Abstract
Background/Aims
Colorectal polyps are removed to prevent progression to colorectal cancer. Polyp size is an important factor for risk stratification of malignant transformation. Endoscopic size estimation correlates poorly with pathological reports and several factors have been suggested to influence size estimation. We aimed to gain insight into the factors influencing endoscopic polyp size estimation.
Methods
Images of polyps in an artificial model were obtained at 1, 3, and 5 cm from the colonoscope’s tip. Participants were asked to estimate the diameter and volume of each polyp.
Results
Fifteen endoscopists from three large-volume centers participated in this study. With an intraclass correlation coefficient of 0.66 (95% confidence interval [CI], 0.62–0.71) for diameter and 0.56 (95% CI, 0.50–0.62) for volume. Polyp size estimated at 3 cm from the colonoscope’s tip yielded the best results. A lower distance between the tip and the polyp was associated with a larger estimated polyp size.
Colorectal cancer screening programs throughout the world are based on early detection of neoplastic colorectal polyps. It is generally accepted that removal of these polyps prevents progression to colorectal cancer.1,2 Currently, risk stratification of polyp recurrence and surveillance intervals are based on the location, type, number, and size of the polyps.3-8
The importance of polyp size in risk stratification has been reported previously.3 Polyp size ≥10 mm is associated with an increased risk of recurrence of advanced adenomas on follow-up colonoscopy, leading to a shorter time interval in these patients. Moreover, polyp size is strongly associated with the risk of malignant transformation, since non-advanced adenomas (<10 mm) have a very low risk of malignant transformation compared to polyps ≥20 mm in size.9-11 In small (≤5 mm) polyps, the “resect and discard” strategy combines visually estimated polyp size with suspected presence of dysplasia to decide whether a polyp should be retrieved for histopathological examination or can be discarded, saving cost and time.10,12,13
Currently, pathologically measured diameter is the preferred standard for polyp size estimation.14,15 It may be adequate for planning surveillance strategies in retrospect. However, it is not useful in visual endoscopic size estimation. Real-time endoscopic polyp size estimation is crucial, especially in the “resect and discard” situations and in cases of incomplete or piecemeal resection. It has been shown that the accuracy of endoscopic polyp size estimation is poorer compared to that of a pathological report.15-19 Multiple tools to aid in visual polyp size estimation have been used and researched. These include polypectomy snares and biopsy forceps. Studies in this field have been heterogeneous in terms of methods and outcomes. Some studies showed promising improvement, while others showed no improvement in visual polyp size estimation.16,19-23
Factors believed to influence visual endoscopic estimation of polyp size include polyp morphology, individual variations among endoscopists, and terminal digit preference.16-18,24-26 Our study aimed to determine additional factors influencing polyp size estimation using a real-time endoscopic model with known polyp sizes.
A colon model was created, which had artificial polyps of different sizes, shapes, and volumes (Fig. 1). Eight dissimilar polyps including five pedunculated polyps (with short stalks) and three nonpedunculated polyps were created using self-hardening DAS modelling clay (Fabbrica Italiana Lapis ed Affini, Florence, Italy). The exact diameter was measured using a caliper. The diameters were 5, 8, 10, 15, and 20 mm for pedunculated polyps and 30, 35, and 38 mm for nonpedunculated polyps. The volume of each polyp was calculated using the density of the clay and the exact volumes were 58, 236, 710, 2,115, 3,755, 1,704, 4,857, and 6,104 mm3. The colon model was created using a tube with an inner diameter of 65 mm. The artificial polyps were affixed inside the tube. Images were obtained using an Olympus video colonoscope (Olympus EVIS EXERA III CF-HQ190L; Olympus, Hamburg, Germany) at fixed distances of 1, 3, and 5 cm (Fig. 2).
A web-based questionnaire was presented to 32 endoscopists from three large-volume endoscopy centers: Medisch Spectrum Twente in Enschede (The Netherlands), Reinier de Graaf Gasthuis in Delft (The Netherlands), and Gold Coast University Hospital in Southport (Queensland, Australia). The invited participants included nurse endoscopists, gastroenterologists in training, and experienced gastroenterologists. Images of the polyps were included in the questionnaire and presented twice in a random fashion (total 48 images). The participants were unaware of the distance between the tip of the colonoscope and the polyps. The inner diameter of the colon model was provided to the endoscopists at the start of the questionnaire. Additionally, an image of a dice with corresponding diameter and volume was provided as a reference object. A reminder to complete the questionnaire was sent to the participants after 2, 6, and 10 weeks and participation was closed after 12 weeks.
