, arrows). Symptomatic thoracic myelopathy recurred at postoperative 2 years, and MR imaging revealed regrowth of the residual OLF (F). |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download