Abstract
Osteoarthritis (OA) is the most common form of arthritis and is a growing public health concern in the aging society. In rapidly aging societies such as in Korea, the increasing prevalence of OA may present serious new health issues. There is no treatment for OA that can either prevent or slow the progression of joint damage. For the development of effective therapeutics, precise understating of its pathogenesis is important. In this review, the current evidence of etiopathogenesis of OA is discussed. First, while epidemiologic study of OA are still dominated by reports from Western countries, findings from Korean epidemiologic studies are highlighted. Then, recent progresses in genetics, especially in the field of genome wide association study and mendelian randomization studies, are reviewed with focus on Asian population. Lastly, sex difference in pain etopathogenesis is reviewed. Studies of OA pathogenesis including epidemiology, genetics, animal model and pain signaling will aid in progress towards treatment of OA.
Osteoarthritis is the most common form of arthritis and is a growing public health concern in the aging society. OA is the leading cause of lower extremity disability in older adults, and hip and knee OA have accounted for 1.12 percent of all years lived with disability (YLD), an overall measure of disease burden. In Global Burden of Disease (GBD) studies, OA is consistently ranked among the leading contributors to global YLDs. In rapidly aging societies such as in Korea, the increasing prevalence of OA may present serious new health issues. There is no treatment for OA that can either prevent or slow the progression of joint damage. For the development of effective therapeutics, precise understating of its pathogenesis is important. In this review, the current evidence of etiopathogenesis of OA is discussed.
Prevalence of OA differs according to involved joints, population studied and definition used. Studies from Western countries report that about 80% of people >75 years old have radiographic osteoarthritis and GBD project report that age-adjusted prevalence, incidence and YLD all have been increasing 8%~10% since 1990. In addition, a US study reported that health care cost due to OA has been increasing 5.7% every year since 1996, amounting to 8 billion dollars in 2016, and OA become the 8th high-cost illness among 154 reference diseases [1]. A recent Korean report using data from the Korea National Health and Nutritional Examination Survey from 2010 to 2013 showed the prevalence of radiographic OA of 35.1% (male 24.4%, female 44.3%) in people >50 years old, with the highest prevalence of 78.7% among female >80 years old. Previous studies have reported various risk factors associated with knee OA such as older age, female sex, hypertension, raised glucose, obesity, history of knee injury, varus/valgus malalignment, quadriceps muscle strength, and physical workload [2]. Since most of these studies for risk factors of knee OA have been performed in persons of European origin, the results cannot be extrapolated to Asian populations. There have only been a few longitudinal studies of risk factors for knee OA among Asians peoples with age, sex and body mass index (BMI) as significant risk factors for incident OA [3]. In a Korean study using the standardized radiographic protocol of community residents in Chuncheon, the presence of hypertension, having a manual occupation and a lower level of education were significantly associated with the presence of radiographic knee OA [4]. Three-year follow-up study of the same population showed that incident OA was observed in 10.2% and progression of OA in 13.6% subjects. Only female sex was significantly associated with the progression of radiographic knee OA [5]. It is of note that the progression rate of knee OA among Koreans as well as Japanese females was higher compared to Western population (7.36% and 6.3% per year among Korean and Japanese females, respectively vs. 3.9% and 2.8% among subjects from the Framingham OA Study and Chingford Women’s Study, respectively), suggesting that lifestyle factors, such as sitting with legs crossed, sitting with knees and feet together on the floor, or genetic factors might play a role [5].
Difference in prevalence of OA according to ethnicity is more remarkable in the hip OA prevalence. A Korean study reported that not only the prevalence of hip OA in people >70 years old assessed with intravenous pyelograms was lower compared to Caucasians, (1.0% overall, 1.4% for male and 0.7% for female), but the rate of total hip replacement (THR) was significantly lower than that of total knee replacement (TKR) in Korean population (THR vs. TKR 1:15.9). This is in contrast to the US, where the rate of TKR is about 20% higher than that of THR or to Sweden, where the rate of TKR is lower than that of THR [6], and suggests that severe hip OA is much less common in Korea as compared to Western countries, and as compared to severe knee OA. A previous report on hip OA estimated by intravenous pyelogram (IVP) among Chinese male aged 60~75 years stated that the prevalence of hip OA among a Chinese population was much lower to that than among a population in the United Kingdom (5.4% vs. 11.0%), further corroborating the Korean study showing lower prevalence of hip OA [7]. Although the reason behind the discrepancy of the prevalence of hip OA between Asians and Caucasians is not known, differences in the frequency of acetabular dysplasia among ethnic groups, as well as genetic factors have been postulated.
