Abstract
Fetal alcohol syndrome (FAS) is a developmental and congenital disorder characterized by neurocognitive impairment, structural defects, and growth restriction due to prenatal alcohol exposure. The estimated global prevalence of alcohol use during pregnancy is 9.8%, and the estimated prevalence of FAS in the general population is 14.6 per 10,000 people. In Korea, the estimated prevalence of alcohol use during pregnancy is 16%, and the prevalence of FAS is 18–51 per 10,000 women, which is higher than the global prevalence. Women’s alcohol consumption rates have increased, especially in women of childbearing age. This could increase the incidence of FAS, leading to higher medical expenses and burden on society. Alcohol is the single most important teratogen that causes FAS, and there is no safe trimester to drink alcohol and no known safe amount of alcohol consumption during pregnancy. Thus, physicians should assess women’s drinking patterns in detail and provide education on FAS to women by understanding its pathophysiology. Moreover, the prevention of FAS requires long-term care with a multidisciplinary approach.
Fetal alcohol syndrome (FAS) is a congenital fetal disorder caused by maternal alcohol consumption during pregnancy. Children with FAS present with characteristic facial features, growth retardation, and intellectual disability and may even have difficulties adjusting to society later by fetal programming [1–3]. Alcohol can affect fetal development in various ways, including any amount of alcohol consumed from 3 months before conception to the end of pregnancy. Some children are born with all the features of FAS, whereas others have only some malformations, especially abnormalities of the central nervous system (CNS).
The global prevalence of alcohol use during pregnancy is reported to be 9.8%, and the estimated prevalence of FAS in the general population is 14.6 per 10,000 people (Table 1) [4–7]. Moreover, it is estimated that one in every 67 women who consumed alcohol during pregnancy would have a child with FAS, which translates to approximately 119,000 children born with FAS worldwide annually [4]. In Korea, the estimated prevalence of FAS is 0.18–0.51% in general schools and 14.9% in facilities for kids with mental retardation according to an epidemiological survey in 2012 [8]. According to Lee et al. [9], approximately 16% of women were reported to drink alcohol during pregnancy, and 1.7% reported binge drinking in Korea. Moreover, the annual drinking rate among Korean women (over 19 years old) was higher in 2015 (70.8%) than in 1998 (59.3%) according to data from the Korea Health Promotion Institute [1]. The increasing rate of child-bearing-aged women who consume alcohol can be considered to increase the chance of alcohol exposure during pregnancy, whether intentionally or unintentionally. Thus, the potential prevalence of FAS may also increase in Korea.
Alcohol consumption during pregnancy is harmful. FAS was first described in French medical literature by Lemoine et al. in 1968 [10]. This was explained based on the abnormal findings observed in 127 children of alcoholic parents at the time [10]. Five years later, Jones et al. (1973) systemically described the association between maternal alcohol abuse and certain types of birth defects and provided the first diagnostic criteria for this condition [11]. This has always been cited in basic and clinical research articles that have identified alcohol-related malformations. In 2005, Hoyme et al. [12] published practical guidelines for operating the Institute of Medicine (IOM) categories, allowing for the standardization of fetal alcohol spectrum disorder (FASD) diagnoses in clinical settings [11]. The updated clinical guidelines for the diagnosis of FASD were published in 2016 [12]. They described four distinct diagnostic categories within FASD: FAS, partial fetal alcohol syndrome (PFAS), alcohol-related neurodevelopmental disorder (ARND), and alcohol-related birth defects (ARBD) (Table 2) [12].
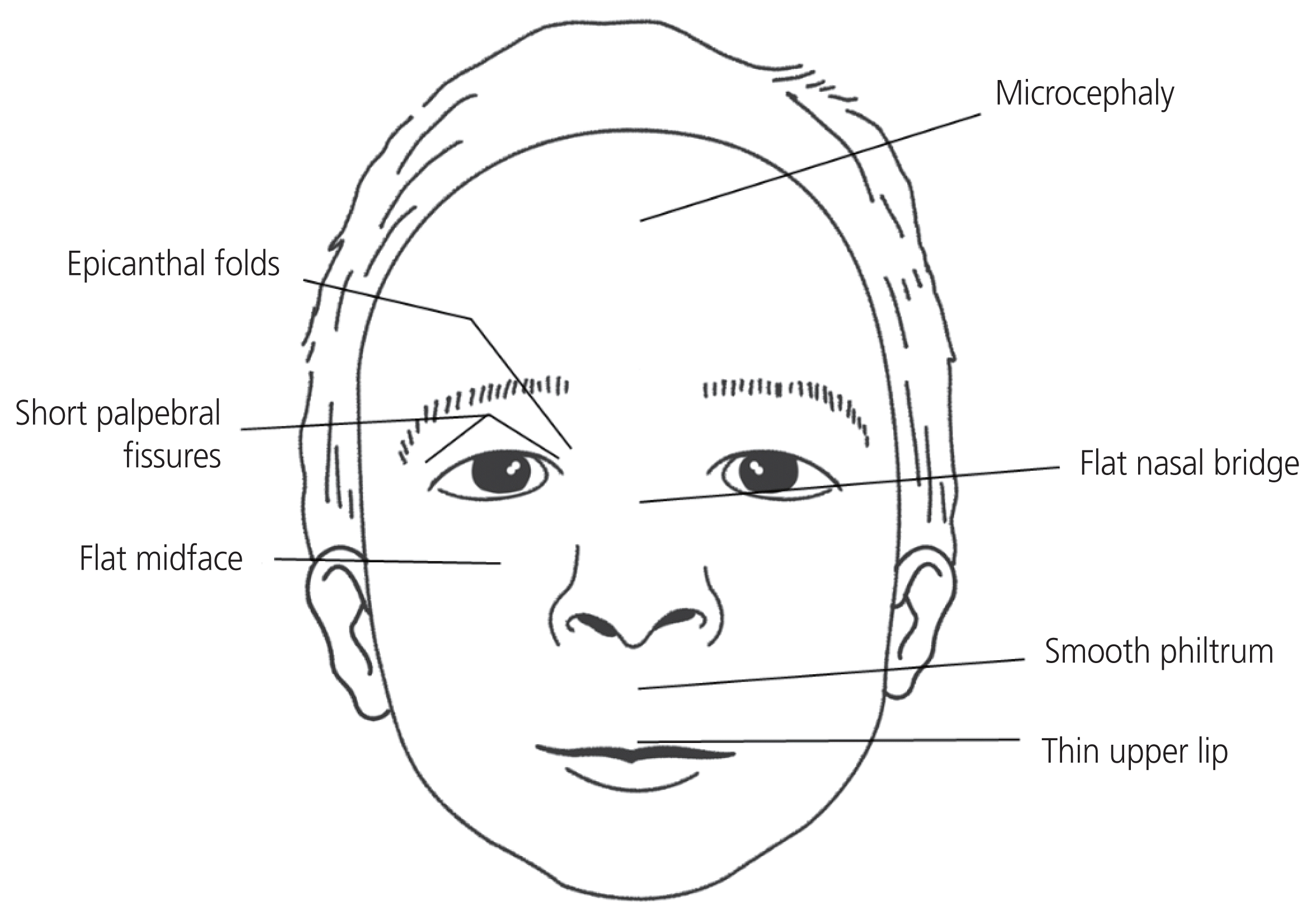
FAS and FASD result from prenatal alcohol exposure and are related to physical malformations and intellectual disabilities [13,14]. Children with FAS have CNS abnormalities, pre- or postnatal growth impairment, and characteristic facial abnormalities [12,13]. CNS abnormalities include microcephaly, tremors, hyperactivity, lack of motor skills, deficits in attention, learning difficulties, intellectual or cognitive impairment, and seizures [12]. Facial abnormalities include short palpebral fissures, epicanthal folds, flat midface, hypoplastic philtrum, and a thin upper vermilion border (Fig. 1) [12,13]. Children with FAS are commonly diagnosed 48.3 months after birth [14]. However, it is often missed or misdiagnosed, preventing affected children from receiving the required services promptly [15].
