1. Devasagayam S, Wyatt B, Leyden J, Kleinig T. Cerebral venous sinus thrombosis incidence is higher than previously thought: a retrospective population-based study. Stroke. 2016; 47:2180–2182.
2. Coutinho JM, Zuurbier SM, Aramideh M, Stam J. The incidence of cerebral venous thrombosis: a cross-sectional study. Stroke. 2012; 43:3375–3377.
3. Salottolo K, Wagner J, Frei DF, Loy D, Bellon RJ, McCarthy K, et al. Epidemiology, endovascular treatment, and prognosis of cerebral venous thrombosis: US center study of 152 patients. J Am Heart Assoc. 2017; 6:e005480.
4. Ropper AH, Klein JP. Cerebral venous thrombosis. N Engl J Med. 2021; 385:59–64.
5. Ferro JM, Canhão P, Stam J, Bousser MG, Barinagarrementeria F; ISCVT Investigators. Prognosis of cerebral vein and dural sinus thrombosis: results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke. 2004; 35:664–670.
6. Borhani Haghighi A, Edgell RC, Cruz-Flores S, Feen E, Piriyawat P, Vora N, et al. Mortality of cerebral venous-sinus thrombosis in a large national sample. Stroke. 2012; 43:262–264.
7. Korathanakhun P, Sathirapanya P, Geater SL, Petpichetchian W. Predictors of hospital outcome in patients with cerebral venous thrombosis. J Stroke Cerebrovasc Dis. 2014; 23:2725–2729.
8. Duman T, Uluduz D, Midi I, Bektas H, Kablan Y, Goksel BK, et al. A multicenter study of 1144 patients with cerebral venous thrombosis: the VENOST Study. J Stroke Cerebrovasc Dis. 2017; 26:1848–1857.
9. Wasay M, Kaul S, Menon B, Dai AI, Saadatnia M, Malik A, et al. Asian study of cerebral venous thrombosis. J Stroke Cerebrovasc Dis. 2019; 28:104247.
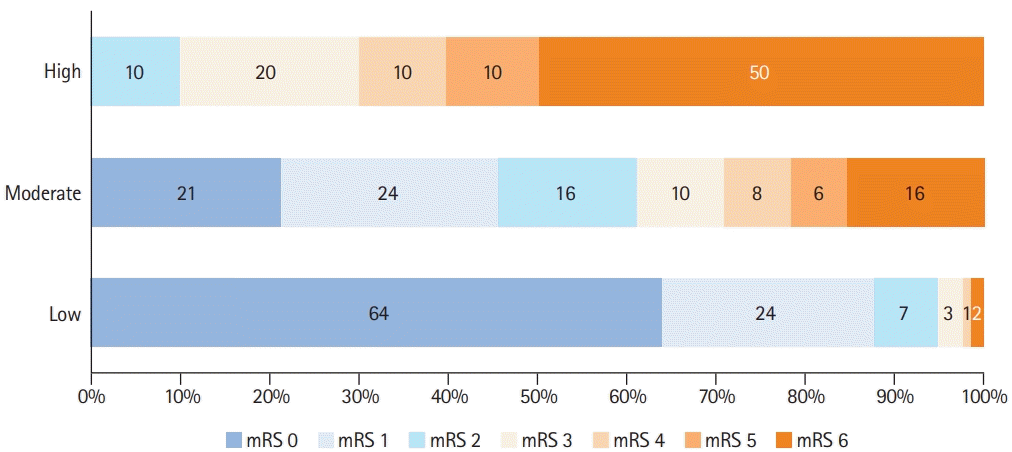
10. Ferro JM, Bacelar-Nicolau H, Rodrigues T, Bacelar-Nicolau L, Canhão P, Crassard I, et al. Risk score to predict the outcome of patients with cerebral vein and dural sinus thrombosis. Cerebrovasc Dis. 2009; 28:39–44.
11. Barboza MA, Chiquete E, Arauz A, Merlos-Benitez M, Quiroz-Compeán A, Barinagarrementería F, et al. A practical score for prediction of outcome after cerebral venous thrombosis. Front Neurol. 2018; 9:882.
12. Poungvarin N, Prayoonwiwat N, Ratanakorn D, Towanabut S, Tantirittisak T, Suwanwela N, et al. Thai venous stroke prognostic score: TV-SPSS. J Med Assoc Thai. 2009; 92:1413–1422.
13. Koopman K, Uyttenboogaart M, Vroomen PC, van der Meer J, De Keyser J, Luijckx GJ. Development and validation of a predictive outcome score of cerebral venous thrombosis. J Neurol Sci. 2009; 276:66–68.
14. Barinagarrementeria F, Cantu C, Arredondo H. Aseptic cerebral venous thrombosis: proposed prognostic scale. J Stroke Cerebrovasc Dis. 1992; 2:34–39.
