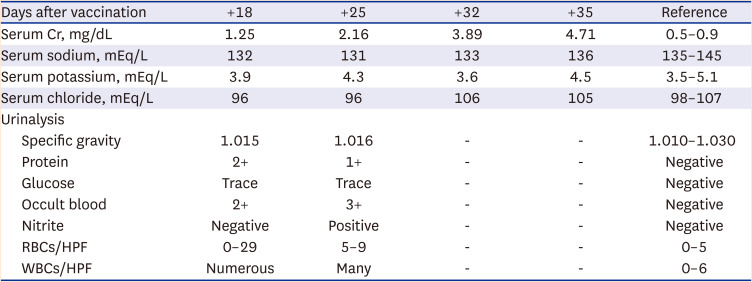
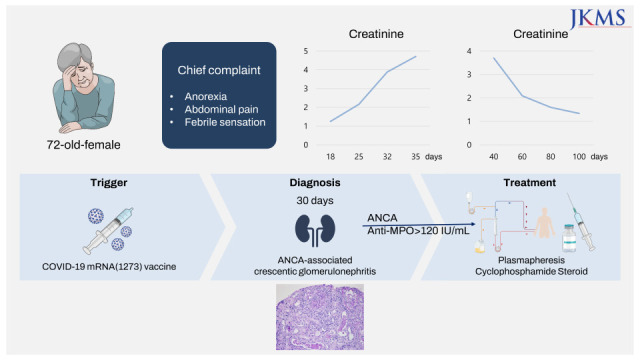
A 72-year-old woman with no specific past medical history presented with tinnitus, nasal congestion, and bloody rhinorrhea. Her constitutional symptoms developed after she received the first dose of the ChAdOx1 nCoV-19 (Oxford AstraZeneca) COVID-19 vaccine on 12 July 2021, and her symptoms were aggravated after she received the second dose of the ChAdOx1 nCoV-19 vaccine on 16 August. She underwent tympanostomy tube insertion and received antibiotic therapy for 2 weeks. However, after she received her third (booster) dose, the mRNA1273 (Moderna) vaccine, on 28 December 2021, she experienced anorexia, abdominal pain, and febrile sensation. Therefore, she was admitted to the gastroenterology department of our hospital. Her endoscopic findings revealed erosive gastritis, and she was treated with intravenous hydration because of her constitutional symptoms of poor oral intake and abdominal pain. She had normal body temperature (36.7°C), pulse rate (78/min), and respiratory rate (18/min) but high blood pressure (152/83 mmHg). A routine laboratory test performed 18 days after vaccination with the mRNA1273 (Moderna) vaccine revealed a serum creatinine (Cr) level of 1.25 mg/dL, and microscopic findings of urinalysis showed microscopic hematuria, occult blood (2+), 10–29 red blood cells (RBCs) per high-power field, and proteinuria (2+). The last previous laboratory test was performed in 2018 and was notable for a serum Cr level of 0.81 mg/dL. On the 25th day after vaccination, her Cr level elevated to 2.16 mg/dL. The spot urine protein to Cr ratio was 0.85 mg/g·Cr, and the spot urine albumin to Cr ratio was 0.19 mg/g·Cr. Despite intravenous hydration, the creatine level remained elevated, and with this finding along with persistent hematuria with proteinuria, we were prompted to perform serological testing. Serological tests revealed positive ANCA titers (> 120 IU/mL) and antibodies against MPO. Anti-nuclear antibody (ANA), anti-phospholipase A2 receptor (PLA2R) antibody, and anti-glomerular basement membrane antibody were all negative. The rheumatoid factor titer was 221 IU/mL and the C3 and C4 levels were 121 and 18 mg/dL, while immunoglobulins levels were all within normal limits (immunoglobulin [Ig] A 197 mg/dL, IgG 1,121 mg/dL, and IgM 96 mg/dL). Kidney imaging showed a left kidney cyst; otherwise, no abnormal findings were present. A kidney biopsy (
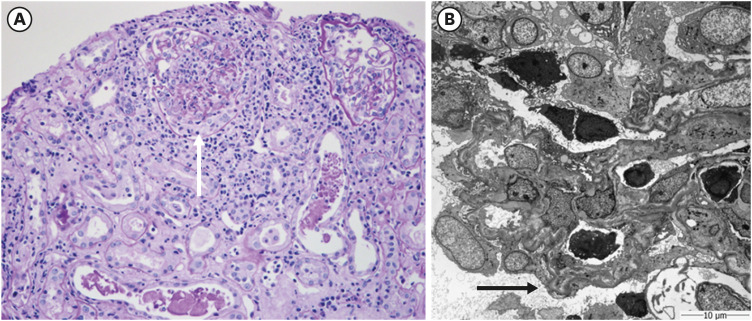
Fig. 1) showed fibrocellular crescents (42.9%) and global sclerosis (14.3%) in glomeruli, with focal moderate tubular atrophy found 30 days after vaccination with the mRNA1273 (Moderna) vaccine. Immune complex-mediated deposits were not seen by electron microscopy. ANCA-associated pauci-immune crescentic glomerulonephritis was diagnosed based on the serological test and kidney biopsy findings. After kidney biopsy, intravenous pulse steroid therapy was initiated; 500 mg of methylprednisolone sodium succinate was injected intravenously over 1 hour once daily for three days. Two weeks after admission, her serum Cr level was elevated from 1.25 mg/dL (glomerular filtration rate [GFR], 42.1 mL/min/1.73 m
2) to 4.71 mg/dL (GFR, 9.1 mL/min/1.73 m
2) (
Table 1). The plasmapheresis is considered when a patient’s serum Cr level is above 5.7 mg/dL as the guideline,
5 however, it was initiated considering the patient’s rapid deterioration of renal function and biopsy findings showing crescents in nearly 50% of glomeruli. Intravenous cyclophosphamide 2.5 mg/kg was administered because of severe glomerulonephritis, and a reduced dose was given based on the patient’s age (> 70 years) and GFR less than 30 mL/min/1.73 m
2. We tapered oral steroids based on the PEXIVIAS trial (oral prednisolone 60 mg per day on 1st week, 30 mg per day on 2nd, 25 mg per day on 3rd and 4th, 20 mg per day on 5th and 6th, 15 mg per day on 7th and 8th, 12.5 mg per day on 9th and 10th, 10 mg per day on 11th and 12th, 7.5 mg per day on 13th and 14th, 6 mg per day on 15th and 16th, and 5 mg per day afterwards).
5 After the third dose of intravenous cyclophosphamide, her renal function improved, and induction therapy with oral cyclophosphamide was administered. Her Cr level improved to 1.34 mg/dL after two months of diagnosis, and her constitutional symptoms improved.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download