Continuous variables were presented as means with ranges and categorical variables were presented as numbers with corresponding percentages. The intraclass correlation coefficient (ICC) was used to assess the agreement between the estimated and true (measured) sizes of pedunculated and nonpedunculated polyps in terms of diameter and volume. Furthermore, the polyps were divided into three categories based on their diameter (≤5, 6–9, and ≥10 mm), since these categories were considered clinically relevant.3,10,13 The level of agreement between the visually estimated diameter category and the true (measured) category was assessed using the kappa test. Strength of agreement was interpreted according to the method described by Koo and Li,27 with values <0.5, 0.5–0.75, 0.75–0.90, and >0.90 indicating poor, moderate, good, and excellent reliability, respectively (Table 1). All images were presented twice and were used as independent images in the analyses.
Among the 32 invited endoscopists, 15 (47%) responded and participated in the study. These included one nurse endoscopist, four gastroenterologists in training, and 10 experienced gastroenterologists. The average age was 39 years (range, 29–58 years) and seven out of 15 participants (47%) were male. Nine out of 15 participants (60%) had endoscopic experience of >48 months and seven out of 15 (47%) performed >300 colonoscopies annually.
The level of agreement between the visually estimated polyp size (via endoscopy) and measured size was moderate. The ICC was 0.66 (95% confidence interval [CI], 0.62–0.71) for diameter and 0.56 (95% CI, 0.50–0.62) for volume. Figure 3 shows the visually estimated and measured polyp sizes in terms of diameter and volume.
After categorization of polyp size (≤5, 6–9, and ≥10 mm), the agreement between the estimated and measured size categories was poor (kappa, 0.49 [95% CI, 0.43–0.56]) (Table 2). Endoscopic experience of >48 months was associated with slightly better ICCs for diameter (0.70 [95% CI, 0.64–0.75] vs. 0.60 [95% CI, 0.50–0.68]), but not for volume (0.57 [95% CI, 0.50–0.64] vs. 0.54 [95% CI, 0.43–0.63]).
There were small differences in the accuracy of polyp size estimation when the distance between the tip of the colonoscope and the polyp was considered. Polyp size estimation at 3 cm and 5 cm showed comparable ICCs of 0.71 (95% CI, 0.63–0.77) and 0.73 (95% CI, 0.66–0.79), respectively when compared with polyp size estimation at 1 cm (ICC, 0.62 [95% CI, 0.52–0.70]). After categorizing the polyp size, the kappa-values were 0.54 (95% CI, 0.43–0.65), 0.50 (95% CI, 0.40–0.60), and 0.43 (95% CI, 0.32–0.54) at 3, 5, and 1 cm, respectively. A smaller distance between the tip of the colonoscope and the polyp was associated with a larger estimated polyp size (Fig. 4).
In case of pedunculated polyps, a more substantial difference was observed in the optimal colonoscope-polyp distance (ICC, 0.79 [95% CI, 0.71–0.85] at 3 cm vs. ICC, 0.71 [95% CI, 0.61–0.79] at 1 cm, and ICC, 0.69 [95% CI, 0.58–0.77] at 5 cm). After analysis of the polyp size categories in this subgroup, the kappa-values were 0.45 (95% CI, 0.32–0.58) at 3 cm, 0.37 (95% CI, 0.24–0.50) at 1 cm, and 0.40 (95% CI, 0.28–0.52) at 5 cm. Polyp size measured at 3 cm from the tip of the colonoscope tended to yield the most accurate results in terms of polyp size estimation.
In case of pedunculated polyps, a moderate agreement was observed between the estimated and measured polyp sizes (ICC, 0.66 [95% CI, 0.60–0.72]). There was no agreement in case of nonpedunculated polyps (ICC, 0.01 [95% CI, −0.12 to 0.15]). Similar findings were observed while comparing polyp volumes (ICC, 0.69 [95% CI, 0.63−0.74] in pedunculated polyps vs. ICC, 0.03 [95% CI, −0.11 to 0.16] in nonpedunculated polyps).
The intraobserver variability analysis showed a good level of agreement for diameter (ICC, 0.80 [95% CI, 0.75−0.84]) and a moderate agreement for volume (ICC, 0.70 [95% CI, 0.64–0.76]). Focusing on pedunculated polyps alone improved the level of agreement (ICC, 0.87 [95% CI, 0.83−0.90] for diameter and ICC, 0.83 [95% CI, 0.77–0.87] for volume). Comparing the first and second size estimations of each nonpedunculated polyp resulted in a poor level of agreement (ICC, 0.36 [95% CI, 0.18−0.52] for diameter and ICC, 0.41 [95% CI, 0.23–0.56] for volume).
