1. Triadafilopoulos G, Hallstone A, Nelson-Abbott H, Bedinger K. Oropharyngeal and esophageal interrelationships in patients with nonobstructive dysphagia. Dig Dis Sci. 1992; 37:551–7.

2. Liu LW, Andrews CN, Armstrong D, Diamant N, Jaffer N, Lazarescu A, et al. Clinical practice guidelines for the assessment of uninvestigated esophageal dysphagia. J Can Assoc Gastroenterol. 2018; 1:5–19.

3. Cockeram AW. Canadian Association of Gastroenterology practice guidelines: evaluation of dysphagia. Can J Gastroenterol. 1998; 12:409–13.

4. Yoon SW, Kim KW, Park HJ, Kim EK, Yu JS, Seo JK, et al. The usefulness of esophagogram with marshmallow bolus in patients with esophageal-related symptoms. J Korean Radiol Soc. 1996; 34:399–404.

5. Smith DF, Ott DJ, Gelfand DW, Chen MY. Lower esophageal mucosal ring: correlation of referred symptoms with radiographic findings using a marshmallow bolus. AJR Am J Roentgenol. 1998; 171:1361–5.

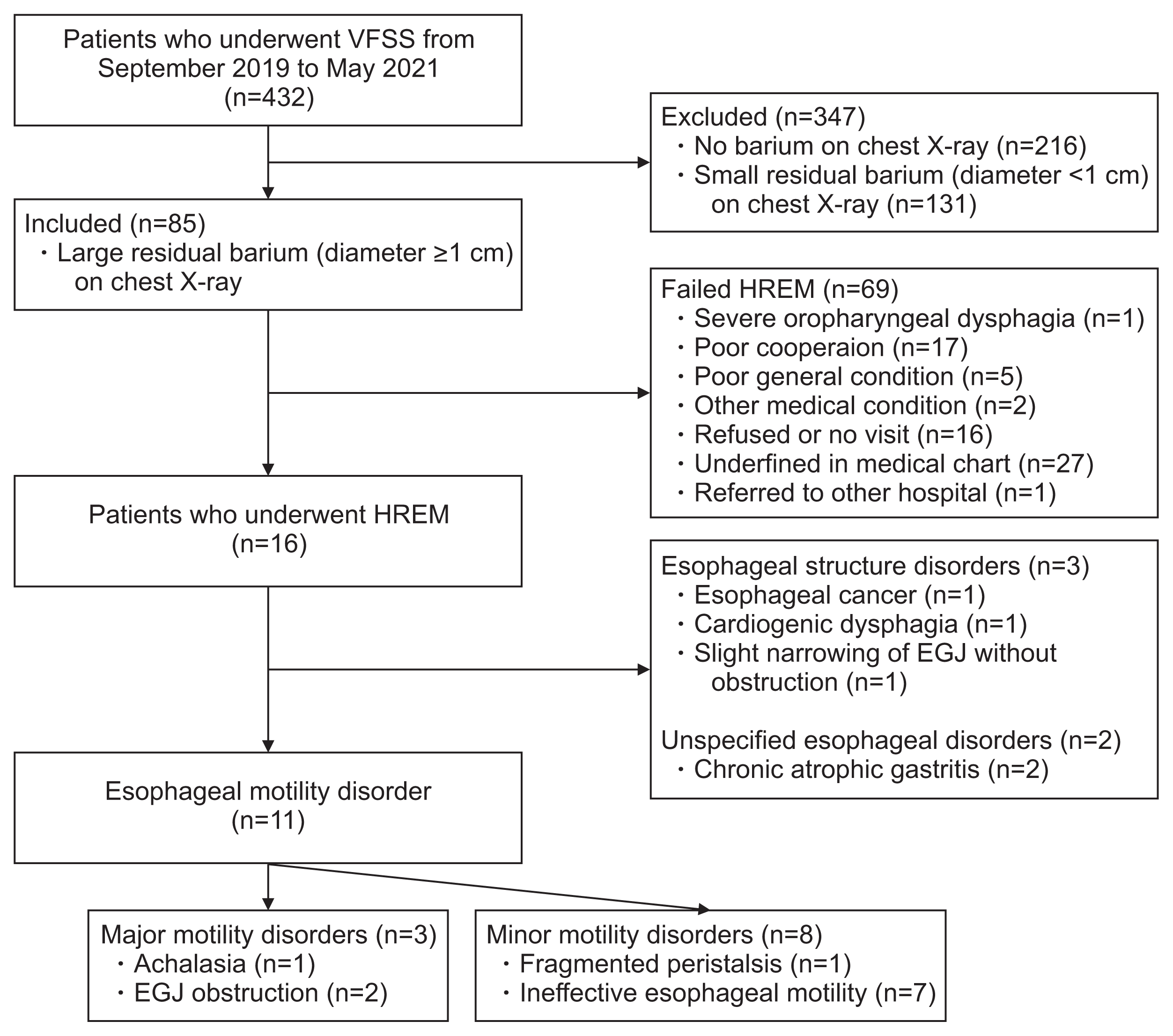
6. Min YK, Baek S, Kang EK, Nam SJ. Characteristics of patients with esophageal dysphagia assessed by chest x-ray imaging after videofluoroscopic swallowing study. Ann Rehabil Med. 2020; 44:38–47.