Clinicians should consider a detailed history of prenatal alcohol exposure to diagnose FAS. Significant prenatal alcohol exposure that can affect the fetus is defined as at least 1 of the following documented findings: (a) 6 or more drinks per week for 2 or more weeks during pregnancy; (b) 3 or more drinks per occasion on 2 or more occasions during pregnancy; (c) alcohol-related social or legal problems during pregnancy; (d) intoxication during pregnancy documented by blood, breath, or urinary alcohol tests; (e) positive test for alcohol exposure biomarkers during pregnancy (fatty acid ethyl esters, phosphatidylethanol, and ethyl glucuronide in maternal hair, fingernails, urine, or blood, or in the placenta or meconium); and (f) increased prenatal risk associated with alcohol use during pregnancy, as assessed by a validated screening tool [12]. If there was no history of prenatal alcohol exposure during the 3 months before pregnancy recognition or at the time of a positive pregnancy test, FAS could be excluded. However, FAS cannot be ruled out if there is a history of alcohol exposure during pregnancy.
FASD is a broad diagnosis that includes FAS, PFAS, ARND, and ARBD according to IOM diagnostic criteria (Table 2) [12]. FAS is the most severe form of the FASD [14]. Another diagnostic strategy is a 4-digit diagnostic code created from the clinical data of the Washington State Fetal Alcohol Syndrome Diagnostic and Prevention Network [16,17]. It reflects the severity of the 4 key diagnostic features of FAS: growth deficiency, FAS facial phenotype, CNS dysfunction, and gestational exposure to alcohol (Table 3) [17]. This is objective and can be useful for surveillance and research purposes. Moreover, Canadian guidelines exist for the diagnosis of FAS [18]. This is similar to the IOM diagnostic category, but the definition of partial FAS is different [18]. There are also other guidelines and checklists for the diagnosis of FASD [19,20]. Differential diagnoses for FASD should include various chromosomal abnormalities, exposure to other teratogenic substances, and behavioral and psychiatric diagnoses, for example, Turner’s syndrome, fragile X syndrome, William’s syndrome, Noonan’s syndrome, DiGeorge syndrome, Zikavirus infection, and diseases by other teratogens (hydantoin, valproate, etc.) [14,21–23].
Clinicians should also identify maternal high-risk groups for prenatal alcohol exposure according to their drinking patterns. Different patterns of alcohol consumption during pregnancy can have various effects on the fetus. Specifically, animal and human studies have shown that binge drinking is more detrimental to fetal development than constant drinking [24–27]. Binge drinking is defined as the consumption of 5 or more drinks on a single occasion (a standard drink is defined as approximately 14 g of pure alcohol) [24,28]. This is because a higher peak blood alcohol concentration worsens fetal brain damage and leads to prolonged alcohol exposure; therefore, metabolizing all the alcohol that has been consumed takes time [24]. Recently, several studies have objectively assessed the patterns of maternal alcohol consumption and identified infants who exhibit FAS-related deficits in growth by biological analysis [29–32].
Alcohol is a potent teratogen [13]. Clinicians should consider the following in determining the teratogenicity of substances: completely characteristic defects, sufficient amounts to affect embryo or fetal development, exposure to critical developmental periods, and evidence of epidemiological investigation [33]. Various processes are involved in the effects of alcohol on the fetus-teratogenic mechanism [34–36]. These processes produce variable outcomes, including stillbirth, structural anomalies in infancy, and neurobehavioral disorders in adolescence (Fig. 2).
First, drinking alcohol and hypoxia are related, and hypoxia is a primary cause of cellular damage [37]. If a mother drinks alcohol and the alcohol is metabolized in the liver, the amount of oxygen in the circulation would significantly decrease. Thus, hypoxia may affect cell damage during fetal development, and this process can explain abortions related to alcohol exposure [37]. Specifically, it can affect the developing brain, such as the hippocampus and cerebellum, which are sensitive to hypoxia and alcohol exposure [37].
Second, free radicals can cause significant damage to cells because they are unstable and reactive [37]. They cause oxidative stress and disrupt stable lipids, proteins, receptors, and chromosomes [37]. In general, fetal cells are more sensitive to oxidative stress because of their lower levels of antioxidants and related enzymes [38]. Specifically, craniofacial and visceral structures are derived from neural crest cells; this sensitivity could account for the characteristic malformations associated with FAS [38]. Facial dysplasia, a prominent feature of FAS, appears to occur when peak blood alcohol levels occur during the embryonic stage of prenatal development [39]. This was also explained by the fact that mice exposed to ethanol on embryonic day 7 or 8 exhibited FAS-related facial dysplasia [39].