The findings of our study suggest that even in an artificial colon model with an established colon diameter, endoscopic polyp size estimation remains challenging and correlates moderately with the actual polyp size. The distance between the tip of the endoscope and the polyp emerged as a new important factor influencing size estimation. A distance of 3 cm between the tip of the endoscope and the polyp yielded the best results.
The use of artificial polyps of known size provides a reliable gold standard. Previous studies have used pathological reports for comparison.15-19 It is still debatable whether a pathological report is the optimal gold standard, since polyp size is potentially influenced by endoscopic manipulation, polypectomy, or fixation in formalin after the procedure.14,15,28 Furthermore, the use of a colon model ensured that no polyps were excluded from the analysis. In a previous study, the majority (56%, 3,396 out of 6,067) of the polyps had to be excluded, as they were non-intact or were not retrieved for histopathological examination.18 Exclusion of a significant portion of resected polyps might be an important influencing factor, since piecemeal resection is frequently performed for larger polyps. Previous research has shown that a small polyp size is associated with failed polyp retrieval.29 The use of artificial polyps in the colon model excluded this potential bias. The importance of correct endoscopic size estimation is emphasized by Eichenseer et al.17 who showed that the suggested surveillance intervals were often inappropriate when based solely on endoscopically estimated size.
To the best of our knowledge, the present study is the first one to examine the effect of the distance between the tip of the colonoscope and the polyp. A small but substantial difference was observed while comparing the estimation of polyp size at 1, 3, and 5 cm. A smaller distance between the tip of the colonoscope and the polyp was associated with a larger estimated polyp size. The optimal distance for visual polyp size estimation seems to be 3 cm, which consistently yielded the best results in all pedunculated polyps. A comparable phenomenon was observed during endoscopic volume estimation, albeit with a lower consistency. Colonoscopes contain multiple lenses that change positions relative to each other to maintain the sharpness of the images. These positional changes in the lenses may also affect the magnification, influencing the size of the image shown on the monitor. This change in magnification at different viewing distances may partially explain the “optimal” viewing distance. Another complicating factor that influences visual size estimation is the broad field of view (“fish-eye”) of the colonoscope, which causes the center of the image to be magnified, while the peripheral objects appear smaller and warped.
Previous research has shown that polyp morphology significantly influences visual size estimation. Anderson et al.18 reported that nonpedunculated polyps were more frequently overestimated in terms of size when compared with pedunculated polyps (61% vs. 34%). A larger disagreement was observed in our study while comparing polyp size estimation based on polyp morphology, with no agreement in case of nonpedunculated polyps and a moderate to good level of agreement in case of pedunculated polyps. This unexpected difference may be due to the similar sizes of nonpedunculated polyps (30, 35, and 38 mm). All nonpedunculated polyps are over 3 cm in size and the clinical relevance of the ability to discriminate between a 30-mm and a 38-mm polyp is unclear. Therefore, our data on nonpedunculated polyps should be interpreted with caution. Further research should involve a wider range of sizes of nonpedunculated polyps for better assessment of the influence of polyp morphology on size estimation.
A limitation of the present study is the use of a colon model with a fixed inner diameter of 65 mm, which cannot reflect the true dynamics during colonoscopy. Moreover, it does not reflect the changes in size in different colonic locations and does not consider difficulties such as colonic folds or polyps within the flexures. Since the inner diameter model was known to the participants, we excluded locations as a factor influencing size estimation. We decided to use images in our questionnaire instead of videos, which enabled us to use exact distances between the colonoscope and the polyp. In addition, the participating endoscopists were not aware of the variations in distance from the tip of the colonoscope to the polyp among different images. Awareness of this information might have led to the use of secondary clues such as subtle changes in the wall of the colon model to estimate the polyp size instead of making a new estimation each time a new image was presented. These measures were employed to gain optimal insight into the distance between the tip of the colonoscope and the polyp as a factor influencing polyp size estimation.
Another limitation is the relatively small number of polyps used in the study. The number of polyps in the study already resulted in a large number of images for classification (eight polyps at three different distances, presented twice: total 48 images). We expected that a higher number of polyps would result in a lower response rate.
