This article has been
cited by other articles in ScienceCentral.
Abstract
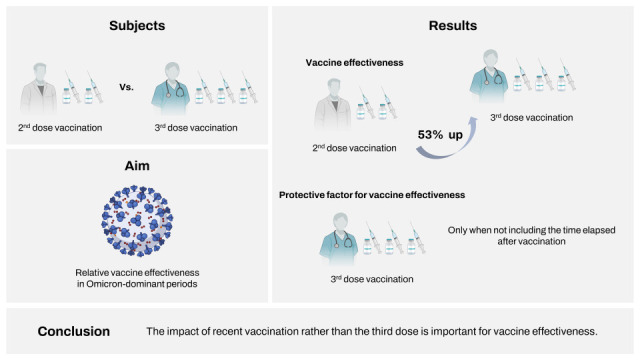
The omicron variant of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is known to have high infectivity and is more likely to evade vaccine immunity. However, booster vaccination is expected to strengthen cross-reactive immunity, thereby increasing the vaccine effectiveness (VE). This study aimed to evaluate the relative VE of the 3-dose (booster) vaccination compared with the 2-dose primary series vaccination in healthcare workers during omicron variant-dominant periods. During the omicron-dominant period from February 1, 2022 to February 28, 2022, a 1:1 matched case-control study was conducted. Healthcare workers with positive SARS-CoV-2 test results were classified as positive cases, whereas those with negative results served as controls. Compared with the 2-dose primary series vaccination, booster vaccination with mRNA vaccine showed moderate VE (53.1%). However, in multivariate analysis including the time elapsed after vaccination, the significant VE disappeared, reflecting the impact of recent vaccination rather than the third dose itself.
Keywords: SARS-CoV-2, COVID-19, Healthcare Worker, Vaccine Effectiveness
Healthcare workers are at an increased risk for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection due to frequent close contact with highly infectious patients and exposure to those with undiagnosed or subclinical infection. Vaccination is considered the most effective measure to prevent disease transmission and control the coronavirus disease 2019 (COVID-19) pandemic.
1 By March 13, 2022, the domestic COVID-19 vaccination rate in Korea was estimated to be 87.5% for the first dose, 86.6% for the second dose, and 62.6% for the third dose.
2 At our hospital, the COVID-19 vaccination rate of the healthcare workers was 98.6% for the first dose, 98.2% for the second dose, and 89.0% for the third dose. COVID-19 vaccine boosters would further enhance or restore the protective effect that decreased over time after the primary series vaccination.
3 Due to concerns about increased transmissibility and vaccine escape of the SARS-CoV-2 omicron variant,
4 we aimed to evaluate the relative effectiveness of booster dose (3-dose) vaccination compared with the 2-dose primary series vaccination in healthcare workers during omicron variant-dominant periods.
We conducted a 1:1 matched case-control study to assess the effectiveness of the booster dose of COVID-19 vaccine among healthcare workers at Korea University Guro Hospital (KUGH) from February 1, 2022 to February 28, 2022. The study inclusion criteria were as follows: 1) no evidence of previous laboratory-confirmed SARS-CoV-2 infection; 2) availability of immunization information; 3) receipt of 2- or 3-dose COVID-19 vaccination series at least 14 days prior to study enrollment; and 4) completion of laboratory tests for COVID-19 within 7 days after symptom onset or close contact with SARS-CoV-2-infected patients. If tests were repeated, only those on the laboratory-diagnosed date were included; otherwise, only the first test date of each uninfected individual was included in the study. However, we excluded subjects who underwent SARS-CoV-2 real-time reverse transcriptase-polymerase chain reaction (rRT-PCR) tests prior to returning to work after release from quarantine and those who underwent periodic tests for simple screening purposes. SARS-CoV-2 infection was confirmed by rRT-PCR (Allplex™ 2019-nCoV Assay; Seegene, Seoul, Korea). Healthcare workers with positive rRT-PCR results were classified as positive cases, whereas those with negative test results served as controls. Controls were matched to the cases on the age, sex, and date of the laboratory tests. All statistical analyses were performed using SPSS version 26 software (IBM Corp., Armonk, NY, USA). Continuous variables were analyzed using an independent t-test and categorical variables were analyzed using the χ
2 test or Fisher’s exact test. Statistical significance was set at
P < 0.05. Logistic regression was used to estimate the odds ratio for laboratory-confirmed COVID-19 in 3-dose vaccinated vs. 2-dose vaccinated participants. Vaccine effectiveness (VE) was defined as [100 × (1 − Odds Ratio for COVID-19 in 3-Dose Vaccinated vs. 2-Dose Vaccinated Individuals)]. Logistic regression models were adjusted for age, comorbidities, occupation category, vaccine type, and household exposure with COVID-19. The intervals from the third-dose vaccination to the time of study enrollment is inevitably shorter than those of the second-dose vaccination, so the time that elapsed after the final vaccination is highly likely to offset the effect of the third vaccination. Therefore, the time elapsed after the final vaccination was excluded from the multivariate analysis for VE; the results including elapsed intervals are presented in
Supplementary Table 1.
