Abstract
Background
Methods
Results
SUPPLEMENTARY MATERIALS
Supplementary Table 1.
Supplementary Table 2.
Supplementary Table 3.
Supplementary Table 4.
Supplementary Table 5.
Supplementary Fig. 1.
Supplementary Fig. 2.
Supplementary Fig. 3.
Notes
CONFLICTS OF INTEREST
The following companies provided study medications, equipment, or supplies: Abbott Laboratories, Amylin Pharmaceutical, AstraZeneca Pharmaceuticals LP, Bayer HealthCare LLC, Closer Healthcare Inc., GlaxoSmithKline Pharmaceuticals, King Pharmaceuticals Inc., Merck & Co. Inc., Novartis Pharmaceuticals Inc., Novo Nordisk Inc., Omron Healthcare Inc., Sanofi-Aventis U.S., and Takeda Pharmaceuticals Inc. However, no potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conception or design: D.N.H., F.R.L.
Acquisition, analysis, or interpretation of data: D.N.H., F.R.L.
Drafting the work or revising: R.Z., J.Z.Z., X.X.W.
Final approval of the manuscript: D.N.H., F.R.L., R.Z., J.Z.Z., X.X.W., X.B.W.
FUNDING
Our study was supported by the National Natural Science Foundation of China (82173607), the Guangdong Basic and Applied Basic Research Foundation (2021A1515011684), Open Project of the Guangdong Provincial Key Laboratory of Tropical Disease Research (2020B1212060042) and Guangzhou Science and Technology Project (202102080597). Funders had no role in the design of the study, the analysis and interpretation of data, and the writing of the paper.
ACKNOWLEDGMENTS
REFERENCES
Fig. 1.
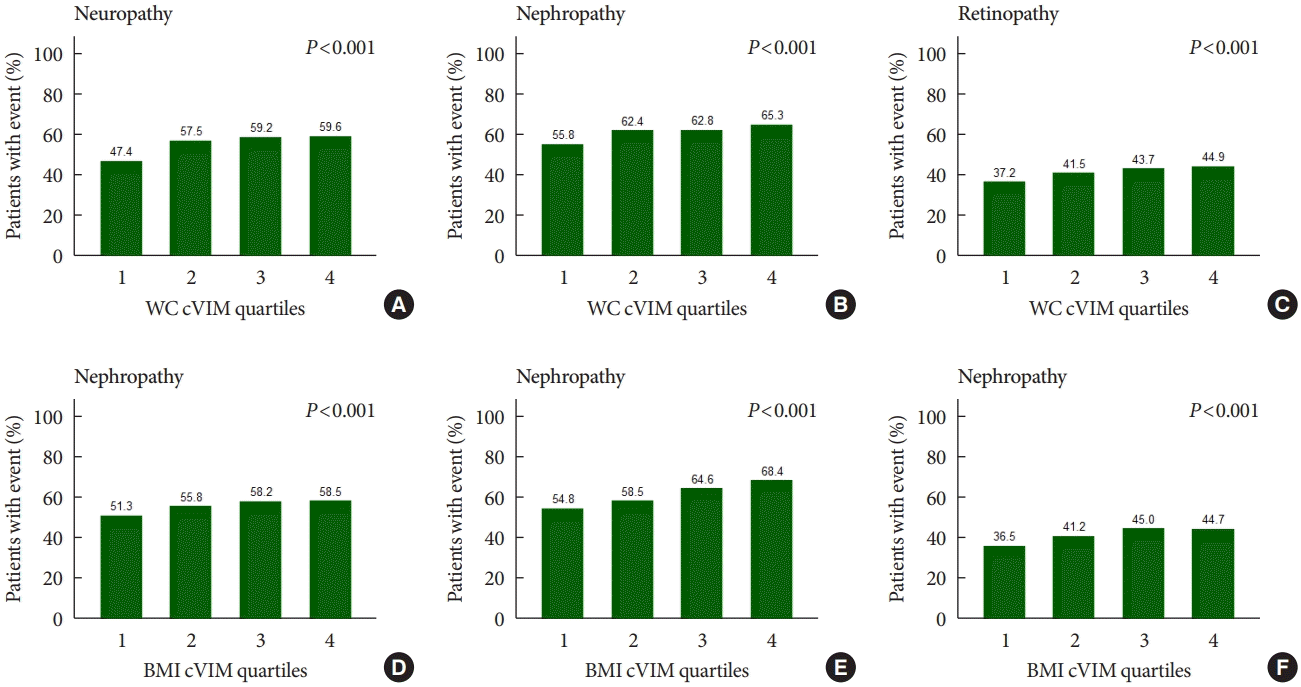
Table 1.