Computed tomography (CT) colonography is another technique used to detect and measure colorectal polyps. Data published in 2011 and a review in 2010 showed that CT colonography measurements were comparable to visual endoscopic estimations, while the pathological specimens were slightly smaller.30,31 Although CT colonography is less invasive and carries a lower risk of complications, its most important disadvantage is the lack of therapeutic possibilities if polyps are detected.
Polyp volume is a novel variable introduced in the present study. It has been discussed in CT colonography studies, but not in the setting of endoscopic polyp size estimation.31-33 Hypothetically, correctly measured polyp volume may be more accurate in risk stratification than polyp diameter, as volume estimation corrects for variations in the shape of the polyps. In CT colonography studies, volumetry has been shown to provide a better insight into polyp growth than measurement of diameter alone.32 However, there are no clear cut-off values for polyp volumes associated with malignant potential or the methods of removal. Thus, the value of polyp volumetry in CT colonography or in an endoscopic setting is yet to be established. In the present study, endoscopic polyp volume estimation was inaccurate for all polyps. This might be because endoscopists are not trained to estimate polyp volume, since it is not a routine practice. Another reason might be the use of static images in a two-dimensional plane. Correct measurement of volume requires depth (three-dimensional) assessment. Although real-time endoscopic images are also presented two-dimensionally during a procedure, the endoscopist can obtain an impression of the depth by moving the colonoscope. Thus, video or real-time endoscopic assessments should be considered in future studies.
The polyp size categories were selected based on their clinical relevance (≤5, 6–9, and ≥10 mm). A margin of ≤5 mm is used in the “resect and discard” strategy, while polyp size ≥10 mm is an independent risk factor for recurrent (advanced) adenomas. Two of the artificial polyps had sizes closer (in diameter) to these margins (polyps sized 5 mm and 10 mm), which might have influenced the level of agreement in the kappa test. However, it also reflects routine practice wherein these small numerical differences in size estimation make an important difference in the suggested surveillance intervals or therapeutic strategies.
Many previous studies have attempted to increase the accuracy of visual polyp size estimation by providing several tools as references to gauge the size of the polyp.16,19-23,34-36 The most commonly researched tools are biopsy forceps, linear measuring probes, and graduated tools such as snares or injection needles. Moreover, training programs and digital measurement systems are also being explored. Although some studies have shown promising results with a clear improvement in the accuracy of visual estimation, others have failed to show this benefit.16,20,21,23,35,36 Furthermore, the studies and outcomes have been heterogeneous and the use of additional tools during colonoscopy has certain disadvantages. The most significant disadvantages are the additional cost and increased procedural time. The use of these tools is not indicated in the current guidelines or advised as a common practice. Hence, we decided not to include them in this study.37
In conclusion, our study confirmed that visual polyp size estimation using an endoscope remains a challenge. Our study demonstrates that the distance between the tip of the colonoscope and the polyp is an important factor influencing polyp size estimation. Polyp size estimation seems optimal at a distance of 3 cm and endoscopists should be aware that a smaller distance between the colonoscope and the polyp results in a larger estimated polyp size. These findings needs further investigation, but may influence endoscopy practice and endoscopic training in the future. With the emergence of artificial intelligence for polyp detection, computer-guided size estimation might be the next tool available to endoscopists. However, until this technique is adapted to clinical practice, a better understanding of endoscopic polyp size estimation is critical.
Notes
Author Contributions
Conceptualization: KRB, JAS, PBM; Data curation: JAS; Formal analysis: KRB, MBK; Investigation: KRB, JAS, SJ, RQ, PBM; Methodology: JAS, MBK, PBM; Project administration: KRB; Supervision: PBM; Validation: MBK, SJ, RQ, PBM; Visualization: KRB; Writing–original draft: KRB, PBM; Writing–review & editing: all authors.
REFERENCES
1. Winawer SJ, Zauber AG, Ho MN, et al. Prevention of colorectal cancer by colonoscopic polypectomy. The National Polyp Study Workgroup. N Engl J Med. 1993; 329:1977–1981.
2. Zauber AG, Winawer SJ, O’Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012; 366:687–696.
3. de Jonge V, Sint Nicolaas J, van Leerdam ME, et al. Systematic literature review and pooled analyses of risk factors for finding adenomas at surveillance colonoscopy. Endoscopy. 2011; 43:560–572.
4. Lieberman DA, Rex DK, Winawer SJ, et al. Guidelines for colonoscopy surveillance after screening and polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012; 143:844–857.