Epidemiologic study of OA are still dominated by reports from Western countries with high prevalences of obesity, however, as obesity increases in other parts of the world, it is likely that OA would also increase. In addition, populations previously adapted to energy scarcity may be particularly susceptible to the development of obesity-related OA from rapid transitions in diet and physical activity [8]. In cross-sectional analyses of an indigenous Mexican population, they were predisposed to accrue greater abdominal adiposity for a given body weight, and were more vulnerable to radiographic and symptomatic knee osteoarthritis at lower levels of body mass index, compared to Americans [9]. Recently, inactivity as well as the level of physical activity has been gaining interest in OA. A recent report showed that in 13.7 million persons with knee OA, a total of 7.5 million quality adjusted life years (QALYs), or 0.55 QALYs per person, were lost due to inactivity or insufficient physical activity [10].
Despite the long-held concept that OA is a degenerative disease stemming from aging and mechanical wear and tear of joints, where environmental factors play a major role in its development, evidence shows that genetic factors are also important factors. Epidemiologic studies have shown that Heberden’s nodes in the distal interphalangeal joints run in a family, especially among female members. Twin studies consistently showed that OA concordance is significantly higher among monozygotic twins than in dizygotic twins [11]. Generally, hand and hip OA show higher genetic predilections compared to knee OA. Recent progresses in technology of molecular biology and genetics enabled the field of OA to enjoy some of the most ground-breaking advances.
1) Identification of risk loci by the use of genomewide association study (GWAS, Figure 1)
The amount of information provided by the hypothesis-free GWAS has accumulated exponentially during the past decade. Not only risk loci, but also potential drug targets have been forwarded. Lower number of OA risk loci identified by GWAS compared to other diseases (rheumatoid arthritis, over 100 risk loci; height, over 3,000 loci; OA, 19 reported up to April 2017), may be related with its stronger risk conferred by environmental factors and aging compared to other diseases [12].
The discovery of significant OA genetic risk loci has since accelerated, with increase in the number of samples included in the analysis and utilization of genotyping and medical data, together with existing OA GWAS cohorts. Recent large studies using a meta-analysis of the UK biobank (UKBB) data together with the Icelandic deCODE genetics or UK arcOGEN datasets exhibits increased power to detect genetic variants with small to modest effects sizes whist reducing the probability of false-negative results [13,14]. Of the novel loci reported by these studies, variants mapping to the LMX1B, COL11A1, and IL11 genes were commonly identified. Joint-specific differences in genetic risk was noted and loci near genes involved in skeletal development and/or associated with rare monogenic bone disease was identified, including the TGFβ pathway genes TGFB1, LTBP1, LTBP3, SMAD3, and ROCR long non-coding RNA (lncRNA) that acts upstream of SOX9 during chondrogenic differentiation. A global collaboration of 18 GWAS cohorts from Europe, Japan, Hong Kong, and the USA, including more than twice the number of OA cases used in previous GWAS studies runs meta-analyses for spine, thumb and finger OA in addition to hip and knee OA, and may enable identification of novel OA pathways more generalizable in the near future (Table 1) [15]. A GWAS from the UK Biobank cohort identified female-specific independent single-nucleotide variant (SNVs), including rs116112221 in the female-only total hip replacement phenotype and is located in a region containing long intergenic non coding RNAs with the closest protein coding gene being FA Complementation Group L (FANCL), the mutations of which are causative for premature ovary insufficiency in humans, a condition associated with increased prevalence of osteoarthritis. In addition, rs10282983, which resides in an intron of C8orf34, was associated with total hip replacement with opposite direction of effects between male and female. This locus has been associated with waist-to-hip ratio and heel bone mineral density, both risk factors for osteoarthritis.