| Characteristic |
Neuropathy |
Nephropathy |
Retinopathy |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No cases (n=3,173) | Cases (n=4,031) | P valuea | No cases (n=3,351) | Cases (n=5,369) | P valuea | No cases (n=3,616) | Cases (n=2,601) | P valuea | ||
| Age, yr | 61.6 (57.4–66.3) | 62.2 (57.7–67.6) | <0.001 | 62.2 (57.7–67.4) | 61.9 (57.7–66.9) | 0.196 | 60 (56.6–64.4) | 61.8 (57.6–66.2) | <0.001 | |
| Sex | <0.001 | 0.002 | 0.134 | |||||||
| Male | 1,859 (58.6) | 2,551 (63.3) | 2,007 (59.9) | 3,390 (63.1) | 2,300 (63.6) | 1,606 (61.7) | ||||
| Female | 1,314 (41.4) | 1,480 (36.7) | 1,344 (40.1) | 1,979 (36.9) | 1,316 (36.4) | 995 (38.3) | ||||
| Race | <0.001 | 0.035 | 0.084 | |||||||
| White | 1,644 (51.8) | 2,648 (65.7) | 2,074 (61.9) | 3,443 (64.1) | 2,223 (61.5) | 1,655 (63.6) | ||||
| Non-white | 1,529 (48.2) | 1,383 (34.3) | 1,277 (38.1) | 1,926 (35.9) | 1,393 (38.5) | 946 (36.4) | ||||
| WC, cm | 104.1 (94.7–114) | 106.7 (97.8–115.6) | <0.001 | 105.5 (96.5–115.6) | 106.7 (97.8–116.8) | <0.001 | 106.7 (96.5–116.8) | 105.4 (96.5–114.5) | 0.023 | |
| BMI, kg/m2 | 31.0 (27.5–35.0) | 31.8 (28.2–35.7) | <0.001 | 31.5 (28.1–35.8) | 32.0 (28.3–36.0) | 0.017 | 32.2 (28.4–36.2) | 31.5 (27.9–35.6) | <0.001 | |
| HbA1c, % | 8 (7.5–8.8) | 8.1 (7.6–8.9) | 0.072 | 8.0 (7.5–8.7) | 8.2 (7.6–8.9) | <0.001 | 8.1 (7.5–8.8) | 8.2 (7.6–9) | <0.001 | |
| Duration of diabetes, yr | 8 (4–14) | 9 (5–14) | 0.001 | 9 (5–15) | 9 (5–15) | 0.001 | 7 (4–12) | 9 (5–14) | <0.001 | |
| Glycemia | 0.006 | 0.383 | <0.001 | |||||||
| Standard | 1,511 (47.6) | 2,050 (50.9) | 1,653 (49.3) | 2,700 (50.3) | 1,728 (47.8) | 1,357 (52.2) | ||||
| Intensive | 1,662 (52.4) | 1,981 (49.1) | 1,698 (50.7) | 2,669 (49.7) | 1,888 (52.2) | 1,244 (47.8) | ||||
| Arm of trial | 0.010 | <0.001 | 0.044 | |||||||
| Standard BP | 721 (22.7) | 946 (23.5) | 1,015 (30.3) | 1,001 (18.6) | 788 (21.8) | 640 (24.6) | ||||
| Intensive BP | 808 (25.5) | 898 (22.3) | 742 (22.1) | 1,302 (24.3) | 861 (23.8) | 572 (22.0) | ||||
| Lipid placebo | 805 (25.4) | 1,111 (27.6) | 1,102 (32.9) | 1,218 (22.7) | 990 (27.4) | 683 (26.3) | ||||
| Lipid fibrate | 839 (26.4) | 1,076 (26.7) | 492 (14.7) | 1,848 (34.4) | 977 (27.0) | 706 (27.1) | ||||
| Insulin use | <0.001 | 0.261 | <0.001 | |||||||
| Yes | 863 (27.2) | 1,288 (32.0) | 1,111 (33.2) | 1,843 (34.3) | 931 (25.8) | 812 (31.2) | ||||
| No | 2,310 (72.8) | 2,743 (68.1) | 2,240 (66.9) | 3,526 (65.7) | 2,685 (74.3) | 1,789 (68.8) | ||||
| Statin use | 0.728 | 0.603 | 0.734 | |||||||
| Yes | 2,014 (63.8) | 2,546 (63.4) | 2,120 (63.5) | 3,424 (64.0) | 2,256 (62.6) | 1,611 (62.2) | ||||
| No | 1,143 (36.2) | 1,470 (36.6) | 2,120 (63.5) | 3,424 (64.0) | 1,345 (37.4) | 978 (37.8) | ||||
| Antihypertensive | 0.913 | 0.011 | 0.202 | |||||||
| Yes | 2,695 (84.9) | 3,420 (84.8) | 2,820 (84.2) | 4,625 (86.1) | 3,048 (84.3) | 2,161 (83.1) | ||||
| No | 478 (15.1) | 611 (15.2) | 531 (15.9) | 744 (13.9) | 568 (15.7) | 440 (16.9) | ||||
| Alcohol consumption | 0.004 | 0.333 | 0.254 | |||||||
| Yes | 719 (22.7) | 1,032 (25.6) | 839 (25.1) | 1,296 (24.1) | 941 (26.0) | 644 (24.8) | ||||
| No | 2,453 (77.3) | 2,997 (74.4) | 2,509 (74.9) | 4,072 (75.9) | 2,673 (74.0) | 1,957 (75.2) | ||||
| Cigarette smoking | 0.125 | 0.214 | 0.148 | |||||||
| Yes | 443 (14.0) | 513 (12.7) | 430 (12.8) | 739 (13.8) | 561 (15.5) | 369 (14.2) | ||||