Brain damage in infants with FAS includes microcephaly, agenesis of the corpus callosum and anterior commissure, and anomalies in the cerebellum and brainstem [40]. According to Olney [41], the administration of a single high dose of ethanol to neonatal rats significantly reduced the thickness of the corpus callosum. Alcohol disrupts the rapid growth of the brain in the 3rd trimester of gestational age, which is characterized by glial development, synaptogenesis, and development of the cerebellum [27,42]. Thus, prenatal alcohol exposure can lead to learning and memory deficits, as well as long-term and neurobehavioral dysfunction [42,43].
Investigating the use of other potential teratogens during pregnancy is also important [12,44]. Smoking and drinking are highly correlated; therefore, alcoholics are more likely to be heavy smokers [37]. Marijuana, cocaine, and caffeine are also significantly correlated with alcohol consumption during pregnancy [37]. Marijuana smoking can increase the level of carbon monoxide in the mother’s body, causing hypoxia and thus increasing the risk of FAS [37]. Cocaine causes uterine artery vasoconstriction and exacerbates hypoxic fetal conditions [37]. Prenatal caffeine exposure can exacerbate zinc deficiency and reinforce the effects of alcohol (zinc is an essential trace mineral important for cellular replication and protein synthesis and is a cofactor for endogenous antioxidative enzymes) [37].
Variations in the alcohol dehydrogenase (ADH) allele related to alcohol metabolism can influence the risk of alcohol-induced malformations in the fetus [42]. According to Warren and Li [45], the more efficient ADH allele ADH1B*3 protects against FASD, and ADH1B*2 reduces the risk of FAS compared with ADH1B*1. Differences in the isoforms of aldehyde dehydrogenase (ALDH), an enzyme that converts acetaldehyde to acetate, also influence the risk of FAS [37]. If the inactive form of ALDH (ALDH2*2), acetaldehyde, is not converted to acetate in the body, it accumulates and causes flushing, tachycardia, and nausea [37]. ALDH2*2, an inactive form of ALDH, is mainly present in Asian populations. Thus, we can consider racial differences in FAS severity, and the delayed breakdown of alcohol can affect the fetus and induce malformations. Asian mothers may need to be more educated on alcohol abstinence than others. Furthermore, studies have shown differential sensitivity to FAS in dizygotic twins and high concordance for FAS in monozygotic twins [37].
Several methods can be employed to explore the teratogenicity of alcohol, and established medical databases can be used as valuable resources for retrospective cohort studies of FAS [46]. However, cohort studies that use past medical databases have systematic errors, such as selection bias, information bias (misclassification), and confounding factors [46]. Moreover, morphological evaluation for the diagnosis of FAS has limitations such as racial differences [11]. Questionnaires are also primarily used to obtain information on pregnant women about alcohol consumption before or during pregnancy. However, this estimate of alcohol use using questionnaires is often underreported because pregnant women are concerned about social stigma [31,47]. Therefore, many studies are being conducted to identify methods to objectively evaluate alcohol exposure in pregnant women [31,32,48,49]. Furthermore, assembling prospective cohorts to observe the occurrence of FAS is expensive and inefficient in terms of time, money, and resources because of the rarity of birth defects related to FAS [46]. Thus, a case-control design is usually used to study FAS; however, the results from such studies could be limited in terms of generalization [46].
Clinical trials for FAS in humans are also difficult. Pregnant women rarely participate in randomized studies on alcohol or other drugs, so there is a lack of data on the safety or prenatal drug exposure itself [46]. Moreover, there are clear limitations to animal experiments because alcohol metabolism is species specific. Therefore, new research methodologies have been investigated recently to verify the negative effects of substances by organizing and analyzing sporadic big data through machine learning [50].