Recently, GWAS has been used to elucidate the inter-relationship between various OA risk factors and genetics. DNA variants associated with height, hip shape, and developmental dysplasia of the hip were explored since subtle differences in joint development and shape may alter joint biomechanics, leading to OA later in life. In the deCODE-UKBB GWAS, FAM101A, and FILIP1 loci associated with increased or COL11A1 and GDF5 associated with decreased height were identified at the genome-wide significance level [13]. A study analyzing hip shape by bone density scans from five different cohorts identified 3 loci overlapping known OA loci near the PTHLH and ASTN2 (for hip OA) and ROCR/SOX9 (for knee OA) [16]. GDF5 SNP rs143384 was found associated with knee OA and developmental dysplasia of the hip in European and Asian population [17,18]. Although GDF5 is not a risk locus for hip OA in Europeans, changes in biomechanics may confer indirect risk to knee joint because GDF rs143384 is also associated with hip intertrochanteric/shaft and trochanter bone area as well as bone area of the lumbar spine. A GWAS of bone size derived from dual-energy X-ray absorptiometry (DXA) scans, identified eight bone area variants associated with hip or knee OA [12].
2) Causal relationships between risk factors and OA idenitified by Mendelian Randomization (MR) studies
MR studies has been employed to investigate causal relationships between risk factors and development of disease independent of confounders by using genetic variations. Malmö Diet and Cancer Study determined the role of cardiometabolic risk factors in hip and knee OA and reported a causal role for lower Low Density Lipoprotein and greater BMI [19]. UK Biobank study evaluated causal factors for hand, hip and knee OA using data from 384,838 individuals, and revealed a strong causal association of genetically determined BMI with knee and hip OA but not hand OA. Increased bone mineral density at the femoral neck and low systolic blood pressure were also causally associated with hip and knee OA [20]. Furthermore, phenome wide health effects (MR-PheWAS) is elucidated with MR. Another UK Biobank study using data from 337,535 individuals based on their genetic calcium score showed OA as a downstream health outcome [21].
3) Sex-specific genetic contributions
Genetic study is used to evaluate sex-specific genetic contribution to OA. A study using the world’s largest twin registry showed that overall heritability of knee OA surgery was 53%, which was higher in female than male [22]. A study of the effects of maternal and paternal factors on hand, hip and knee OA in offspring showed that OA in the mother increased the risk of surgically and clinically defined hip, knee, and hand OA in the offspring, particularly in daughters. On the other hand, having a father with OA was less likely to increase the risk of any OA in daughters or sons, implicating maternal genes and/or maternal-specific factors such as the fetal environment in the heredity of OA [23].
4) Epigenetic studies
An evolutionary aspect of OA in terms of human bipedalism was explored by epigenetical profiling of human and mouse joint chondrocytes [24]. Open chromatin regions specific to distal femur and proximal tibia, reflecting site-specific regulatory elements involved in knee chondrogenesis, exhibited signals of the effects of ancient selection during primate and hominin evolution. These include reduced sequence conservation among primates relative to pleiotropic elements, enrichments for human accelerated regions, and significantly reduced diversity relative to pleiotropic elements and certain genome features within humans but not chimps. A variant rs6060369 of GDF5, a BMP family member essential to knee development across mammals was identified as causative. The proposed model suggests that evolutionary mechanisms such as genetic drift and antagonistic pleiotropy (where positive selection increases a beneficial allele together with linked deleterious alleles) leads to violations to constraint of functional conserved sequences which are tolerated during knee development but can increase risk of OA development [15,25-27]. A study profiling regional patterns of protein turnover and miRNA expression in human hip, knee, and ankle joints evaluated molecular clocks in the form of nonenzymatically deamidated proteins [28]. Ankle cartilage as well as surface zone cartilage, had lower ratios of deamidated proteins indicating higher tissue anabolism and greater repair capacity. A position-dependent gradient (distal high, proximal low) of protein turnover, indicative of a gradient of tissue anabolism reflecting innate tissue repair capacity in human lower limb cartilages that is associated with expression of limb-regenerative microRNAs was revealed. miRNAS signal of blastemal, a mass of cells capable of differentiation and growth into organs or appendages, essential to limb regeneration in certain animal species (miR-121, miR-31, and miR-181c) were enriched in ankle joint, superficial zone and OA cartilage, suggesting that strategies incorporating miRNAs to enhance cartilage anabolism and endogenous repair may be useful.
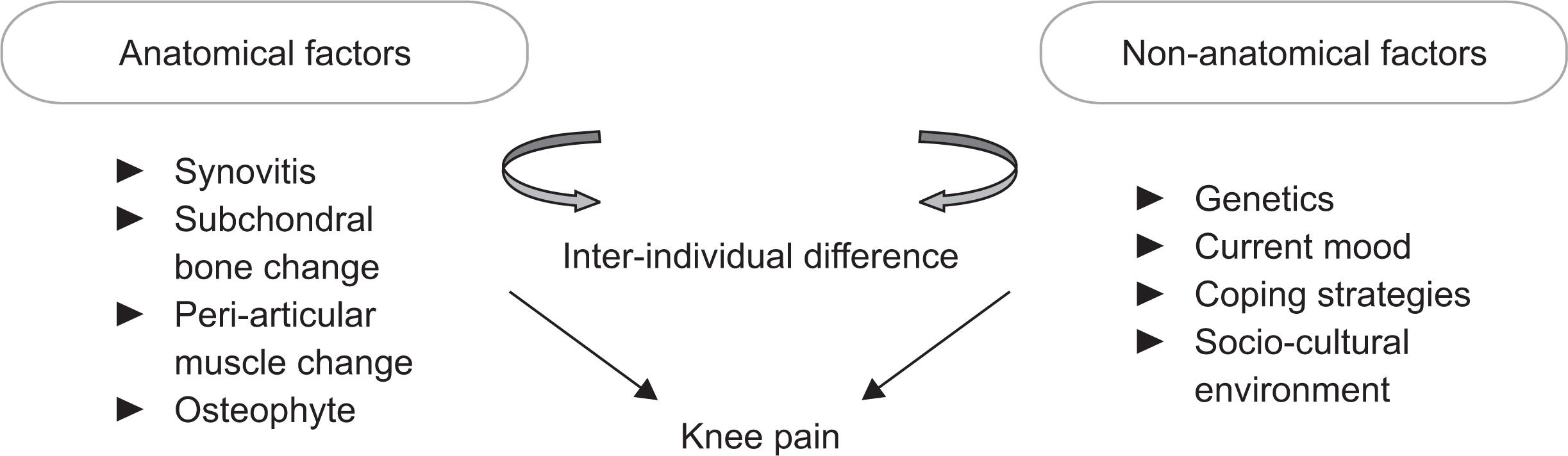