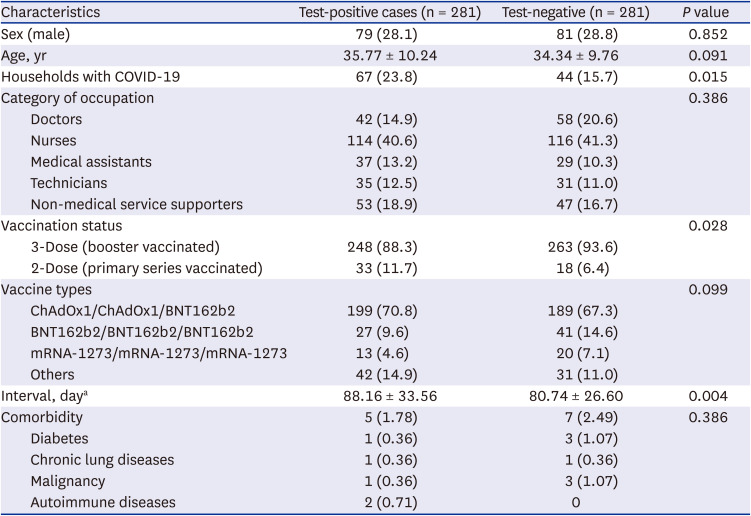
In total, 281 patients were identified during the study period. The mean ages of the test-positive cases and test-negative controls were 35.77 ± 10.24 years and 34.34 ± 9.76 years, respectively (
P = 0.091) (
Table 1). Test-positive cases were more likely to have SARS-CoV-2-infected household members from whom they might have developed COVID-19 (23.8% vs. 15.7%,
P = 0.015). The booster vaccination rate was 88.3% (n = 248) in the test-positive cases and 93.6% (n = 263) in the test-negative controls (
P = 0.028). The mean intervals from the time of final dose of vaccination (second or third dose) to testing were 88.16 ± 33.56 days in test-positive cases and 80.74 ± 26.60 days in test-negative controls, respectively (
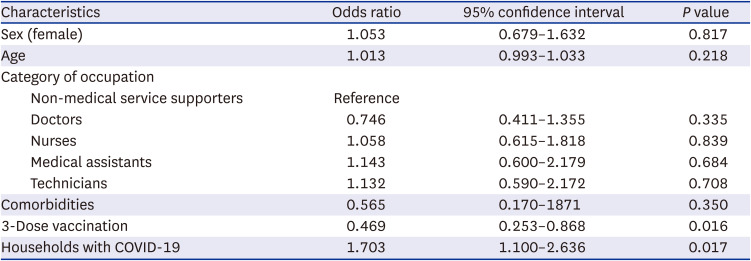
P = 0.004). There were no significant differences in comorbidities or vaccine types between the test-positive cases and test-negative controls. In the multivariate analysis, booster vaccination showed an additional effectiveness of 53.1% in preventing SARS-CoV-2 infection among healthcare workers during omicron dominant periods (
Table 2). As expected, multivariate analysis, including the time elapsed after vaccination was performed, revealed that the 3-dose vaccination series did not show significant VE against COVID-19 compared to 2-dose vaccination, while the elapsed interval after vaccination appeared to be associated with SARS-CoV-2 infection (
P = 0.064) (
Supplementary Table 1). This reflected the waning VE after vaccination, suggesting the benefit of the booster (third) dose. In the subgroup analysis of heterologous booster vaccination (ChAdOx1/ChAdOx1/BNT162b2), the third-dose vaccination showed similar VE (48.3%) (
Supplementary Table 2). The risk of contracting SARS-CoV-2 increased by 1.703-fold when there was a household member infected with COVID-19. Among the test-positive cases, 83.3% (n = 234) had symptoms such as fever, sore throat, and cough. None of the cases required hospitalization, and all recovered without complications.
According to a previous study, neutralizing antibody immunity to wild-type SARS-CoV-2 and Delta variant was maintained until 5 months after 2-dose primary series vaccination with Pfizer/BioNTech messenger ribonucleic acid (mRNA) vaccine (BNT162b2), but cross-neutralizing antibody to the omicron variant was not detectable.
5 In comparison, after the booster vaccination (third dose), sufficient neutralizing antibodies were induced against the omicron variant (neutralizing antibody titer at 50% inhibition, 100). Similarly, after booster vaccination with mRNA-1273, cross-neutralizing antibodies against the omicron variant significantly increased.
6 If booster vaccination is performed after memory B-cells have sufficiently matured with 2-dose primary series vaccination, anti-SARS-CoV-2 antibodies with high affinity and wide diversity might be produced, forming cross-neutralizing antibody immunity against omicron variants. In a study evaluating real-world effectiveness in England, significant VE was maintained against Delta variants up to 25 weeks after 2-dose primary series vaccination but not for omicron variants.