5. van Heijningen EM, Lansdorp-Vogelaar I, Kuipers EJ, et al. Features of adenoma and colonoscopy associated with recurrent colorectal neoplasia based on a large community-based study. Gastroenterology. 2013; 144:1410–1418.
6. Hassan C, Quintero E, Dumonceau JM, et al. Post-polypectomy colonoscopy surveillance: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2013; 45:842–851.
7. Hassan C, Antonelli G, Dumonceau JM, et al. Post-polypectomy colonoscopy surveillance: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - update 2020. Endoscopy. 2020; 52:687–700.
8. Gupta S, Lieberman D, Anderson JC, et al. Recommendations for follow-up after colonoscopy and polypectomy: a consensus update by the US multi-society task force on colorectal cancer. Am J Gastroenterol. 2020; 115:415–434.
9. Wieszczy P, Kaminski MF, Franczyk R, et al. Colorectal cancer incidence and mortality after removal of adenomas during screening colonoscopies. Gastroenterology. 2020; 158:875–883.e5.
10. Ponugoti PL, Cummings OW, Rex DK. Risk of cancer in small and diminutive colorectal polyps. Dig Liver Dis. 2017; 49:34–37.
11. Yoshida N, Naito Y, Siah KT, et al. High incidence of metachronous advanced adenoma and cancer after endoscopic resection of colon polyps ≥20 mm in size. Dig Endosc. 2016; 28:194–202.
12. Rex DK. Real-time endoscopic pathology assessment of colorectal polyps. Curr Gastroenterol Rep. 2013; 15:354.
13. Kessler WR, Imperiale TF, Klein RW, et al. A quantitative assessment of the risks and cost savings of forgoing histologic examination of diminutive polyps. Endoscopy. 2011; 43:683–691.
14. Schoen RE, Gerber LD, Margulies C. The pathologic measurement of polyp size is preferable to the endoscopic estimate. Gastrointest Endosc. 1997; 46:492–496.
15. Moug SJ, Vernall N, Saldanha J, et al. Endoscopists’ estimation of size should not determine surveillance of colonic polyps. Colorectal Dis. 2010; 12:646–650.
16. Rubio CA, Höög CM, Broström O, et al. Assessing the size of polyp phantoms in tandem colonoscopies. Anticancer Res. 2009; 29:1539–1545.
17. Eichenseer PJ, Dhanekula R, Jakate S, et al. Endoscopic mis-sizing of polyps changes colorectal cancer surveillance recommendations. Dis Colon Rectum. 2013; 56:315–321.
18. Anderson BW, Smyrk TC, Anderson KS, et al. Endoscopic overestimation of colorectal polyp size. Gastrointest Endosc. 2016; 83:201–208.
19. Watanabe T, Kume K, Yoshikawa I, et al. Usefulness of a novel calibrated hood to determine indications for colon polypectomy: visual estimation of polyp size is not accurate. Int J Colorectal Dis. 2015; 30:933–938.
20. Gopalswamy N, Shenoy VN, Choudhry U, et al. Is in vivo measurement of size of polyps during colonoscopy accurate? Gastrointest Endosc. 1997; 46:497–502.
21. Margulies C, Krevsky B, Catalano MF. How accurate are endoscopic estimates of size? Gastrointest Endosc. 1994; 40:174–177.
22. Kaz AM, Anwar A, O’Neill DR, et al. Use of a novel polyp “ruler snare” improves estimation of colon polyp size. Gastrointest Endosc. 2016; 83:812–816.
23. Kim JH, Park SJ, Lee JH, et al. Is forceps more useful than visualization for measurement of colon polyp size? World J Gastroenterol. 2016; 22:3220–3226.
24. Plumb AA, Nickerson C, Wooldrage K, et al. Terminal digit preference biases polyp size measurements at endoscopy, computed tomographic colonography, and histopathology. Endoscopy. 2016; 48:899–908.
25. Sakata S, Klein K, Stevenson ARL, et al. Measurement bias of polyp size at colonoscopy. Dis Colon Rectum. 2017; 60:987–991.
26. Elwir S, Shaukat A, Shaw M, et al. Variability in, and factors associated with, sizing of polyps by endoscopists at a large community practice. Endosc Int Open. 2017; 5:E742–E745.
27. Koo TK, Li MY. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 2016; 15:155–163.
28. Morales TG, Sampliner RE, Garewal HS, et al. The difference in colon polyp size before and after removal. Gastrointest Endosc. 1996; 43:25–28.