15. Eskey CJ, Meyers PM, Nguyen TN, Ansari SA, Jayaraman M, McDougall CG, et al. Indications for the performance of intracranial endovascular neurointerventional procedures: a scientific statement from the American Heart Association. Circulation. 2018; 137:e661–e689.
16. Ferro JM, Coutinho JM, Dentali F, Kobayashi A, Alasheev A, Canhão P, et al. Safety and efficacy of dabigatran etexilate vs dose-adjusted warfarin in patients with cerebral venous thrombosis: a randomized clinical trial. JAMA Neurol. 2019; 76:1457–1465.
17. Yaghi S, Shu L, Bakradze E, Salehi Omran S, Giles JA, Amar JY, et al. Direct oral anticoagulants versus warfarin in the treatment of cerebral venous thrombosis (ACTION-CVT): a multicenter international study. Stroke. 2022; 53:728–738.
18. Alimohammadi A, Kim DJ, Field TS. Updates in cerebral venous thrombosis. Curr Cardiol Rep. 2022; 24:43–50.
19. Shu L, Bakradze E, Salehi Omran S, Giles JA, Amar JY, Henninger N, et al. Predictors of recurrent venous thrombosis after cerebral venous thrombosis: analysis of the ACTION-CVT Study. Neurology. 2022 Forthcoming.
20. Siegler JE, Shu L, Yaghi S, Salehi Omran S, Elnazeir M, Bakradze E, et al. Endovascular therapy for cerebral vein thrombosis: a propensity-matched analysis of anticoagulation in the treatment of cerebral venous thrombosis. Neurosurgery. 2022; Aug. 23. [Epub].
https://doi.org/10.1227/neu.0000000000002098.
21. Liberman AL, Kamel H, Mullen MT, Messé SR. International Classification of Diseases, ninth revision (ICD-9) diagnosis codes can identify cerebral venous thrombosis in hospitalized adults. Neurohospitalist. 2016; 6:147–150.
22. Handley JD, Emsley HC. Validation of ICD-10 codes shows intracranial venous thrombosis incidence to be higher than previously reported. Health Inf Manag. 2020; 49:58–61.
23. Burke JF, Freedman VA, Lisabeth LD, Brown DL, Haggins A, Skolarus LE. Racial differences in disability after stroke: results from a nationwide study. Neurology. 2014; 83:390–397.
25. Canhão P, Ferro JM, Lindgren AG, Bousser MG, Stam J, Barinagarrementeria F, et al. Causes and predictors of death in cerebral venous thrombosis. Stroke. 2005; 36:1720–1725.
26. Girot M, Ferro JM, Canhão P, Stam J, Bousser MG, Barinagarrementeria F, et al. Predictors of outcome in patients with cerebral venous thrombosis and intracerebral hemorrhage. Stroke. 2007; 38:337–342.
27. Otite FO, Patel S, Sharma R, Khandwala P, Desai D, Latorre JG, et al. Trends in incidence and epidemiologic characteristics of cerebral venous thrombosis in the United States. Neurology. 2020; 95:e2200–e2213.
28. Williams O, Ovbiagele B. Stroking out while Black: the complex role of racism. JAMA Neurol. 2020; 77:1343–1344.
29. Levine DA, Duncan PW, Nguyen-Huynh MN, Ogedegbe OG. Interventions targeting racial/ethnic disparities in stroke prevention and treatment. Stroke. 2020; 51:3425–3432.
30. Churchwell K, Elkind MS, Benjamin RM, Carson AP, Chang EK, Lawrence W, et al. Call to action: structural racism as a fundamental driver of health disparities: a presidential advisory from the American Heart Association. Circulation. 2020; 142:e454–e468.
31. Skolarus LE, Sharrief A, Gardener H, Jenkins C, Boden-Albala B. Considerations in addressing social determinants of health to reduce racial/ethnic disparities in stroke outcomes in the United States. Stroke. 2020; 51:3433–3439.
32. Magwood GS, Ellis C, Nichols M, Burns SP, Jenkins C, Woodbury M, et al. Barriers and facilitators of stroke recovery: perspectives from African Americans with stroke, caregivers and healthcare professionals. J Stroke Cerebrovasc Dis. 2019; 28:2506–2516.
33. Barlas RS, Honney K, Loke YK, McCall SJ, Bettencourt-Silva JH, Clark AB, et al. Impact of hemoglobin levels and anemia on mortality in acute stroke: analysis of UK regional registry data, systematic review, and meta-analysis. J Am Heart Assoc. 2016; 5:e003019.
34. Silvis SM, Reinstra E, Hiltunen S, Lindgren E, Heldner MR, Mansour M, et al. Anaemia at admission is associated with poor clinical outcome in cerebral venous thrombosis. Eur J Neurol. 2020; 27:716–722.
35. Liu K, Pei L, Gao Y, Zhao L, Fang H, Bunda B, et al. Dehydration status predicts short-term and long-term outcomes in patients with cerebral venous thrombosis. Neurocrit Care. 2019; 30:478–483.