| No | 2,730 (86.0) | 3,518 (87.3) | 2,921 (87.2) | 4,630 (86.2) | 3,055 (84.5) | 2,232 (85.8) | ||||
| CVD history | 0.192 | <0.001 | 0.152 | |||||||
| Yes | 1,029 (32.4) | 1,366 (33.9) | 1,050 (31.3) | 1,939 (36.1) | 1,154 (31.9) | 875 (33.6) | ||||
| No | 2,144 (67.6) | 2,665 (66.1) | 2,301 (68.7) | 3,430 (63.9) | 2,462 (68.1) | 1,726 (66.4) | ||||
| SBP, mm Hg | 136 (125–147) | 135 (125–147) | 0.099 | 133 (123–143) | 136 (125–147) | <0.001 | 134 (124–145) | 136 (126–147) | <0.001 | |
| DBP, mm Hg | 75 (69–82) | 74 (68–82) | <0.001 | 74 (68–81) | 75 (68–82) | 0.071 | 76 (69–83) | 75 (68–82) | 0.017 | |
| HDL, mg/dL | 41 (35–49) | 40 (34–48) | <0.001 | 41 (35–49) | 39 (34–47) | <0.001 | 39 (34–47) | 40 (34–48) | 0.028 | |
| LDL, mg/dL | 102 (82–128) | 101 (81–124) | 0.103 | 102 (82–126) | 100 (80–124) | 0.004 | 102 (81–125) | 103 (83–128) | 0.071 | |
| eGFR, mL/min/1.73 m² | - | - | - | 80.3 (68.6–92.1) | 92.1 (79.6–108.8) | <0.001 | - | - | - | |
| UACR, mg/g | - | - | - | 10 (6–22) | 15 (7–44) | <0.001 | - | - | - | |
| Serum creatinine, mg/dL | - | - | - | 0.9 (0.8–1.1) | 0.8 (0.7–1.0) | <0.001 | - | - | - | |
Values are presented as median (interquartile range) or number (%).
WC, waist circumference; BMI, body mass index; HbA1c, glycosylated hemoglobin; BP, blood pressure; CVD, cardiovascular disease; SBP, systolic blood pressure; DBP, diastolic blood pressure; HDL, high density lipoprotein; LDL, low density lipoprotein; eGFR, estimated glomerular filtration rate; UACR, urinary albumin creatinine ratio.
Table 2.
Model 1: unadjusted model; Model 21: Adjusted for age, sex race, duration of diabetes, allocation to glycemia treatment arm, blood pressure (BP) vs. lipid treatment arm, systolic BP, diastolic BP, low density lipoprotein, high density lipoprotein, glycosylated hemoglobin, insulin use, alcohol consumption, and baseline BMI (or WC).
WC, waist circumference; BMI, body mass index; HR, hazard ratio; CI, confidence interval; cVIM, corrected variability independent of mean; ASV, average successive variability; CV, coefficient of variation; SD, standard deviation.
Table 3.
Model 1: unadjusted model; Model 22: Adjusted for age, sex, race, duration of diabetes, blood pressure (BP) vs. lipid treatment arm, systolic BP, diastolic BP, low density lipoprotein, high density lipoprotein, glycosylated hemoglobin, antihypertensive, cardiovascular disease history, estimated glomerular filtration rate, urinary albumin creatinine ratio, and baseline BMI (or WC).
WC, waist circumference; BMI, body mass index; HR, hazard ratio; CI, confidence interval; cVIM, corrected variability independent of mean; ASV, average successive variability; CV, coefficient of variation; SD, standard deviation.
Table 4.
Model 1: unadjusted model; Model 23: Adjusted for age, sex, race, duration of diabetes, allocation to glycemia treatment arm, blood pressure (BP) vs. lipid treatment arm, systolic BP, diastolic BP, low density lipoprotein, high density lipoprotein, glycosylated hemoglobin, insulin use, and baseline BMI (or WC).
WC, waist circumference; BMI, body mass index; HR, hazard ratio; CI, confidence interval; cVIM, corrected variability independent of mean; ASV, average successive variability; CV, coefficient of variation; SD, standard deviation.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download