29. Komeda Y, Suzuki N, Sarah M, et al. Factors associated with failed polyp retrieval at screening colonoscopy. Gastrointest Endosc. 2013; 77:395–400.
30. Barancin C, Pickhardt PJ, Kim DH, et al. Prospective blinded comparison of polyp size on computed tomography colonography and endoscopic colonoscopy. Clin Gastroenterol Hepatol. 2011; 9:443–445.
31. Summers RM. Polyp size measurement at CT colonography: what do we know and what do we need to know? Radiology. 2010; 255:707–720.
32. Pickhardt PJ, Pooler BD, Kim DH, et al. The natural history of colorectal polyps: overview of predictive static and dynamic features. Gastroenterol Clin North Am. 2018; 47:515–536.
33. Johnson CD, Chen MH, Toledano AY, et al. Accuracy of CT colonography for detection of large adenomas and cancers. N Engl J Med. 2008; 359:1207–1217.
34. Leng Q, Jin HY. Measurement system that improves the accuracy of polyp size determined at colonoscopy. World J Gastroenterol. 2015; 21:2178–2182.
35. Hyun YS, Han DS, Bae JH, et al. Graduated injection needles and snares for polypectomy are useful for measuring colorectal polyp size. Dig Liver Dis. 2011; 43:391–394.
36. Jin HY, Leng Q. Use of disposable graduated biopsy forceps improves accuracy of polyp size measurements during endoscopy. World J Gastroenterol. 2015; 21:623–628.
37. Ferlitsch M, Moss A, Hassan C, et al. Colorectal polypectomy and endoscopic mucosal resection (EMR): European Society of Gastrointestinal Endoscopy (ESGE) clinical guideline. Endoscopy. 2017; 49:270–297.
Fig. 1.
The colon model with a colonoscope inside it, showing the image of an artificial polyp on the monitor. The inner diameter of the colon model is 65 mm.
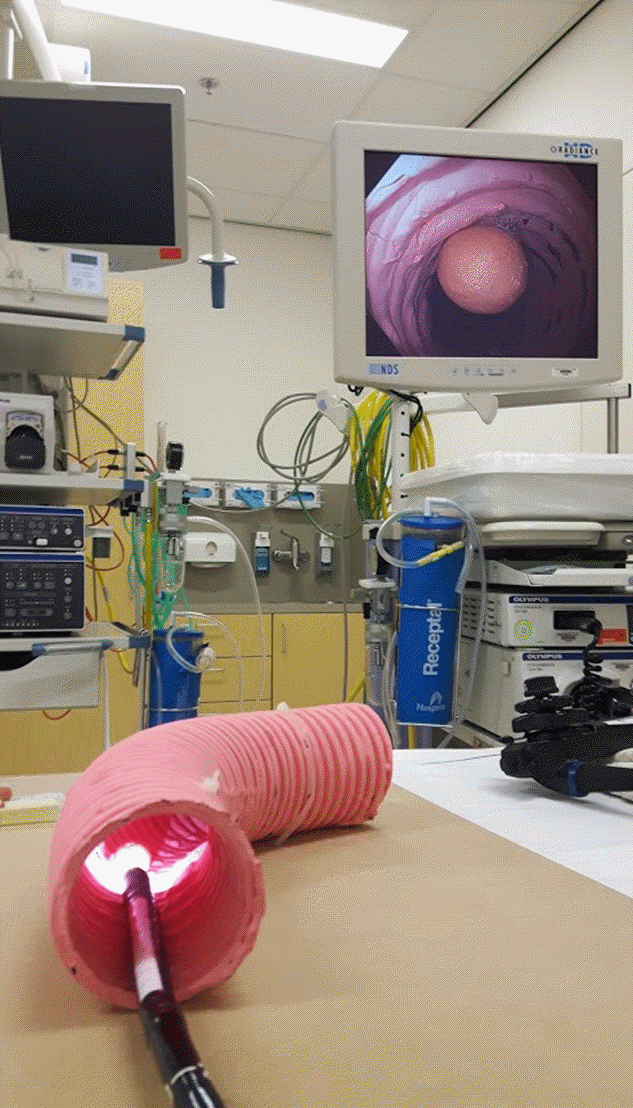
Fig. 2.
The pedunculated and nonpedunculated polyps used in the study, with the photograph obtained at a distance of 3 cm between the tip of the colonoscope and the polyp. A pedunculated polyp sized (A) 5 mm, (B) 8 mm, (C) 10 mm, (D) 15 mm, and (E) 20 mm; a nonpedunculated polyp sized (F) 30 mm, (G) 35 mm, and (H) 38 mm.