36. Coutinho JM, Ferro JM, Canhão P, Barinagarrementeria F, Cantú C, Bousser MG, et al. Cerebral venous and sinus thrombosis in women. Stroke. 2009; 40:2356–2361.
37. Ruiz-Sandoval JL, Chiquete E, Bañuelos-Becerra LJ, Torres-Anguiano C, González-Padilla C, Arauz A, et al. Cerebral venous thrombosis in a Mexican multicenter registry of acute cerebrovascular disease: the RENAMEVASC study. J Stroke Cerebrovasc Dis. 2012; 21:395–400.
38. Coutinho JM, Zuurbier SM, Bousser MG, Ji X, Canhão P, Roos YB, et al. Effect of endovascular treatment with medical management vs standard care on severe cerebral venous thrombosis: the TO-ACT Randomized Clinical Trial. JAMA Neurol. 2020; 77:966–973.
39. Quinn TJ, Dawson J, Walters MR, Lees KR. Reliability of the modified Rankin Scale: a systematic review. Stroke. 2009; 40:3393–3395.
40. Dentali F, Poli D, Scoditti U, Di Minno MN, De Stefano V, Siragusa S, et al. Long-term outcomes of patients with cerebral vein thrombosis: a multicenter study. J Thromb Haemost. 2012; 10:1297–1302.
41. Nguyen TN, Qureshi MM, Klein P, Yamagami H, Abdalkader M, Mikulik R, et al. Global impact of the COVID-19 pandemic on cerebral venous thrombosis and mortality. J Stroke. 2022; 24:256–265.
42. Siegler JE, Abdalkader M, Michel P, Nguyen TN. Therapeutic trends of cerebrovascular disease during the COVID-19 pandemic and future perspectives. J Stroke. 2022; 24:179–188.





 PDF
PDF Citation
Citation Print
Print



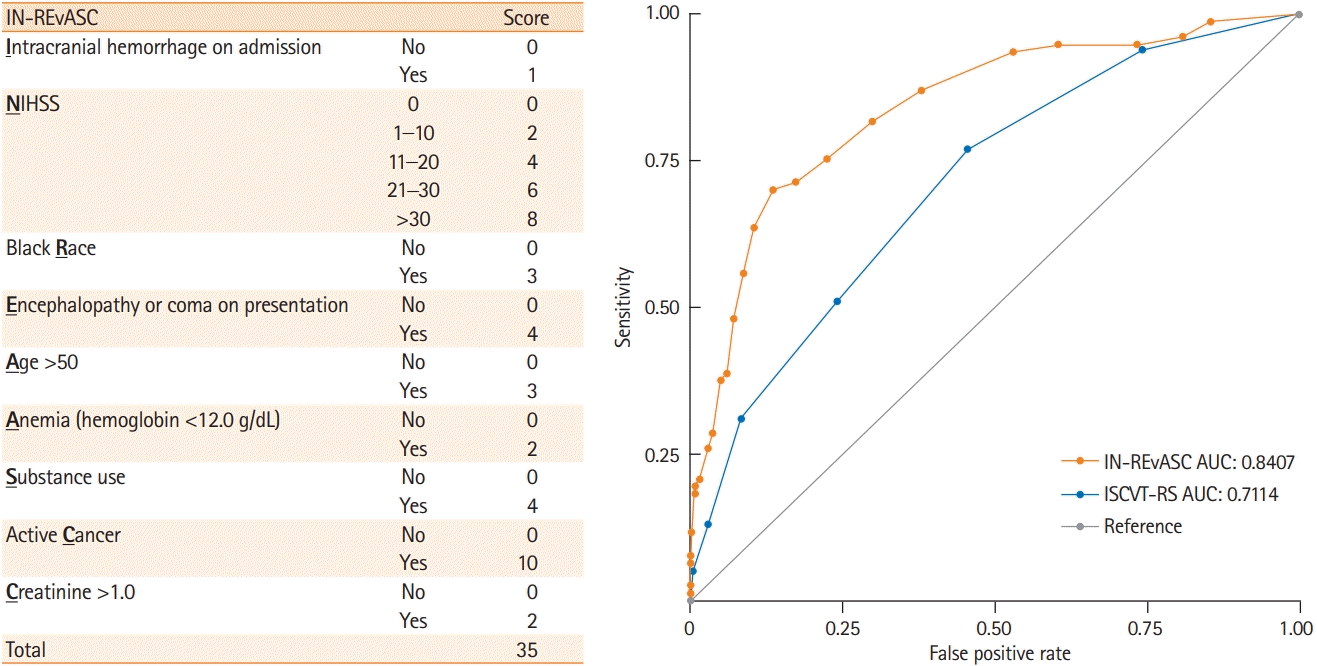
 XML Download
XML Download