Radiographic changes in OA are poorly correlated with pain and physical function, and the risk factors for radiographic OA are not the same as those for OA pain [29]. As pain is strongly correlated with person-level psychological factors aside from joint damage, studies involving human OA subjects have a high risk for being influenced by confounding effects between individual subjects [30]. Study using a within-person, case–control design minimizes inter-personal difference by using less symptomatic knee in the same person as a control. The severity of radiographic knee OA in persons with knees discordant for the presence of pain or pain severity, is indeed strongly associated with pain in diverse ethnic groups [31,32]. On the other hand, a study of community subjects and the Korean National Health and Nutrition Examination Survey (KNHANES) showed that a significant number of subjects with Kellgren and Lawrence (KL) grade 4 OA, which is characterized by marked loss of articular cartilage and joint damage, did not report pain [33]. Considering that hyaline cartilage, the main focus of research in OA, is an aneural tissue and devoid of nociceptive input, therapeutic decision based on the degree of cartilage loss in knee X-ray would lead to suboptimal result. In addition, treatment options focusing solely on cartilage engineering, should be viewed with caution. Bone marrow lesions (BMLs), which are regions of hyperintense marrow signals, revealed in magnetic resonance imaging (MRI) are strongly associated with knee pain as well as progression of cartilage damage [34]. Expression of nerve growth factor (NGF) in osteochondral channels in the subchondral bone was found to correlate with pain in humans matched for macroscopic cartilage damage score [35]. An animal study showed that reduction of osteoclast formation by knockout of receptor activator of nuclear factor kappa-B ligand (Rankl) in osteocytes inhibited the growth of sensory nerves into subchondral bone, dorsal root ganglion neuron hyperexcitability, and behavioral measures of pain hypersensitivity in OA mice [36]. Netrin-1 secreted by osteoclasts during aberrant subchondral bone remodeling was found to play a role in inducing sensory innervation and OA pain through its receptor DCC (deleted in colorectal cancer). These results suggest that aberrant subchondral bone remodeling is associated with symptomatic knee OA, independent of cartilage lesions.
Tissue damage and inflammation in the joint may induce peripheral and central sensitization, perpetuating chronic OA pain. A study of 2,126 subjects with or at risk for knee OA reported that mechanical temporal summation (an augmented response to repetitive mechanical stimulation and a measure of central sensitization) and pressure pain thresholds (a measure of sensitivity to pain evoked by mechanical stimulation of nociceptors and a reflection of activity-dependent peripheral sensitization) were associated with pain severity but not radiographic OA [37]. Pain occurs intermittently in response to strenuous activities in early OA while it becomes constant and occurs even at rest in late stage. It is speculated that aggravation of pain arises from neuropathy due to damage to the neurons innervating the joint. End-stage OA shows tidemark breaching by vascular channels containing sympathetic and sensory nerves, which corroborate these hypothesis [38].
A GWAS of 171,516 individuals from the UK Biobank cohort identified genetic variants associated with knee pain [39]. Two loci were identified, rs143384 located in GDF5, a gene playing a role in joint development, and rs2808772 located near COL27A1, a member of the fibrillar collagen family. It is of note that GDF5 variants are also associated with increased OA risk. The largest genome-wide association study meta-analysis across 826,690 individuals (177,517 with osteoarthritis) identified high correlation between osteoarthritis and sciatica, fibromyalgia, headaches, and other back pain phenotypes, where the highest correlation is with spine osteoarthritis [40].
Although decades of epidemiologic and clinical studies have demonstrated that woman are more sensitive to pain than men, and OA is much more prevalent in female, research into the underlying mechanism of sex difference in the arthritis induced pain is still at its infancy due to the bias in biomedical research which largely used male subjects and animals. Aside from biomechanical difference such as knee adduction moment according to sex, the influence of sex hormone on OA has been studied to elucidate the mechanism behind the sex difference. However, the result has been conflicting, with a cross-sectional study showing positive association between estradiol levels and radiographic knee OA, while another cohort study showing negative association [41]. A recent study using animal models have demonstrated that spinal microglia cells, macrophage-like immune cells which reside in the central nervous system, proliferate in the spinal cord following nerve injury, but only male mice use microglia as mediators for the persistent pain hypersensitivity [42]. Whether such sex difference in pain modulation plays a role in human OA is an interesting subject for future research. The search for sex-specific loci associated with pain phenotype of OA remains to be investigated.
Notes
REFERENCES
1. Dieleman JL, Cao J, Chapin A, Chen C, Li Z, Liu A, et al. 2020; US health care spending by payer and health condition, 1996-2016. JAMA. 323:863–84. DOI: 10.1001/jama.2020.0734. PMID: 32125402. PMCID: PMC7054840.