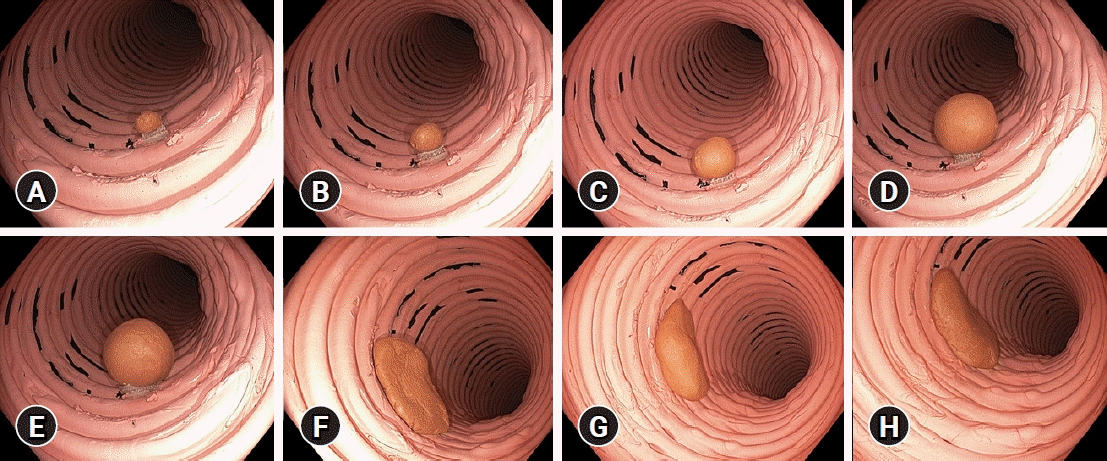
Fig. 3.
Comparison of the endoscopically estimated size and measured size for all pedunculated and nonpedunculated polyps. (A) Polyp size in diameter (mm). (B) Polyp size in volume (mm3).
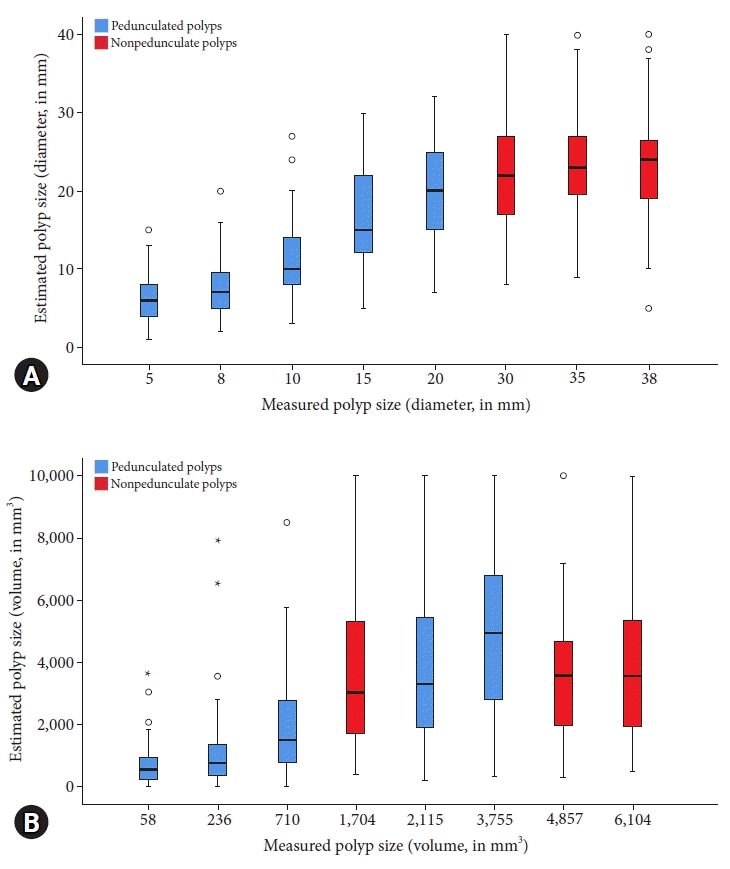
Fig. 4.
Comparison of the endoscopically estimated size and measured size for all pedunculated and nonpedunculated polyps. The endoscopic size estimations were further divided according to the distance between the tip of the colonoscope and the polyp (1, 3, or 5 cm). A clear pattern was observed, suggesting that a smaller distance between the tip of the colonoscope and the polyp (1 cm) resulted in a larger estimated polyp size. (A) Polyp size in diameter (mm). (B) Polyp size in volume (mm3).