FAS is a disease in which differential diagnosis is important. FAS-related features that can be identified by prenatal ultrasonography include fetal growth restrictions, microcephaly, and extremity and heart malformations. If characteristic features of FAS are seen during antenatal care, other common causes (e.g., infection, genetic factors, placental insufficiency, and other teratogens) should be differentially diagnosed first. If fetal growth restriction or microcephaly appears, the patterns and degrees of growth restriction should be documented [12]. It is necessary to suspect and evaluate maternal alcohol consumption in cases of fetal growth restriction with microcephaly. During the maternal interview, we should investigate not only maternal alcohol intake but also medical history, nutrition, husband’s alcohol use, and home environment. According to several studies, women who have consumed alcohol during pregnancy have a low educational level, low rates of planned pregnancy, and a low level of knowledge related to the risks of drinking alcohol during pregnancy [9,34,51]. Low educational level and unplanned pregnancy are significant risk factors for alcohol consumption in pregnant women [9]. It is also important to consider the overall drinking pattern just before pregnancy recognition, as it is common for the drinking pattern before 3 months of pregnancy to continue into the 1st trimester [12]. Once maternal alcohol consumption is confirmed, physicians should provide objective information on FAS and educate patients on neutral grounds [52]. Above all, it is important for women to be aware of their overall drinking patterns. Women should be educated about immediate alcohol discontinuation once pregnancy is confirmed and encouraged to undergo antenatal care.
FASD is a developmental disorder with a specific phenotype requiring long-term management [34]. It has a high recurrence rate, and younger siblings tend to be more severely affected [34]. FAS recurrence would reach approximately 75% if mothers continue to drink alcohol in subsequent pregnancies [34]. Thus, sustainable follow-up of the mother and child is required, and a multidisciplinary approach with pediatrics and psychiatric departments is important. If a baby with suspected FAS is born, obstetricians should hand over the clinical information to the pediatrician for continuous assessment and management. Psychiatrists must also participate in treatment until neurobehavioral problems appear in adolescence. Counselors can help potential patients avoid exposure during subsequent pregnancies by intervening in alcohol abuse. Furthermore, a cohort study linked to obstetrics and gynecology, pediatrics, and psychiatry should be conducted to prevent FAS by accumulating data on FAS mothers and children; an example is the Washington State Fetal Alcohol Syndrome Diagnostic & Prevention Network, which started at the University of Washington in Seattle in 1993 [53]. It was sponsored by the centers for disease control and prevention, began diagnosing patients in 1993 and has diagnosed over 3,000 patients to date [53]. They try to prevent FAS through screening, diagnosis, intervention, education, and research [53].
In this way, we should establish a big data cohort at the national and social levels that enables accurate diagnosis and sustainable follow-up of FAS to prevent FAS through a multidisciplinary approach. This multidisciplinary team for the prevention of FAS should consist of obstetricians, gynecologists, pediatricians, psychiatrists, biologic scientists, data professionals, and policymakers. An example of such a multidisciplinary team is the FAS prevention center located at the Ewha Womans University Mokdong Hospital in Korea. It is the first FAS-specialized center in Korea, consisting of physicians, biologic scientists, data professionals, etc. They work together to conduct research on FAS, in addition to counseling and campaigning to prevent FAS, sending missions to developing countries, and publishing books for public education. Moreover, the Korean Mothersafe Counseling Center has been running a campaign regarding abstinence from alcohol and other teratogens for pregnant women.
Alcohol is the single most important factor in FAS, and there is no safe trimester or known safe amount to drink alcohol during pregnancy [12]. FAS is a developmental disorder that can be prevented. Physicians must accurately obtain the medical history of the mother and provide safe antenatal care and education for FAS once maternal alcohol consumption is confirmed. If the prevalence of FAS is decreased, it will be possible to reduce the financial burden on society and unrealized human suffering [12].
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported. Young Ju Kim has been an Editorial Board of Obstetrics & Gynecology Science; however, she was not involved in the peer reviewer selection, evaluation, or decision process of this article. Otherwise, no other potential conflicts of interest relevant to this article were reported.
References
1. Seo BA, Kim SG, Huh SY, Lee DH, An SH, Lee SY, et al. Changes of drinking behavior in Korean pregnancy women for the last 20 year. J Public Health (Oxf). 2021; 43:e632–6.
2. Öztürk HNO, Türker PF. Fetal programming: could intrauterin life affect health status in adulthood? Obstet Gynecol Sci. 2021; 64:473–83.