2. Bastick AN, Runhaar J, Belo JN, Bierma-Zeinstra SM. 2015; Prognostic factors for progression of clinical osteoarthritis of the knee: a systematic review of observational studies. Arthritis Res Ther. 17:152. DOI: 10.1186/s13075-015-0670-x. PMID: 26050740. PMCID: PMC4483213.

3. Muraki S, Akune T, Oka H, Ishimoto Y, Nagata K, Yoshida M, et al. 2012; Incidence and risk factors for radiographic knee osteoarthritis and knee pain in Japanese men and women: a longitudinal population-based cohort study. Arthritis Rheum. 64:1447–56. DOI: 10.1002/art.33508. PMID: 22135156.

4. Kim I, Kim HA, Seo YI, Song YW, Jeong JY, Kim DH. 2010; The prevalence of knee osteoarthritis in elderly community residents in Korea. J Korean Med Sci. 25:293–8. DOI: 10.3346/jkms.2010.25.2.293. PMID: 20119586. PMCID: PMC2811300.

5. Yoo JJ, Kim DH, Kim HA. 2018; Risk factors for progression of radiographic knee osteoarthritis in elderly community residents in Korea. BMC Musculoskelet Disord. 19:80. DOI: 10.1186/s12891-018-1999-5. PMID: 29530005. PMCID: PMC5848445.