7. Kahrilas PJ, Bredenoord AJ, Fox M, Gyawali CP, Roman S, Smout AJ, et al. The Chicago classification of esophageal motility disorders, v3.0. Neurogastroenterol Motil. 2015; 27:160–74.

8. Blonski W, Kumar A, Feldman J, Richter JE. Timed barium swallow: diagnostic role and predictive value in untreated achalasia, esophagogastric junction outflow obstruction, and non-achalasia dysphagia. Am J Gastroenterol. 2018; 113:196–203.

9. Kostic S, Andersson M, Hellstrom M, Lonroth H, Lundell L. Timed barium esophagogram in the assessment of patients with achalasia: reproducibility and observer variation. Dis Esophagus. 2005; 18:96–103.

10. Rosenbek JC, Robbins JA, Roecker EB, Coyle JL, Wood JL. A penetration-aspiration scale. Dysphagia. 1996; 11:93–8.

11. Park J, Yoo W, Yoo B. Standard recipes for the preparation of thickened barium liquids used in the diagnosis of dysphagia. Clin Nutr Res. 2019; 8:265–71.

12. Richter JE, Wu WC, Johns DN, Blackwell JN, Nelson JL, Castell JA, et al. Esophageal manometry in 95 healthy adult volunteers: variability of pressures with age and frequency of “abnormal” contractions. Dig Dis Sci. 1987; 32:583–92.
13. Varadarajulu S, Eloubeidi MA, Patel RS, Mulcahy HE, Barkun A, Jowell P, et al. The yield and the predictors of esophageal pathology when upper endoscopy is used for the initial evaluation of dysphagia. Gastrointest Endosc. 2005; 61:804–8.

14. Qureshi NA, Hallissey MT, Fielding JW. Outcome of index upper gastrointestinal endoscopy in patients presenting with dysphagia in a tertiary care hospital: a 10 years review. BMC Gastroenterol. 2007; 7:43.
15. Spieker MR. Evaluating dysphagia. Am Fam Physician. 2000; 61:3639–48.
16. Lin SH, Chang JY. Esophageal cancer: diagnosis and management. Chin J Cancer. 2010; 29:843–54.

17. Park J, Baek S, Kim G, Nam SJ, Cho BR. Dysphagia secondary to esophageal compression in a patient with decompensated heart failure. Korean J Helicobacter Up Gastrointest Res. 2022; 22:146–51.

18. Sweis R, Anggiansah A, Wong T, Kaufman E, Obrecht S, Fox M. Normative values and inter-observer agreement for liquid and solid bolus swallows in upright and supine positions as assessed by esophageal high-resolution manometry. Neurogastroenterol Motil. 2011; 23:509-e198.

19. Xiao Y, Nicodeme F, Kahrilas PJ, Roman S, Lin Z, Pandolfino JE. Optimizing the swallow protocol of clinical high-resolution esophageal manometry studies. Neurogastroenterol Motil. 2012; 24:e489–96.

20. Yadlapati R, Kahrilas PJ, Fox MR, Bredenoord AJ, Prakash Gyawali C, Roman S, et al. Esophageal motility disorders on high-resolution manometry: Chicago classification version 4.0. Neurogastroenterol Motil. 2021; 33:e14058.
21. Shinjo Y, Okitsu A, Ukeda I, Miyagi A, Domen K, Koyama T. Effects of posture on subjective swallowing difficulty during screening tests for dysphagia. Int J Phys Med Rehabil. 2013; 1:133.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download