3. Lee S, Kwon EJ, You YA, Du JE, Jo I, Kim YJ. Long-term effects of pro-opiomelanocortin methylation induced in food-restricted dams on metabolic phenotypes in male rat offspring. Obstet Gynecol Sci. 2020; 63:239–50.

4. Popova S, Lange S, Probst C, Gmel G, Rehm J. Estimation of national, regional, and global prevalence of alcohol use during pregnancy and fetal alcohol syndrome: a systematic review and meta-analysis. Lancet Glob Health. 2017; 5:e290–99.

5. Persson A, Lindmark S, Petersson K, Gabriel E, Thorsell M, Lindström K, et al. Alcohol and illicit and non-medical prescription drug use before and during pregnancy in Stockholm, Sweden: a cross-sectional study. Sex Reprod Healthc. 2021; 29:100622.

6. Miyake Y, Tanaka K, Okubo H, Sasaki S, Arakawa M. Alcohol consumption during pregnancy and birth outcomes: the Kyushu Okinawa maternal and child health study. BMC Pregnancy Childbirth. 2014; 14:79.

7. May PA, Blankenship J, Marais AS, Gossage JP, Kalberg WO, Barnard R, et al. Approaching the prevalence of the full spectrum of fetal alcohol spectrum disorders in a South African population-based study. Alcohol Clin Exp Res. 2013; 37:818–30.

8. Lee HG. Development of diagnostic system for fetal alcohol spectrum disorders (2011E6100200). cheongju: Korea Disease Control and Prevention Agency;c2011. [cited 2022 Apr 24]. Available from: https://nih.go.kr/board.es?mid=a40801000000&cbid=0050&act=view&list_no=20485
.
9. Lee SH, Shin SJ, Won SD, Kim EJ, Oh DY. Alcohol use during pregnancy and related risk factors in Korea. Psychiatry Investig. 2010; 7:86–92.

10. Lemoine P, Harousseau H, Borteyru JP, Menuet JC. Children of alcoholic parents--observed anomalies: discussion of 127 cases. Ther Drug Monit. 2003; 25:132–6.