6. Kim HA, Koh SH, Lee B, Kim IJ, Seo YI, Song YW, et al. 2008; Low rate of total hip replacement as reflected by a low prevalence of hip osteoarthritis in South Korea. Osteoarthritis Cartilage. 16:1572–5. DOI: 10.1016/j.joca.2008.04.024. PMID: 18558502. PMCID: PMC4373077.

7. Lau EM, Lin F, Lam D, Silman A, Croft P. 1995; Hip osteoarthritis and dysplasia in Chinese men. Ann Rheum Dis. 54:965–9. DOI: 10.1136/ard.54.12.965. PMID: 8546528. PMCID: PMC1010061.

8. Peat G, Thomas MJ. 2021; Osteoarthritis year in review 2020: epidemiology & therapy. Osteoarthritis Cartilage. 29:180–9. DOI: 10.1016/j.joca.2020.10.007. PMID: 33242603.

9. Wallace IJ, Felson DT, Worthington S, Duryea J, Clancy M, Aliabadi P, et al. 2019; Knee osteoarthritis risk in non-industrial societies undergoing an energy balance transition: evidence from the indigenous Tarahumara of Mexico. Ann Rheum Dis. 78:1693–8. Erratum. DOI: 10.1136/annrheumdis-2019-215886. PMID: 31519654.

10. Losina E, Silva GS, Smith KC, Collins JE, Hunter DJ, Shrestha S, et al. 2020; Quality-adjusted life-years lost due to physical inactivity in a US population with osteoarthritis. Arthritis Care Res (Hoboken). 72:1349–57. DOI: 10.1002/acr.24035. PMID: 31350803. PMCID: PMC6982563.

11. Kasper DL, Fauci AS, Hauser SL, Longo DL, Jameson JL, Loscalzo J. 2015. Harrison's principles of internal medicine. 19th ed. McGraw Hill Education;New York: p. 2226.
12. Reynard LN, Barter MJ. 2020; Osteoarthritis year in review 2019: genetics, genomics and epigenetics. Osteoarthritis Cartilage. 28:275–84. DOI: 10.1016/j.joca.2019.11.010. PMID: 31874234.

13. Styrkarsdottir U, Lund SH, Thorleifsson G, Zink F, Stefansson OA, Sigurdsson JK, et al. 2018; Meta-analysis of Icelandic and UK data sets identifies missense variants in SMO, IL11, COL11A1 and 13 more new loci associated with osteoarthritis. Nat Genet. 50:1681–7. DOI: 10.1038/s41588-018-0247-0. PMID: 30374069.

14. Tachmazidou I, Hatzikotoulas K, Southam L, Esparza-Gordillo J, Haberland V, Zheng J, et al. 2019; Identification of new therapeutic targets for osteoarthritis through genome-wide analyses of UK Biobank data. Nat Genet. 51:230–6. DOI: 10.1038/s41588-018-0327-1. PMID: 30664745. PMCID: PMC6400267.

15. Ratneswaran A, Kapoor M. 2021; Osteoarthritis year in review: genetics, genomics, epigenetics. Osteoarthritis Cartilage. 29:151–60. DOI: 10.1016/j.joca.2020.11.003. PMID: 33227439.

16. Baird DA, Evans DS, Kamanu FK, Gregory JS, Saunders FR, Giuraniuc CV, et al. 2019; Identification of novel loci associated with hip shape: a meta-analysis of genomewide association studies. J Bone Miner Res. 34:241–51. DOI: 10.1002/jbmr.3605. PMID: 30320955. PMCID: PMC6375741.

17. Hatzikotoulas K, Roposch A, Shah KM, Clark MJ, Bratherton S, Limbani V, et al. 2018; Genome-wide association study of developmental dysplasia of the hip identifies an association with GDF5. Commun Biol. 1:56. DOI: 10.1038/s42003-018-0052-4. PMID: 30273415. PMCID: PMC6123669.
18. Sun Y, Wang C, Hao Z, Dai J, Chen D, Xu Z, et al. 2015; A common variant of ubiquinol-cytochrome c reductase complex is associated with DDH. PLoS One. 10:e0120212. DOI: 10.1371/journal.pone.0120212. PMID: 25848760. PMCID: PMC4388640.