7 However, after the booster mRNA vaccination, VE against omicron increased to 62.4–73.9% at 2–4 weeks, decreasing to 39.6–45.7% at 10 or more weeks.
7 In other studies, 2-dose primary series vaccination did not provide significant protection against omicron variants; however, after the booster dose, 37–66% of omicron variant infections were prevented.
8 The VE estimates in this study are generally consistent with those previously reported.
78
This study has several limitations. First, it was a single-center retrospective observational study, so its results should be interpreted with caution. Second, although this study was conducted during the omicron variant-dominant period, viral sequencing data were not available. Thus, other variant infection cases might have been included. Nevertheless, as of the beginning of February 2022, the omicron variant has accounted for more than 90% of the SARS-CoV-2 cases in South Korea.
9 Finally, there might be some unadjusted confounding factors including healthy vaccinee bias (general health status and behavior). However, we included healthcare workers who received at least 2 doses of the COVID-19 vaccine and provided free SARS-CoV-2 rRT-PCR tests for all healthcare workers. In KUGH during the entire COVID-19 pandemic period, SARS-CoV-2 rRT-PCR testing was performed for all individuals who had fever/respiratory symptoms or close contact with laboratory-confirmed patients regardless of vaccination status.
In summary, booster mRNA vaccination may provide moderate protection (53.1%) against the omicron variant of SARS-CoV-2 for 3 months after immunization. However, in multivariate analysis including the time elapsed after vaccination, the significant VE disappeared, reflecting the impact of recent vaccination rather than the third dose. While this study was not designed to evaluate the relative VE after the concurrent completion of the second and third dose vaccination, the waning of COVID-19 VE is a critical issue considering that COVID-19 pandemic is ongoing with everchanging viral antigenicity.
Ethics statement
The study was approved by the Institutional Review Board of the Korea University Guro Hospital (2022GR0282). Written informed consent was waived.
Table 1
Characteristics of the study cases and controls
|
Characteristics |
Test-positive cases (n = 281) |
Test-negative (n = 281) |
P value |
|
Sex (male) |
79 (28.1) |
81 (28.8) |
0.852 |
|
Age, yr |
35.77 ± 10.24 |
34.34 ± 9.76 |
0.091 |
|
Households with COVID-19 |
67 (23.8) |
44 (15.7) |
0.015 |
|
Category of occupation |
|
|
0.386 |
|
Doctors |
42 (14.9) |
58 (20.6) |
|
Nurses |
114 (40.6) |
116 (41.3) |
|
Medical assistants |
37 (13.2) |
29 (10.3) |
|
Technicians |
35 (12.5) |
31 (11.0) |
|
Non-medical service supporters |
53 (18.9) |
47 (16.7) |
|
Vaccination status |
|
|
0.028 |
|
3-Dose (booster vaccinated) |
248 (88.3) |
263 (93.6) |
|
2-Dose (primary series vaccinated) |
33 (11.7) |
18 (6.4) |
|
Vaccine types |
|
|
0.099 |
|
ChAdOx1/ChAdOx1/BNT162b2 |
199 (70.8) |
189 (67.3) |
|
BNT162b2/BNT162b2/BNT162b2 |
27 (9.6) |
41 (14.6) |
|
mRNA-1273/mRNA-1273/mRNA-1273 |
13 (4.6) |
20 (7.1) |
|
Others |
42 (14.9) |
31 (11.0) |
|
Interval, daya
|
88.16 ± 33.56 |
80.74 ± 26.60 |
0.004 |
|
Comorbidity |
5 (1.78) |
7 (2.49) |
0.386 |
|
Diabetes |
1 (0.36) |
3 (1.07) |
|
Chronic lung diseases |
1 (0.36) |
1 (0.36) |
|
Malignancy |
1 (0.36) |
3 (1.07) |
|
Autoimmune diseases |
2 (0.71) |
0 |
Table 2
Multivariate analysis for the risk factors of COVID-19
|
Characteristics |
Odds ratio |
95% confidence interval |
P value |
|
Sex (female) |
1.053 |
0.679–1.632 |
0.817 |
|
Age |
1.013 |
0.993–1.033 |
0.218 |
|
Category of occupation |
|
|
|
|
Non-medical service supporters |
Reference |
|
|
|
Doctors |
0.746 |
0.411–1.355 |
0.335 |
|
Nurses |
1.058 |
0.615–1.818 |
0.839 |
|
Medical assistants |
1.143 |
0.600–2.179 |
0.684 |
|
Technicians |
1.132 |
0.590–2.172 |
0.708 |
|
Comorbidities |
0.565 |
0.170–1871 |
0.350 |
|
3-Dose vaccination |
0.469 |
0.253–0.868 |
0.016 |
|
Households with COVID-19 |
1.703 |
1.100–2.636 |
0.017 |






 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download