11. Calhoun F, Warren K. Fetal alcohol syndrome: historical perspectives. Neurosci Biobehav Rev. 2007; 31:168–71.

12. Hoyme HE, Kalberg WO, Elliott AJ, Blankenship J, Buckley D, Marais AS, et al. Updated clinical guidelines for diagnosing fetal alcohol spectrum disorders. Pediatrics. 2016; 138:e20154256.

13. Cunningham FG, Leveno KJ, Bloom SL, Dashe JS, Hoffman BL, Casey BM, et al. Williams obstetrics. 25th ed. New York (NY): McGraw-Hill;2018.
14. Denny L, Coles S, Blitz R. Fetal alcohol syndrome and fetal alcohol spectrum disorders. Am Fam Physician. 2017; 96:515–22.
15. Moberg DP, Bowser J, Burd L, Elliott AJ, Punyko J, Wilton G. Fetal alcohol syndrome surveillance: age of syndrome manifestation in case ascertainment. Birth Defects Res A Clin Mol Teratol. 2014; 100:663–69.

16. Astley SJ, Clarren SK. Diagnostic guide for fetal alcohol syndrome and related conditions: the 4-digit diagnostic code. Seattle (WA): University of Washington;1999.
17. Astley SJ, Clarren SK. Diagnosing the full spectrum of fetal alcohol-exposed individuals: introducing the 4-digit diagnostic code. Alcohol Alcohol. 2000; 35:400–10.

18. Chudley AE, Conry J, Cook JL, Loock C, Rosales T, LeBlanc N. Fetal alcohol spectrum disorder: Canadian guidelines for diagnosis. CMAJ. 2005; 172(5 suppl):S1–21.

19. Cook JL, Green CR, Lilley CM, Anderson SM, Baldwin ME, Chudley AE, et al. Fetal alcohol spectrum disorder: a guideline for diagnosis across the lifespan. CMAJ. 2016; 188:191–7.

20. Burd L, Klug MG, Li Q, Kerbeshian J, Martsolf JT. Diagnosis of fetal alcohol spectrum disorders: a validity study of the fetal alcohol syndrome checklist. Alcohol. 2010; 44:605–14.

21. Douzgou S, Breen C, Crow YJ, Chandler K, Metcalfe K, Jones E, et al. Diagnosing fetal alcohol syndrome: new insights from newer genetic technologies. Arch Dis Child. 2012; 97:812–17.

22. Adams DJ, Clark DA. Common genetic and epigenetic syndromes. Pediatr Clin North Am. 2015; 62:411–26.

23. Adibi JJ, Marques ETA Jr, Cartus A, Beigi RH. Teratogenic effects of the Zika virus and the role of the placenta. Lancet. 2016; 387:1587–90.

24. Maier SE, West JR. Drinking patterns and alcohol-related birth defects. Alcohol Res Health. 2001; 25:168–74.
25. Bonthius DJ, West JR. Alcohol-induced neuronal loss in developing rats: increased brain damage with binge exposure. Alcohol Clin Exp Res. 1990; 14:107–18.

26. Strandberg-Larsen K, Nielsen NR, Grønbaek M, Andersen PK, Olsen J, Andersen AM. Binge drinking in pregnancy and risk of fetal death. Obstet Gynecol. 2008; 111:602–9.

27. Gursky ZH, Savage LM, Klintsova AY. Executive functioning-specific behavioral impairments in a rat model of human third trimester binge drinking implicate prefrontal-thalamo-hippocampal circuitry in fetal alcohol spectrum disorders. Behav Brain Res. 2021; 405:113208.

28. National Institute on Alcohol Abuse and Alcoholism. What is a standard drink? [Internet]. Bethesda (MD): National Institute on Alcohol Abuse and Alcoholism;c2021. [cited 2021 Aug 26]. Available from: https://www.niaaa.nih.gov/alcohols-effects-health/overview-alcohol-consumption/what-standard-drink
.
29. Breunis LJ, Wassenaar S, Sibbles BJ, Aaldriks AA, Bijma HH, Steegers EA, et al. Objective assessment of alcohol consumption in early pregnancy using phosphatidylethanol: a cross-sectional study. BMC Pregnancy Childbirth. 2021; 21:1–7.

30. Maugeri A, Barchitta M, Magnano San Lio R, La Rosa MC, La Mastra C, Favara G, et al. The effect of alcohol on telomere length: a systematic review of epidemiological evidence and a pilot study during pregnancy. Int J Environ Res Public Health. 2021; 18:5038.

31. Adler J, Rissmann A, Kropf S, Mohnicke K, Taneva E, Ansorge T, et al. Estimated prevalence of harmful alcohol consumption in pregnant and nonpregnant women in Saxony-Anhalt (NorthEast Germany) using biomarkers. Alcohol Clin Exp Res. 2021; 45:819–27.