19. Hindy G, Åkesson KE, Melander O, Aragam KG, Haas ME, Nilsson PM, et al. 2019; Cardiometabolic polygenic risk scores and osteoarthritis outcomes: a Mendelian randomization study using data from the Malmö Diet and Cancer Study and the UK Biobank. Arthritis Rheumatol. 71:925–34. DOI: 10.1002/art.40812. PMID: 30615301. PMCID: PMC6563114.

20. Funck-Brentano T, Nethander M, Movérare-Skrtic S, Richette P, Ohlsson C. 2019; Causal factors for knee, hip, and hand osteoarthritis: a Mendelian randomization study in the UK Biobank. Arthritis Rheumatol. 71:1634–41. DOI: 10.1002/art.40928. PMID: 31099188. PMCID: PMC6790695.
21. Zhou A, Morris HA, Hyppönen E. 2019; Health effects associated with serum calcium concentrations: evidence from MR-PheWAS analysis in UK Biobank. Osteoporos Int. 30:2343–8. DOI: 10.1007/s00198-019-05118-z. PMID: 31392400.

22. Magnusson K, Turkiewicz A, Englund M. 2019; Nature vs nurture in knee osteoarthritis - the importance of age, sex and body mass index. Osteoarthritis Cartilage. 27:586–92. DOI: 10.1016/j.joca.2018.12.018. PMID: 30634033.

23. Weldingh E, Johnsen MB, Hagen KB, Østerås N, Risberg MA, Natvig B, et al. 2019; The maternal and paternal effects on clinically and surgically defined osteoarthritis. Arthritis Rheumatol. 71:1844–8. DOI: 10.1002/art.41023. PMID: 31237417.

24. Richard D, Liu Z, Cao J, Kiapour AM, Willen J, Yarlagadda S, et al. 2020; Evolutionary selection and constraint on human knee chondrocyte regulation impacts osteoarthritis risk. Cell. 181:362–81.e28. DOI: 10.1016/j.cell.2020.02.057. PMID: 32220312. PMCID: PMC7179902.

25. Zhao T, Zhao J, Ma C, Wei J, Wei B, Liu J. 2020; Common variants in LTBP3 gene contributed to the risk of hip osteoarthritis in Han Chinese population. Biosci Rep. 40:BSR20192999. DOI: 10.1042/BSR20192999. PMID: 32452514. PMCID: PMC7284319.

26. Wang B, Sun Y, Liu H, Cao Y, Lei T. 2020; Evaluation of relationship between DNA methyltransferase 3 β gene and the risk of hip osteoarthritis: a case-control study based on a Han Chinese population. Int J Rheum Dis. 23:1404–11. DOI: 10.1111/1756-185X.13943. PMID: 32776659.

27. Chu M, Zhu X, Wang C, Rong J, Wang Y, Wang S, et al. 2017; The rs4238326 polymorphism in ALDH1A2 gene potentially associated with non-post traumatic knee osteoarthritis susceptibility: a two-stage population-based study. Osteoarthritis Cartilage. 25:1062–7. DOI: 10.1016/j.joca.2017.01.003. PMID: 28089900.

28. Hsueh MF, Önnerfjord P, Bolognesi MP, Easley ME, Kraus VB. 2019; Analysis of "old" proteins unmasks dynamic gradient of cartilage turnover in human limbs. Sci Adv. 5:eaax3203. DOI: 10.1126/sciadv.aax3203. PMID: 31633025. PMCID: PMC6785252.

29. Bedson J, Croft PR. 2008; The discordance between clinical and radiographic knee osteoarthritis: a systematic search and summary of the literature. BMC Musculoskelet Disord. 9:116. DOI: 10.1186/1471-2474-9-116. PMID: 18764949. PMCID: PMC2542996.

30. Hong JI, Park IY, Kim HA. 2020; Understanding the molecular mechanisms underlying the pathogenesis of arthritis pain using animal models. Int J Mol Sci. 21:533. DOI: 10.3390/ijms21020533. PMID: 31947680. PMCID: PMC7013391.