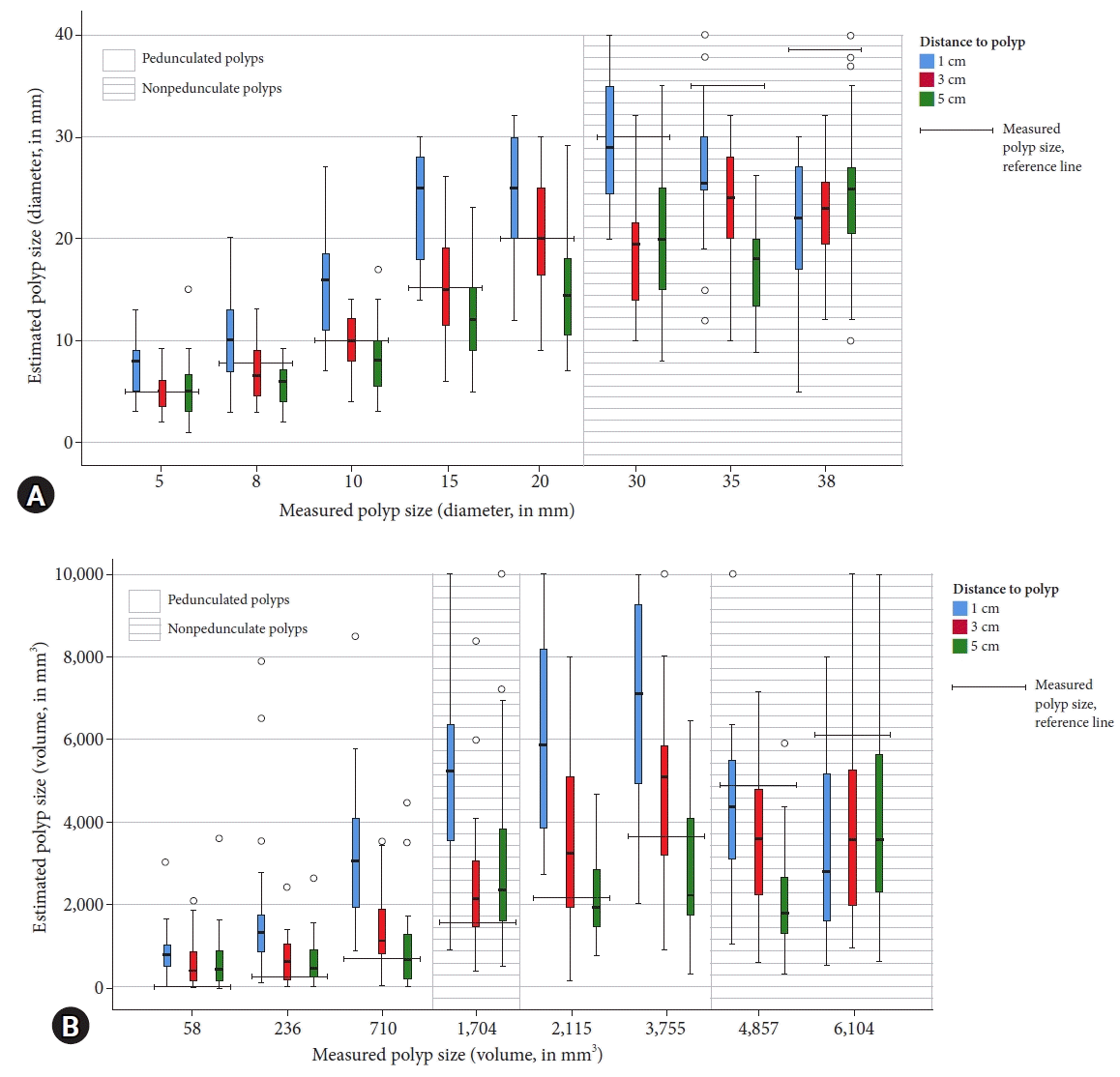
Table 1.
Strength of agreement, according to Koo and Li27
| Kappa and interclass correlation value | Strength of agreement |
|---|---|
| <0 | No correlation |
| 0–0.50 | Poor |
| 0.50–0.75 | Moderate |
| 0.75–0.90 | Good |
| 0.90–1.00 | Excellent |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download