32. Mahnke AH, Sideridis GD, Salem NA, Tseng AM, Carter RC, Dodge NC, et al. Infant circulating microRNAs as biomarkers of effect in fetal alcohol spectrum disorders. Sci Rep. 2021; 11:1429.

34. Paintner A, Williams AD, Burd L. Fetal alcohol spectrum disorders--implications for child neurology, part 2: diagnosis and management. J Child Neurol. 2012; 27:355–62.

35. Kim JY, Lee DY, Lee YJ, Park KJ, Kim KH, Kim JW, et al. Chronic alcohol consumption potentiates the development of diabetes through pancreatic β-cell dysfunction. World J Biol Chem. 2015; 6:1–15.

36. Lee YJ, Kim JY, Lee DY, Park KJ, Kim GH, Kim JE, et al. Alcohol consumption before pregnancy causes detrimental fetal development and maternal metabolic disorders. Sci Rep. 2020; 10:10054.

37. Abel EL, Hannigan JH. Maternal risk factors in fetal alcohol syndrome: provocative and permissive influences. Neurotoxicol Teratol. 1995; 17:445–62.

38. Davis WL, Crawford LA, Cooper OJ, Farmer GR, Thomas DL, Freeman BL. Ethanol induces the generation of reactive free radicals by neural crest cells in vitro. J Craniofac Genet Dev Biol. 1990; 10:277–93.
39. Sulik KK. Genesis of alcohol-induced craniofacial dysmorphism. Exp Biol Med (Maywood). 2005; 230:366–75.

40. Jones KL, Smith DW. Recognition of the fetal alcohol syndrome in early infancy. Lancet. 1973; 302:999–1001.

42. Guerri C, Bazinet A, Riley EP. Foetal alcohol spectrum disorders and alterations in brain and behaviour. Alcohol Alcohol. 2009; 44:108–14.

43. Roediger DJ, Krueger AM, de Water E, Mueller BA, Boys CA, Hendrickson TJ, et al. Hippocampal subfield abnormalities and memory functioning in children with fetal alcohol spectrum disorders. Neurotoxicol Teratol. 2021; 83:106944.

44. Bayih WA, Belay DM, Ayalew MY, Tassew MA, Chanie ES, Feleke DG, et al. The effect of substance use during pregnancy on neonatal outcomes in Ethiopia: a systematic review and meta-analysis. Heliyon. 2021; 7:e06740.

45. Warren KR, Li TK. Genetic polymorphisms: impact on the risk of fetal alcohol spectrum disorders. Birth Defects Res A Clin Mol Teratol. 2005; 73:195–203.

46. Ehrenstein V, Sørensen HT, Bakketeig LS, Pedersen L. Medical databases in studies of drug teratogenicity: methodological issues. Clin Epidemiol. 2010; 2:37–43.

47. Mullally A, Cleary BJ, Barry J, Fahey TP, Murphy DJ. Prevalence, predictors and perinatal outcomes of peri-conceptional alcohol exposure--retrospective cohort study in an urban obstetric population in Ireland. BMC Pregnancy Childbirth. 2011; 11:27.
48. Auriacombe M, Moriceau S, Serre F, Denis C, Micoulaud-Franchi JA, de Sevin E, et al. Development and validation of a virtual agent to screen tobacco and alcohol use disorders. Drug Alcohol Depend. 2018; 193:1–6.

49. O’Keeffe LM, Kearney PM, McCarthy FP, Khashan AS, Greene RA, North RA, et al. Prevalence and predictors of alcohol use during pregnancy: findings from international multicentre cohort studies. BMJ Open. 2015; 5:3006323.

50. Lee JY, Lee YS, Kim DH, Lee HS, Yang BR, Kim MG. The use of social media in detecting drug safety-related new black box warnings, labeling changes, or withdrawals: scoping review. JMIR Public Health Surveill. 2021; 7:e30137.

51. May PA, Baete A, Russo J, Elliott AJ, Blankenship J, Kalberg WO, et al. Prevalence and characteristics of fetal alcohol spectrum disorders. Pediatrics. 2014; 134:855–66.

52. Jasper JD, Goel R, Einarson A, Gallo M, Koren G. Effects of framing on teratogenic risk perception in pregnant women. Lancet. 2001; 358:1237–8.