31. Neogi T, Felson D, Niu J, Nevitt M, Lewis CE, Aliabadi P, et al. 2009; Association between radiographic features of knee osteoarthritis and pain: results from two cohort studies. BMJ. 339:b2844. DOI: 10.1136/bmj.b2844. PMID: 19700505. PMCID: PMC2730438.

32. Wang K, Kim HA, Felson DT, Xu L, Kim DH, Nevitt MC, et al. 2018; Radiographic knee osteoarthritis and knee pain: cross-sectional study from five different racial/ethnic populations. Sci Rep. 8:1364. DOI: 10.1038/s41598-018-19470-3. PMID: 29358690. PMCID: PMC5777996.

33. Son KM, Hong JI, Kim DH, Jang DG, Crema MD, Kim HA. 2020; Absence of pain in subjects with advanced radiographic knee osteoarthritis. BMC Musculoskelet Disord. 21:640. DOI: 10.1186/s12891-020-03647-x. PMID: 32993609. PMCID: PMC7526196.

34. Yusuf E, Kortekaas MC, Watt I, Huizinga TW, Kloppenburg M. 2011; Do knee abnormalities visualised on MRI explain knee pain in knee osteoarthritis? A systematic review. Ann Rheum Dis. 70:60–7. DOI: 10.1136/ard.2010.131904. PMID: 20829200.

35. Aso K, Shahtaheri SM, Hill R, Wilson D, McWilliams DF, Walsh DA. 2019; Associations of symptomatic knee osteoarthritis with histopathologic features in subchondral bone. Arthritis Rheumatol. 71:916–24. DOI: 10.1002/art.40820. PMID: 30663865.

36. Zhu S, Zhu J, Zhen G, Hu Y, An S, Li Y, et al. 2019; Subchondral bone osteoclasts induce sensory innervation and osteoarthritis pain. J Clin Invest. 129:1076–93. DOI: 10.1172/JCI121561. PMID: 30530994. PMCID: PMC6391093.

37. Neogi T, Frey-Law L, Scholz J, Niu J, Arendt-Nielsen L, Woolf C, et al. Multicenter Osteoarthritis (MOST) Study. 2015; Sensitivity and sensitisation in relation to pain severity in knee osteoarthritis: trait or state? Ann Rheum Dis. 74:682–8. DOI: 10.1136/annrheumdis-2013-204191. PMID: 24351516. PMCID: PMC4062615.

38. Suri S, Gill SE, Massena de Camin S, Wilson D, McWilliams DF, Walsh DA. 2007; Neurovascular invasion at the osteochondral junction and in osteophytes in osteoarthritis. Ann Rheum Dis. 66:1423–8. DOI: 10.1136/ard.2006.063354. PMID: 17446239. PMCID: PMC2111605.

39. Meng W, Adams MJ, Palmer CNA, et al. 23andMe Research Team, Shi J, Auton A. 2019; Genome-wide association study of knee pain identifies associations with GDF5 and COL27A1 in UK Biobank. Commun Biol. 2:321. Erratum. DOI: 10.1038/s42003-020-0880-x. PMID: 32218487. PMCID: PMC7098975.
40. Boer CG, Hatzikotoulas K, Southam L, Stefánsdóttir L, Zhang Y, Coutinho de Almeida R, et al. 2021; Deciphering osteoarthritis genetics across 826,690 individuals from 9 populations. Cell. 184:4784–818.e17.
41. Kim JR, Kim HA. 2020; Molecular mechanisms of sex-related differences in arthritis and associated pain. Int J Mol Sci. 21:7938. DOI: 10.3390/ijms21217938. PMID: 33114670. PMCID: PMC7663489.

42. Sorge RE, Mapplebeck JC, Rosen S, Beggs S, Taves S, Alexander JK, et al. 2015; Different immune cells mediate mechanical pain hypersensitivity in male and female mice. Nat Neurosci. 18:1081–3. DOI: 10.1038/nn.4053. PMID: 26120961. PMCID: PMC4772157.

Fig. 1
Overview of steps for conducting OA GWAS. OA: osteoarthritis, GWAS: genomewide association study.

Table 1
Characterization of OA risk variants in Han Chinese populations




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download