53. FAS Diagnostic & Prevention Network. FAS DPN [Internet]. Seattle (WA): FAS Diagnostic & Prevention Network;c2021. [cited 2021 Aug 26]. Available from: https://depts.washington.edu/fasdpn
.
Fig. 2
Conceptual framework for fetal alcohol syndrome. There are variable processes in the effects of alcohol on the fetus. These influences produce variable outcomes, from stillbirths, structural anomaly in infancy to neurobehavioral disorders in adolescence. ALDH, aldehyde dehydrogenase.
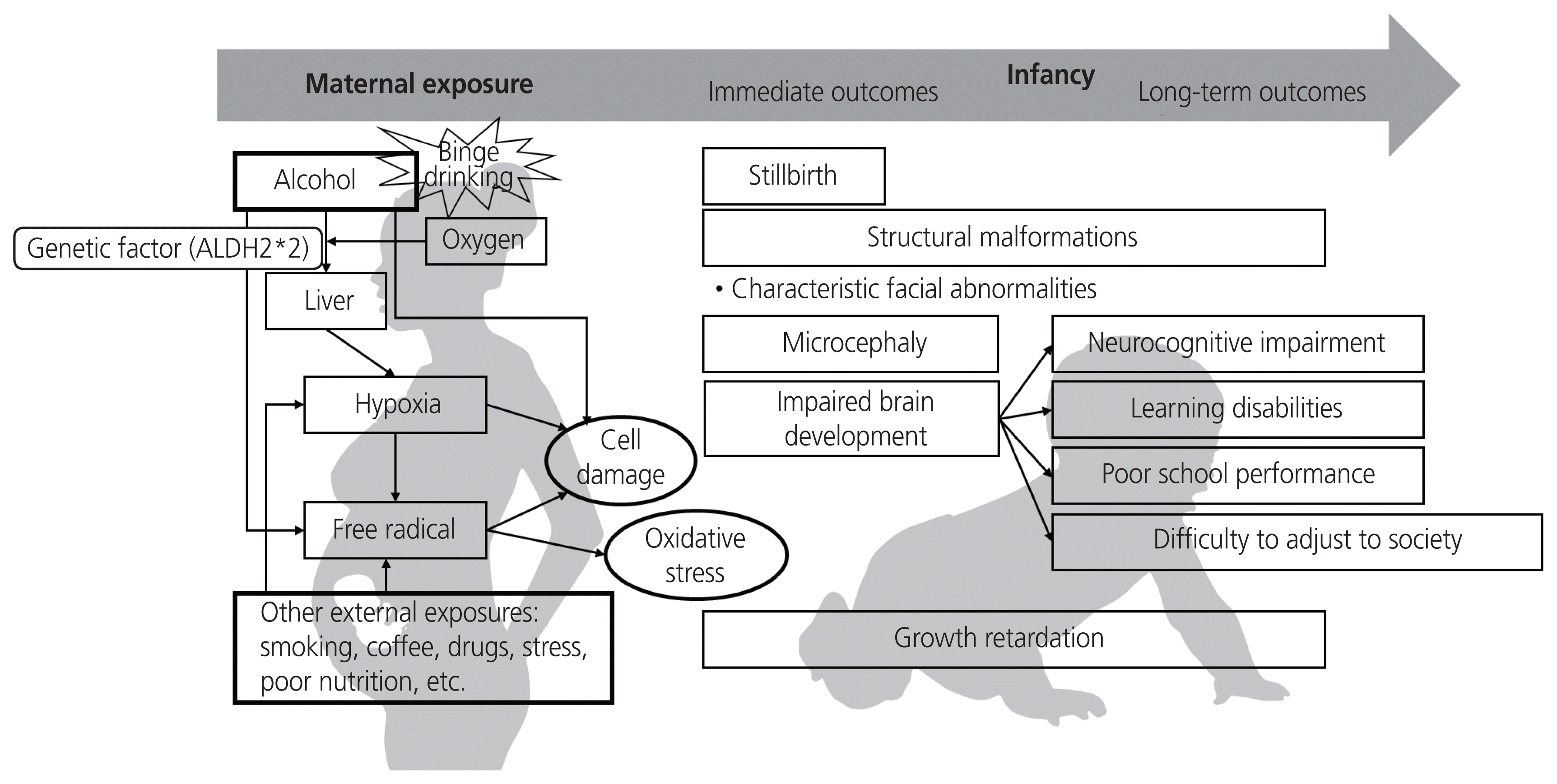
Table 1
Global prevalence of alcohol use (any amount) during pregnancy FAS in the general population in 2012, by WHO region [4]
Table 2
Updated Institute of Medicine (IOM) diagnostic criteria for the diagnosis of fetal alcohol spectrum disorders [12]
Table 3
4-digit diagnostic code criteria for fetal alcohol spectrum disorders [17]




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download